

Entropy
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
Standards-aligned
Thomas Pickett
Used 112+ times
FREE Resource
1 Slide • 11 Questions
1
Entropy
By Kaye Pickett
2
Multiple Choice
Entropy is a measure of
accuracy
precision
the disorder of a system
the attraction of a nucleus for an electron
3
Multiple Choice
Which of these processes represents a decrease in entropy?
sublimation of CO2
boiling water
condensation of steam on cold glass
NaCl dissolving in water
4
Multiple Choice
Which of these reactions shows a DECREASE in entropy?
CaCO3(s) → CaO(s) + CO2(g)
3O2(g) → 2O3(g)
2NH3(g) → 3H2(g) + N2(g)
C6H6(l) → C6H6(g)
5
Multiple Choice
The entropy will usually increase when
a molecule is broken into two or more smaller molecules
a reaction occurs that results in an increase in the number of moles of gas
a solid changes to a liquid
all of these
6
Multiple Choice
Which sample has the lowest entropy?
1 mole of KNO3(l)
1 mole of KNO3(s)
1 mole of H2O(l)
1 mole of H2O(g)
7
Multiple Choice
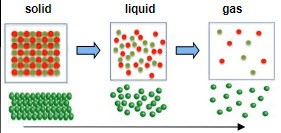
Entropy increases from solid, liquid to gas. Why?
Molecular disorder increases
Molecules increase in number from solid to gas
Molecules are heavier in solid
Molecules are more reactive
8
Multiple Choice
Which phase change represents a decrease in entropy?
solid to liquid
gas to liquid
liquid to gas
solid to gas
9
Multiple Choice
What phase of matter has the most entropy?
solid
liquid
gas
10
Multiple Choice
If Ice melts, its entropy increases.
Trulala
Falsemama
It will not change. So... none of the above.
11
Multiple Choice
Which of these reactions shows a DECREASE in entropy?
CaCO3(s) → CaO(s) + CO2(g)
3O2(g) → 2O3(g)
2NH3(g) → 3H2(g) + N2(g)
C6H6(l) → C6H6(g)
12
Multiple Choice
What is a spontaneous process ?
slow process
fast process
process that needs an external intervention to occur
process that does not need external intervention to occur / keep happening
Entropy
By Kaye Pickett
Show answer
Auto Play
Slide 1 / 12
SLIDE
Similar Resources on Wayground

7 questions
Ch. 10.3 - Percent Composition and Chemical Formulas
Presentation
•
9th - 11th Grade

6 questions
Specific Heat + Unit 1 Performance Task
Presentation
•
9th - 12th Grade

12 questions
Midpoint
Presentation
•
9th - 12th Grade

8 questions
Ideal Gas Law
Presentation
•
10th - 12th Grade

11 questions
Multistep Stoichiometry
Presentation
•
10th - 12th Grade

10 questions
Engagement Questions
Presentation
•
9th - 12th Grade

7 questions
Heating/Cooling Curves Conceptual
Presentation
•
9th - 12th Grade

10 questions
Ch. 3.4 - Density
Presentation
•
9th - 11th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade