

Atoms, Periodic Table, Ions and Bonding
Presentation
•
Science
•
9th Grade
•
Hard
+3
Standards-aligned
Kate NICHOLLS
Used 7+ times
FREE Resource
1 Slide • 25 Questions
1
Atoms, Periodic Table, Ions and Bonding
By Kate NICHOLLS
2
Multiple Choice
Subatomic particle with a negative charge, has very little mass compared to other particles
Proton
Neutron
Electron
Alpha Particle
3
Multiple Choice
Subatomic particle with positive charge identifies the atom
Proton
Neutron
Electron
Alpha Particle
4
Multiple Choice
Atomic mass is equal to the number of protons plus _______.
Protons
Neutrons
Electrons
Alpha particles
5
Multiple Choice
An atom with high ionization energy has 7 valence electrons, when this atom gains an electron, it becomes a __________ ion.
Positive
Negative
Neutral
Covalent
6
Multiple Choice
_________ give away electrons to become positive ions and __________ steal electrons to become negative ions.
Metals, metals
Nonmetals, nonmetals
Nonmetals, metals
Metals, nonmetals
7
Multiple Select
Which combination of elements form ionic bonds? Select all that apply.
metal and nonmetal
nonmetal and nonmetal
two of the same elements
two elements with electronegativity difference > 1.7
8
Multiple Choice
What part of the atom does bonding involve?
protons
neutrons
all electrons
valence electrons
9
Multiple Choice
How many valence electrons do MOST atoms need to be stable?
6
8
3
5
10
Multiple Choice
Which of the following is the correct ionic formula for calcium sulfide?
Ca2S2
CaS
S2Ca2
SCa
11
Multiple Choice
Name the compound from it's formula: BaI2
Iodine barium
Iodine bariumide
Barium iodine
Barium iodide
12
Multiple Choice
What is the charge for each iron atom (Fe) in Fe2O3:
13
Multiple Choice
How many valence electrons does Calcium have?
1
2
3
4
5
14
Multiple Choice
How many valence electrons does Sulfur have?
4
5
6
7
8
15
Multiple Choice
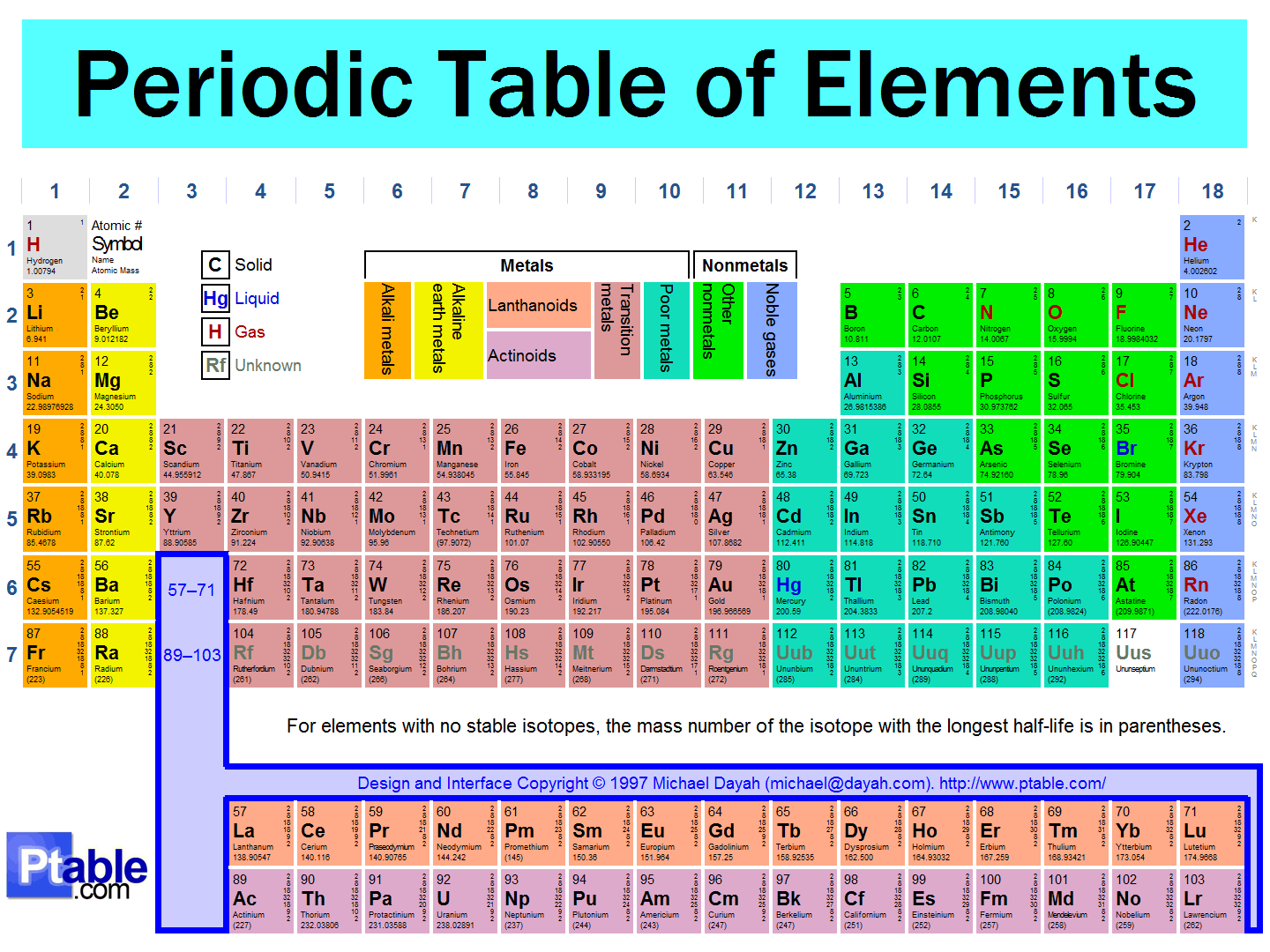
16
Multiple Choice
Magnesium Hydroxide is an ionic compound made up from Mg2+ and OH- ions. Which is its correct formula?
MgOH
MgOH2
Mg(OH)2
MgO
17
Multiple Choice

18
Multiple Choice
Which type of ion results from the loss of electrons and has a positive charge?
cation
anion
19
Multiple Choice
Which of the following groups on the Periodic Table will only form ions with positive charges?
Group 2
Group 15
Group 17
Group 18
20
Multiple Choice
A compound is
anything that has mass
any element that cannot be broken down into a simpler substance.
a substance consisting of two or more different elements in a fixed ratio
any element on the periodic table
21
Multiple Choice
What is a group?
There are seven in the periodic table, with each one beginning at the far left.
A horizontal row of the periodic table.
A vertical column on the periodic table
Begins when a new principal energy level begins filling with electrons.
22
Multiple Select
Metals (select all that apply)
are found on the right side of the periodic table.
are found on the left side of the periodic table.
are good conductors of heat and electricity.
are brittle.
are malleable and ductile.
23
Multiple Choice

Which element is found in group 2, period 6?
Oxygen (O)
Barium (Ba)
Selenium (Se)
Carbon (C)
24
Multiple Choice
25
Multiple Choice
26
Multiple Choice
Atoms, Periodic Table, Ions and Bonding
By Kate NICHOLLS
Show answer
Auto Play
Slide 1 / 26
SLIDE
Similar Resources on Wayground

21 questions
Sound Waves
Presentation
•
8th - 9th Grade

20 questions
Sustainability Lesson with Videos
Presentation
•
9th Grade

20 questions
States of Matter Review
Presentation
•
3rd Grade

20 questions
Earth's Compositional Layers
Presentation
•
9th Grade

18 questions
Waves
Presentation
•
9th Grade

20 questions
Deep water Horizon
Presentation
•
9th Grade

21 questions
acids and bases
Presentation
•
8th - 9th Grade

21 questions
Types of Fossils
Presentation
•
KG - 8th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

11 questions
EOC Domain 1: Cells
Presentation
•
9th - 10th Grade

14 questions
EOC Domain 2: Genetics
Presentation
•
9th - 10th Grade

10 questions
Exploring the Human Respiratory System
Interactive video
•
6th - 10th Grade

10 questions
Exploring Newton's Laws of Motion
Interactive video
•
6th - 10th Grade