

Interpreting Heating Curves
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
+2
Standards-aligned
Amy Dahl
Used 74+ times
FREE Resource
11 Slides • 18 Questions
1
Interpreting Heating Curves
By Amy Dahl
2
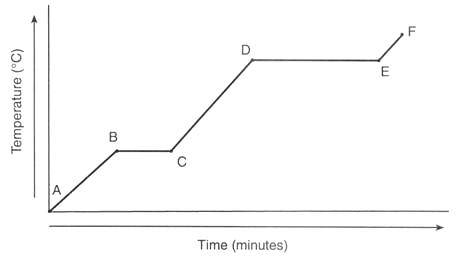
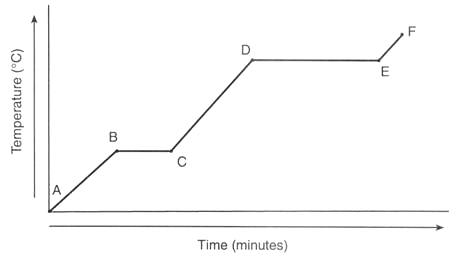
It shows a plot of temperature at various times as a substance is heated.
It is notable for having two "flat spots" where the temperature remains constant even though heat continues to be added.
A heating curve looks like this

3
You may not instantly know the answer
Use what you know to figure out the answer!
The "flat spots" are the key. We can use them to reason out the answer to any question having to do with heating curves.
Some text here about the topic of discussion
4

The vertical axis is temperature. So the higherr the curve gets, the higher the temperature.
Temperature is a measure of particle speed. The higher the temp, the faster they move.
Some text here about the topic of discussion.
First, notice the axes
5

The horizontal axis is heat added (sometimes it is labeled time, but you know it is being heated the entire time).
The farther you go to the right, the more energy has been added.
Some text here about the topic of discussion.
First, notice the axes
6

We expect temperature to go up when the substance is being heated.
But from B-C and D-E it doesn't!
This is because temperature does not change when the phase is changing.
So during flat spots, the phase is changing. Always.
Some text here about the topic of discussion.
Next, the flat spots!
7

Use logic to determine which flat spot is which.
Phase changes that occure when something gets heated are melting and vaporization.
You know melting occurs at a lower temp than vaporization.
Thus B-C is melting, D-E is vaporization.
Some text here about the topic of discussion.
Which is which?
8

This heating curve, for water, shows water's melting point of 0 oC and its boiling point of 100 oC.
Other substances have different melting and boiling points. Just read them off the temp scale.
Some text here about the topic of discussion.
Melting and boiling points
9

Since melting is a change from solid to liquid, before melting you have a solid, and after melting you have a liquid.
So A-B is solid and C-D is liquid.
(during B-C both solid and liquid are present).
Since A-B and C-D slope upwards, T increases and we say they are warming up!
Some text here about the topic of discussion.
More logic
10

Since vaporization is a change from liquid to gas, before vaporization you have a liquid, and after vaporization you have a gas (vapor).
So C-D is liqulid and E-F is gas.
(during D-E both liquid and vapor are present).
Like A-B and C-D, E-F slopes upwards, so the gas gets hotter with time and we say it is warming up.
Some text here about the topic of discussion.
Similarly
11
Let's try some!
Some text here about the topic of discussion.
12
Multiple Choice
Vaporizing
13
Multiple Choice
14
Multiple Choice
15
Multiple Choice

Vaporizing
16
Multiple Choice

Vaporizing
17
Multiple Choice

Vaporizing
18
Multiple Choice

19
Multiple Choice

20
Multiple Choice

21
Multiple Choice

22
Multiple Choice

In which segment is the particle speed remaining constant?
23
Multiple Choice

24
Multiple Choice

25
Multiple Choice
26
Multiple Choice
27
Multiple Choice

From point A to point E, the sample is going through an
exothermic process by releasing heat to the surroundings
exothermic process by absorbing heat from the surroundings
endothermic process by releasing heat to the surroundings
endothermic process by absorbing heat from the surroundings
28
Multiple Choice

The melting point of the sample is
-60 ºC
-100 ºC
60 ºC
100 ºC
29
Multiple Choice
Interpreting Heating Curves
By Amy Dahl
Show answer
Auto Play
Slide 1 / 29
SLIDE
Similar Resources on Wayground

25 questions
Separating a Mixture
Presentation
•
9th - 12th Grade

20 questions
DLD Performance Final
Presentation
•
9th - 12th Grade

22 questions
Debate Vocabulary
Presentation
•
9th - 12th Grade

22 questions
Dependent and Independent Clauses
Presentation
•
9th - 12th Grade

27 questions
Acids & Bases - Part 2
Presentation
•
9th - 12th Grade

24 questions
History of the Periodic Table
Presentation
•
9th - 12th Grade

20 questions
Atomic Structure Review
Presentation
•
9th - 12th Grade

20 questions
Water, Solubility, Solutions, Molarity
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Unit 7 Chemical Reactions
Quiz
•
10th Grade

25 questions
Unit 8 Stoichiometry Review
Quiz
•
10th Grade

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

15 questions
Nuclear Chemistry: Balancing
Quiz
•
10th Grade

20 questions
Naming Acids and Bases
Quiz
•
10th Grade

10 questions
Exploring Stoichiometry in Chemistry
Interactive video
•
6th - 10th Grade