

Review Unit 11, 12, & 13
Presentation
•
Chemistry
•
9th - 12th Grade
•
Hard
Standards-aligned
Ricardo Mora
FREE Resource
8 Slides • 30 Questions
1
Review Unit 11 (Gas Laws), 12 (Solutions), & 13 (Acids / Bases)
By Ricardo Mora
2

In this unit, you learned about how gasses behave when conditions change and when conditions are @STP. We also dealt with conditions that are IDEAL for gasses to react. The conditions (Variables) deal with pressure, volume, temperature, and number of moles of a gas.
Some text here about the topic of discussion.
Unit 11 Gas Laws
3
Multiple Choice
4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
7
Multiple Choice
8
Multiple Choice
9
Multiple Choice
The pressure of a 6.5 L sample of oxygen gas is measured to be 3.8 atm. If the gas is transferred into a 12 L tank, what is the new pressure?
6.6 atm
3.9 atm
4.5 atm
2.1 atm
10
Multiple Choice
11

In this unit, you learned about the concentration of solutions using Molarity (M=mol/liters)and how to dilute a concentrated solution by using M1V1=M2V2. We also discussed the properties of water and why water is THE BEAST in being able to dissolve solutes.
Some text here about the topic of discussion.
Unit 10: Solutions
12
Multiple Choice
13
Multiple Choice

14
Multiple Choice
15
Multiple Choice

16
Multiple Choice

17
Multiple Choice

The fact that water can stick to other substances is called:
Ph
Adhesion
Cohesion
Insulation
18
Multiple Choice

Heat Capacity is a measure of the heat required to raise the temperature of 1g of a substance by 1°C.
So...
True or False:
Water has a very high heat capacity, which means it resists changing temperature when heat is added or removed.
True
False
19
Multiple Choice
What is the molarity of 4 g of NaCl (MM=58.45g/mol) in 3,800 mL of solution?
0.018 M
0.0011 M
1.052 M
0.062 M
20
Multiple Choice
What is the molarity of 122.5 g of AlCl3 in 1.0 L of solution? (MM = 133 g/mol)
1.225 M
0.92 M
0.1225 M
21
Multiple Choice
If I have 340 mL of a 0.5 M NaBr solution, what will the concentration be if I add 560 mL more water to it?
.188 M
3.78 M
.389 M
1.76 M
22
Multiple Choice
If I dilute 250 mL of 0.10 M lithium acetate solution to a volume of 750 mL, what will the concentration of this solution be?
2 M
0.02 M
0.03 M
0.08 M
23

In this unit, you learned about the two different theories of an Acid and a Base. You were also shown how water can act like an acid or a base according to the BrΦnsted-Lowry definition.
Some text here about the topic of discussion.
Unit 13: Acids / Bases
24
25
Ants and bees have acids
Acids on your skin sting because they are corrosive and attack your body tissues. This is why lemon juice stings if you get it in a cut on your finger. Certain types of ants and bees sting because they inject you with formic acid.

26
Dilute and concentrated acids
Most fruits contain acids but you can eat them because the acid in them is very dilute. A dilute acid contains large amounts of water and a small amount of acid. Some of the acids that we use at school laboratories are concentrated acids. They contain large amount of acid and very little water.

27
Multiple Choice
A substance is found to have the following characteristics:
Very bitter taste
Feels slippery to the touch
Produces OH- ions when dissolved in water
In what category would the substance be classified?
acid
base
enzyme
fatty acid
28
Multiple Choice
HNO2
a base
a salt
an acid
an ionic compound
29
Multiple Choice
30
Multiple Choice
KOH is ...
an acid
a base
a salt
an ionic compound
31
Multiple Choice
An Arrhenius base:
donates H+
accepts H+
produces H+
produces OH-
32
Multiple Choice
An Arrhenius acid:
donates H+ to another substance
accepts H+ from another substance
produces H+
produces OH-
33
Multiple Choice
A Bronsted Lowry base:
donates H+ to another substance
accepts H+ from another substance
produces H+
produces OH-
34
Multiple Choice
According to Bronsted-Lowry, what is the definition of an BASE?
a substance that donates a hydrogen (H+) ion
a substance that donates a hydroxide (OH-) ion
a substance that accepts a hydrogen (H+) ion
a substance that accepts a hydroxide (OH-) ion
35
Multiple Choice
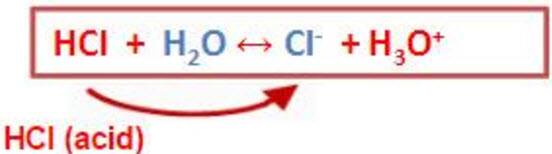
36
Multiple Choice
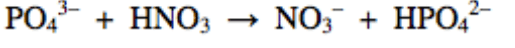
37
Multiple Choice
A sour taste is a characteristic of:
acids
bases
neutral
pH scale
38

I have truly enjoyed hanging out with you and teaching you a few things about Chemistry! Have an awesome summer and be safe!!!!! I will see you next year. :)
Some text here about the topic of discussion.
Thank you for being such awesome students!!
Review Unit 11 (Gas Laws), 12 (Solutions), & 13 (Acids / Bases)
By Ricardo Mora
Show answer
Auto Play
Slide 1 / 38
SLIDE
Similar Resources on Wayground

35 questions
Ionic Radius Trends and Multiple Ionization Energies
Presentation
•
10th - 12th Grade

32 questions
Types of Chemical Reactions
Presentation
•
9th - 12th Grade

33 questions
Standard 4 Exam Review
Presentation
•
9th - 12th Grade

31 questions
Doppler Effect
Presentation
•
9th - 12th Grade

30 questions
Oxidation and Chemical Formulas
Presentation
•
9th - 12th Grade

34 questions
Stoichiometry Review
Presentation
•
10th - 12th Grade

29 questions
Interpreting Heating Curves
Presentation
•
9th - 12th Grade

29 questions
Isotopes & Radioactivity
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade