

Lesson: Solutions, Acids, and Bases
Presentation
•
Science, Chemistry
•
9th Grade
•
Medium
Standards-aligned
William Jared Lovering
Used 11+ times
FREE Resource
22 Slides • 15 Questions
1

2

3

4

5

6

7

8

9

10

11

12

13

14

15

16

17

18

19

20

21

22

23
Multiple Choice
When found in food, acids often taste ________.
Sour
Bitter
Salty
Slippery
24
Multiple Choice
When found in food, bases often taste _______.
Sour
Bitter
Salty
Soggy
25
Multiple Choice
Four solutions are tested and found to have the following pH values. Which of these substances would be considered the weakest acid?
Lemon Juice - pH=2
Apple Juice - pH=4.5
Shower Cleaner - pH=5.5
Hydrogen Peroxide - pH=7
26
Multiple Choice
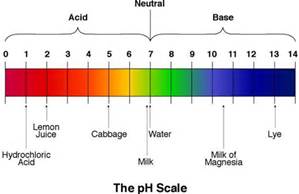
27
Multiple Choice
NaOH may be found in drain cleaners and as a component of soaps. Is NaOH an acid or a base?
Acid
Base
Neither (Neutral)
28
Multiple Choice
A substance doing the dissolving, usually the part of a solution present in the larger amount
solvent
solute
solution
solubility
29
Multiple Choice
a substance being dissolved, usually the part of the solution in the lesser amount
solvent
solute
solubility
concentration
30
Multiple Choice
a substance that vigorously reacts with most metals to produce hydrogen; it readily conducts electricity; it turns litmus red and has a pH of less than 7; it typically is corrosive or sour-tasting
acid
base
neutral
solution
31
Multiple Choice
a substance that does not react with most metals or readily conduct electricity; it turns litmus blue and has a pH of greater than 7; it typically feels slippery and is bitter-tasting
acid
base
neutral
solution
32
Multiple Choice
a substance that has a pH of 7; water is an example
acid
base
neutral
concentration
33
Multiple Choice
indicator that turns red in the presence of an acid and blue in the presence of a base
solvent
solute
litmus
temperature
34
Multiple Choice
describes how much solute has been dissolved in a solution and is expressed in some kind of ratio
conductivity
solute
concentration
solubility curve
35
Multiple Choice
the measure of a solution's ability to conduct electricity
concentration
conductivity
solubility curve
temperature
36
Multiple Choice

The picture shows how you might change an variable in an experiment about what affects the rate of dissolving. What results do you expect to see?
A will dissolve slowest because it has the largest number of particles.
D will dissolve slowest because it has the smallest surface area.
All pieces will dissolve at an equal rate.
C will dissolve slowest because it has the most bonds between sugar molecules.
37
Multiple Choice
What does a neutralization reaction produce?
acids
bases
water and a salt
carbonated water

Show answer
Auto Play
Slide 1 / 37
SLIDE
Similar Resources on Wayground

34 questions
Lab Safety
Presentation
•
9th Grade

30 questions
Chemical Equation
Presentation
•
8th Grade

33 questions
What are Ecosystems?
Presentation
•
9th Grade

33 questions
CHEMICAL ENERGETICS
Presentation
•
9th Grade

33 questions
C9: Chem Calculation (Concentration)
Presentation
•
9th Grade

34 questions
PhySci Practice: Naming Compounds
Presentation
•
9th Grade

31 questions
Half Life Calculations Practice
Presentation
•
9th Grade

31 questions
Catch the Wave What are Waves
Presentation
•
8th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

40 questions
NCFE Earth and Environmental Science Released Test
Quiz
•
9th - 12th Grade

10 questions
Exploring Weather Influences and Map Reading
Interactive video
•
6th - 10th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

61 questions
End of the Year Trivia Fun
Quiz
•
9th Grade

15 questions
Protein synthesis
Quiz
•
9th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade