

CHEMICAL ENERGETICS
Presentation
•
Chemistry
•
9th Grade
•
Hard
Sharidah Nadhirah
Used 9+ times
FREE Resource
33 Slides • 0 Questions
1
CHAPTER 8: CHEMICAL ENERGETICS
Chemical & Energy

2
Chemicals
???
3
Energy
???
4
AB + C = AC + B
???
5
When chemical reaction takes place
Energy is being absorbed/released
energy & chemical reaction
6
Lets sketch how energy is being absorbed and released during a chemical reaction..
pens and paper..
7

8

Energy will start to transfer to or from the environment (surrounding)?
9
When chemical reaction takes place and energy is released to the environment
the process is called..
10
EXOTHERMIC
EXIT
11
When chemical reaction takes place and energy is absorbed from the environment
it is called
12
ENDOTHERMIC
ENTER
13
HOW CAN WE REPRESENT ENERGY COMING IN AND GOING OUT??
ENERGY LEVEL DIAGRAM
14
ENERGY LEVEL DIAGRAM
A diagram showing the energy change of the reaction on vertical axis and the reactants and products on the horizontal axis.
15
WHY IS THERE ENERGY CHANGE?
What is happening in a chemical reaction??
16
17
Lets draw the energy level diagram
IMPORTANT!!!!
18
Lets recap
exo & endo
19
20
EXOTHERMIC (EXIT)
Gives out energy
Surrounding temperature rise/increase
The reaction can be described as: reactants = products + energy
formation of new bonds

21
EXOTHERMIC (eg:)
combustion of fuels (O2 + gas)
neutralization (acid + base)
respiration in our body

22
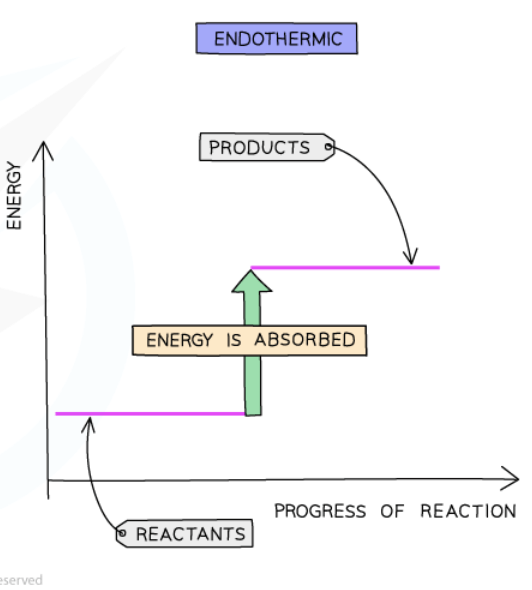
ENDOTHERMIC(ENTER)
Take in energy from the surrounding
Surrounding temperature fall
The reaction can be described as: reactant + energy = products
Since energy is absorbed products must have higher energy than reactants.
Bonds breaking
23
ENDOTHERMIC (eg)
reactions that take place in cooking
photosynthesis: plants absorbed sunlight energy to convert CO2 + H2O = Glucose
ice melting

24
9.2
MAKING & BREAKING BONDS
25
MAKING & BREAKING BONDS
In a chemical reactions, bonds must first be broken. Then new bonds form. Breaking bonds takes in energy. Making bonds releases energy.
26
27
28
BOND ENERGIES
...is the energy needed to make or break bonds.
Energy needed (enter) to break bonds. (Endothermic = absorbed)
Energy given out (exit) when making bonds (Exothermic = released)
29
Bond energy = (kJ/mole)
units
30

31

32
BOND ENERGY CALCULATION
Each chemical bond has a specific bond energy
This is the amount of energy require to break or form the bond
This energy can be used to calculate how much heat would be released or absorbed in a reaction
Identify bond present in reactants & products

33
LETS DO CALCULATION
CALCULATOR!!
CHAPTER 8: CHEMICAL ENERGETICS
Chemical & Energy

Show answer
Auto Play
Slide 1 / 33
SLIDE
Similar Resources on Wayground

24 questions
Chemical Formulas, Equations, & Reactions Post Review
Presentation
•
8th Grade

26 questions
Cell Membrane and Transport
Presentation
•
9th Grade

26 questions
Circuits and Symbols
Presentation
•
9th - 12th Grade

26 questions
DNA structure and replication
Presentation
•
9th Grade

27 questions
Ecological Relationships
Presentation
•
9th Grade

29 questions
Ionic Bonding
Presentation
•
10th Grade

28 questions
Ch 18 Ionic Bonds
Presentation
•
9th Grade

30 questions
Chemical Equation
Presentation
•
8th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Le Chatelier's Principle
Quiz
•
9th - 12th Grade

6 questions
DOL: Endothermic and Exothermic Reactions
Quiz
•
9th - 11th Grade

12 questions
Rate of reaction
Quiz
•
8th - 9th Grade

13 questions
Nuclear Chemistry
Presentation
•
9th - 12th Grade

5 questions
DOL: Ideal Gas Law
Quiz
•
9th - 12th Grade

40 questions
Thermochemistry Review
Quiz
•
9th - 12th Grade

13 questions
Acid Base - Neutralization Reactions
Quiz
•
9th - 12th Grade