

1st Semester Review-Plummer
Presentation
•
•
Medium
Stephanie Plummer
Used 6+ times
FREE Resource
5 Slides • 230 Questions
1
Match
Group 18
Group 1
Group 2
Groups 3-12
Group 17
Noble Gases
Alkali Metals
Alkaline Earth Metals
Transition metals
Halogens
Noble Gases
Alkali Metals
Alkaline Earth Metals
Transition metals
Halogens
2
Match
Elements
Molecules
Compounds
Mixture
Contain only one type of atom
at least 2 atoms combined together
At least 2 DIFFERENT atoms combined together
2 or more substances that are NOT chemically combined
Contain only one type of atom
at least 2 atoms combined together
At least 2 DIFFERENT atoms combined together
2 or more substances that are NOT chemically combined
3
Multiple Choice

This periodic table shows elements 1 and 2 if element 1 has an atomic number of 19 what is the atomic number of element 2 ?
20
28
30
34
4
Match
Located in the nucleus; have a positive charge; and a mass of 1 amu
Located outside of the nucleus; have a negative charge; and a mass of 1/2000th of an amu
located in the nucleus; have no charge; and no charge
Protons
electrons
neutrons
Protons
electrons
neutrons
5
Multiple Choice
Which of the following is a compound
water
hydrogen
nitrogen gas
oxygen
6
Multiple Choice
Which of the following is a compound and a molecule
O2
H2
Ca
CH
7
Fill in the Blanks
Type answer...
8
Fill in the Blanks
Type answer...
9
Multiple Choice
elements are placed in the periodic table based on the number of
neutrons
electrons
protons
protons + neutrons
10
Reorder
Put the following in order
reactants
--> (yields)
products
11
Fill in the Blanks
Type answer...
12
Multiple Choice
The number of atoms of each element must be equal on both sides of a reaction.
True
False
13
Multiple Choice
Methane + 2O2 --> CO2 + 2H2O
what is the chemical formula for methane based on this equation? (hint: remember that the number of atoms of each elements has to match on both sides)
C2H4
CH4
CH4O
CH2
14
Multiple Choice
which of the following is not an example of a chemical change?
Burning wood
sawing wood
baking a cake
digesting food
15
Multiple Choice
which of the following is not an example of a physical change?
Mixing and pouring concrete
Melting ice
burning wood
tearing paper
16
Match
Periods
Groups
Families
Horizontal rows on the periodic table
vertical columns on the periodic table
The specific names for the group of elements within a vertical column on the periodic table
Horizontal rows on the periodic table
vertical columns on the periodic table
The specific names for the group of elements within a vertical column on the periodic table
17
Multiple Choice

The following image represents
Einstein Model
Bohr's Model
Roy's Model
The Model
18
Multiple Choice

Elements are organized on the
periodic table
element table
hypothesis
experiment table
19
Multiple Choice

As we go from left to right on the periodic table - elements tend to become more __.
+
o/neutral
+/-
-
20
Multiple Choice
All of the following are an example of a subatomic particle except
valence
proton
neutron
electron
21
Multiple Choice

As we go from right to left on the periodic table electronegativity decreases
true
false
22
Multiple Choice
When people make inferences they are not using their senses such as: (smell/taste/hearing).
true
false
23
Multiple Choice

These elements are described as: shiny and great conductors of heat + electricity
metalloid
non metals
metals
halogens
24
Multiple Choice
The following are also called semiconductors.
metalloids
halogens
metal
non metal
25
Multiple Choice

Groups on the periodic table go from left to right and are organized by the number of electrons that are in the outside shell
false
true
26
Multiple Choice
Matter is made of elements, elements are made of ____s the smallest and are subatomic.
matter
elements
protons
atoms
27
Multiple Choice
Protons and ___ take up the atomic mass
neutrons
electrons
valence
variation
28
Multiple Choice

What is located in the nucleus of an atom?
protons and neutrons
protons and electrons
electrons and neutrons
29
Multiple Choice
How is the Periodic Table arranged?
alphabetical order
atomic number
the year the element was discovered
Albert Einstein decided how to arrange it.
30
Multiple Choice
What is located on the horizontal rows on the Periodic Table?
families
groups
periods
31
Multiple Choice
In which direction would you find groups/families?
horizontal
vertical
32
Multiple Choice
How many different types of atoms make up an element?
1
2
3
33
Multiple Choice
How are symbols for elements written?
numbers
pictures
letters
34
Multiple Choice
What is the symbol for carbon?
Ca
C
Na
35
Multiple Choice
H is the symbol for____.
Helium
Hydrogen
Iron
36
Multiple Choice
What is the charge of a proton?
positive
negative
neutral
37
Multiple Choice
What is the charge of a neutron?
positive
neutral
negative
38
Multiple Choice
Negative is the charge for _____.
protons
neutrons
electrons
39
Multiple Choice
What is a subscript found in a formula?
letter
number that tells how many atom are needed
star
40
Chemistry Unit Review

Atoms, Periodic Table, Formulas, and Changes
41

Parts Of The Atom
42
Multiple Choice
43
Multiple Choice
44
Multiple Choice
45
Multiple Choice
46
Multiple Choice
47
Multiple Choice
48
Multiple Choice
49
Multiple Choice
50
Multiple Choice

51
Multiple Choice

52
Multiple Choice

53
Multiple Choice

54
Multiple Choice

55
Multiple Choice
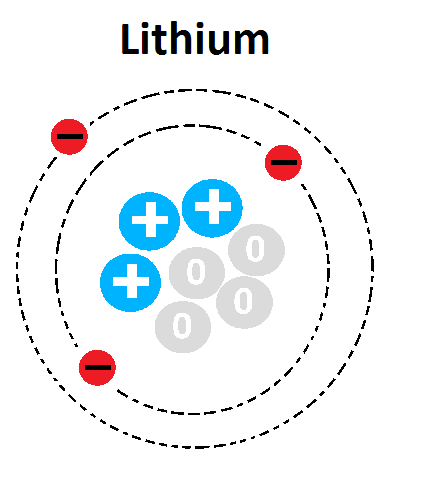
What is the mass number of this atom?
1
3
4
7
56
Multiple Choice

Name the element
Boron
Carbon
Nitrogen
Oxygen
57
Multiple Choice

58

The Periodic Table
59

The GROUP an atom is in tells you the number of VALENCE ELECTRONS and how it will REACT
The PERIOD an atom is in tells you how many ENERGY LEVELS an atom has.
60
Multiple Choice
61
Multiple Choice

62
Multiple Choice
63
Multiple Choice
64
Multiple Choice
65
Multiple Choice
66
Multiple Choice
67
Multiple Choice
68
Multiple Choice
69
Multiple Choice
70
Multiple Choice
71
Multiple Choice
72
Multiple Choice
73
Multiple Choice
74
Multiple Choice
75
Multiple Choice

76
Multiple Choice

77
Multiple Choice

78
Multiple Choice

79
Multiple Choice
80
Multiple Choice
81

Chemical Formulas And Reactions
82
Multiple Choice

83
Multiple Choice
84
Multiple Choice

85
Multiple Choice

86
Multiple Choice

87
Multiple Choice

88
Multiple Choice

89
Multiple Choice

90
Multiple Choice

91
Multiple Choice
92
Multiple Choice
Al + O2 --> 2Al2O3
93
Multiple Choice
94
Multiple Choice
2H2 + O2 --> 2H20
5g 20g ?
95
Multiple Choice
H2 + O2 → H2O
96
Multiple Choice
97
Multiple Choice
98
Multiple Choice
99
Multiple Choice
CaO + 3 C → CaC2 + CO
100
Multiple Choice
HCl + Zn → H2 + ZnCl2
101
Multiple Choice
102
Multiple Choice
103
Multiple Choice
Which is an example of a heterogeneous mixture?
sugar water
brass
table salt
salad
104
Multiple Choice
A pure substance that cannot be broken down into any other substances
atom
element
compound
mixture
105
Multiple Choice
Smallest and basic particle of a pure substance
element
atom
compound
mixture
106
Multiple Choice
A pure substance made of two or more elements chemically combined in a set ratio
element
atom
compound
mixture
107
Multiple Choice
A combination of two or more substances that are not chemically combined
element
atom
compound
mixture
108
Multiple Choice
A mixture in which different materials can be easily distinguished
heterogeneous mixture
homogeneous mixture
chemical formula
molecule
109
Multiple Choice
Shows the elements in the compound and the ratio of atoms
heterogenous mixture
homogenous mixture
chemical formula
molecule
110
Multiple Choice
A group of atoms bonded together
chemical formula
chemical bond
molecule
solution
111
Multiple Choice
A mixture that forms when one substance dissolves another
molecule
solution
pure substance
compound
112
Multiple Choice
Matter that always has exactly the same composition
molecule
solution
pure substance
Chemistry
113
Multiple Choice
The study of the properties of matter and how matter changes
Changeology
Chemistry
Mastery
Biology
114
Multiple Choice
What is CO2 an example of?
Element
Compound
Atom
Mixture
115
Multiple Choice
Lemonade consists of several substances that are NOT chemically combined, so lemonade is classified as
pure substance
a compound
a mixture
an element
116
Multiple Select
What are examples of chemical formulas?
H2O
C12H22O11
Salad
Milk
Lemonade
117
Multiple Choice
CO2, NaCl, and HCl may all be classified as
compounds and pure substances.
elements and pure substances.
elements and molecules.
elements and compounds.
118
Multiple Choice
The chemical formula of glucose is C6H12O6, so it is classified as a(n)
element.
mixture.
solution.
pure substance.
119
Multiple Choice
One difference between mixtures and pure substances is that
mixtures can be physically separated.
mixtures are made of one type of atom.
pure substances have no chemical bonds.
pure substances can be physically separated.
120
Multiple Choice
Suppose you have a container filled with iron and sand. You can separate the iron from the sand if you ____________ so this is a _________.
use a magnet; mixture
melt the sand; mixture
add baking soda; chemical compound
dissolve the sand in water; solution
121
Multiple Choice
In the classification of matter, how would we classify air?
pure mixture
pure substance
homogeneous mixture
heterogeneous mixture
122
Multiple Choice

The table lists the chemical formulas or symbols of a few common substances. Which two substances are compounds?
aluminum and iron
iron and sodium chloride
carbon dioxide and aluminum
sodium chloride and carbon dioxide
123
Multiple Choice

Ms. Roberts asked Tim to put the label "compound" on the correct jar in the picture. Which of the four substances is a compound?
chlorine
nitrogen
sodium
water
124
Multiple Choice
Water, H2O, is made up of different elements, hydrogen and oxygen. Is this type of combination a mixture or a pure substance?
Because water is composed of two separate elements, it is a mixture.
Because water can be heated and fairly easily turned to steam, it is a mixture.
Because water is composed of elements found in the periodic table, it is a pure substance.
Because water molecules all have a consistent ratio of hydrogen to oxygen, it is a pure substance.
125
Multiple Choice
What is a force of attraction between two atoms?
mixture
compound
chemical bonding
chemical property
126
Multiple Choice
What chart is used to organize the elements by atomic number?
Atomic Table
Periodic Table
Kitchen Table
127
Multiple Choice
What is an atom?
Smallest particle of a substance
Raw materials made of only one kind of atom
128
Multiple Choice
What is a compound?
Substances with molecules that have atoms from different elements
Two or more atoms joined together
129
Multiple Choice
What is a neutron?
Particle in an atom with a POSITIVE charge
Particle in an atom with a NEGATIVE charge
Particle in an atom with a NO charge
130
Multiple Choice
What is a proton?
Particle in an atom with a POSITIVE charge
Particle in an atom with a NEGATIVE charge
Particle in an atom with a NO charge
131
Multiple Select
Which particles are located in the nucleus?
Electrons
Neutrons
Protons
132
Multiple Choice
The number of protons is also equal to which number?
Atomic Number
Phone Number
Prime Number
133
Multiple Choice

Which element has 11 protons?
Sodium
Magnesium
Potassium
Calcium
134
Multiple Choice
What makes up an atom?
Protons, Neutrons, Electrons
Aluminum, Carbon, Neon
Solid, Liquid, Gas
Metals, Non-metals, Metalloids
135
Multiple Choice
What particle is located in the nucleus and has a positive charge?
Protons
Electrons
Neutrons
136
Multiple Choice
Which particle is located in the nucleus and has a neutral charge?
Protons
Electrons
Neutrons
137
Multiple Choice
What particle is found OUTSIDE orbiting the nucleus?
Protons
Electrons
Neutrons
138
Multiple Choice
An atom is the smallest unit of matter.
Agree
Disagree
139
Multiple Choice
140
Multiple Choice

141
Multiple Choice
You should NEVER wear safety goggles when working with chemicals.
Agree
Disagree
142
Multiple Choice

143
Multiple Choice

This model represents an element in the Periodic Table. Which element does this atomic model represent?
144
Multiple Choice
Substances that are made up of ONLY one type of atom (element) or compound.
Heterogeneous mixture
Pure substance
Suspension
Homogeneous mixture
145
Multiple Choice
146
Multiple Choice

The particle diagram shown is a:
pure substance
mixture
147
Multiple Choice

148
Multiple Choice
149
Multiple Choice
150
Multiple Choice
151
Multiple Choice
152
Multiple Choice

153
Multiple Choice
154
Multiple Choice
What is at the center of every atom?
nuclear
nucleus
electron
molecule
155
Multiple Choice
An electron of an atom...
lives inside the nucleus and has a neutral charge.
circles the nucleus and has a positive charge.
lives inside the nucleus and has a negative charge.
circles the nucleus and has a negative charge.
156
Fill in the Blanks
Type answer...
157
Fill in the Blanks
Type answer...
158
Multiple Choice
A subscript...
the number of protons in an atom.
the number of molecules present.
shows the number of atoms of a specific element in a compound.
the number an element is on the periodic table.
159
Multiple Choice
How many different elements are present in H2COCH2
5
4
3
2
160
Multiple Choice
Calculate the number of Oxygen atoms in Al(OH)3
1
2
3
4
161
Multiple Choice
The ______ are numbers found on the lower right-hand side of an element symbol.
Coefficient
Exponent
Multiplier
Subscript
162
Multiple Choice

How many NEUTRON are there in this Hydrogen?
2
1
0
163
Multiple Choice
What makes up the nucleus?
Protons + Neutrons
Electrons+ Protons
Neutrons + Electrons
164
Multiple Choice
What are three subparticles of an atom?
Proton, Neutron, Electron
Hydrogen, Oxygen, Helium
One Oxygen and two Hydrogen. H2O
165
Multiple Choice
How many different elements are present in H2COCH2
5
4
3
2
166
Multiple Choice
Calculate the number of Oxygen atoms in Al(OH)3
1
2
3
4
167
Multiple Choice
How many atoms are there TOTAL in H2SO4 ?
6
5
7
3
168
Multiple Choice
What does the subscript in a chemical formula tell you?
How many molecules are present
How many atoms are present
How many elements are present
How many neutrons are present
169
Multiple Choice
How many elements are in this chemical formula? NaHCO3
1
2
3
4
170
Multiple Choice

NaCl is the chemical formula for salt. Is NaCl considered a compound or an element?
element
compound
both
neither
171
Multiple Choice
What charge does an atom have when it GAINS electrons?
Positive
Negative
Neutral
172
Multiple Choice
What subatomic particle has most likely been changed in an ion?
The number of electrons
The number of protons
The number of neutrons
173
Multiple Choice

174
Multiple Choice
3
24
48
175
Multiple Choice
How many Hydrogen atoms are in Water?
176
Multiple Choice
177
Multiple Choice

How many valence electrons are in the element Carbon?
2 electrons
6 electrons
4 electrons
8 electrons
178
Multiple Choice
What happens in an ionic bond?
Atoms gain or lose electrons and bond through charge.
Atoms share valence electrons.
Atoms connect through similar affinities (e.g. both hydrophobic)
179
Multiple Choice
What happens in a covalent bond?
Atoms gain or lose electrons and bond through charge.
Atoms share valence electrons.
Atoms connect through similar affinities (e.g. both hydrophobic)
180
Multiple Choice

Carbon and other nonmetals are found in which area of the periodic table?
in the bottom rows
on the left-most side
on the right side
in the middle columns
181
Multiple Choice

The order of the elements in the periodic table is based on what?
atomic mass
the number of protons in the nucleus
the electric charge of the nucleus
the number of neutrons in the nucleus
182
Multiple Choice
How many neutrons does this isotope have? 89 36Kr
183
Multiple Choice
Which of the following is a Replacement Reaction?
4Na + O2 > 2Na2O
HCl + NaOH > H2O + NaCl
2H2 + O2 > 2H2O
H2O2 > H2 + O2
184
Multiple Choice

What is the atomic weight of H2O?
33
12
24
18
185
Multiple Choice
Where is the mass of the atom?
In the shells
The valence electrons
The nucleus
The electrons
186
Multiple Choice
Which part of the atom takes up the most space? (What makes up most of the volume of an atom?)
The Electrons
The Protons
The Neutrons
The Nucleus
187
Multiple Choice
The Nucleus of an atom is made up of:
protons and electrons
protons and neutrons
electrons and neutrons
electrons
188
Multiple Choice

The atomic number of boron is 5. Boron-11 has an atomic mass of 11. It contains:
5 protons and 6 electrons.
6 protons and 5 neutrons.
5 electrons and 6 protons.
5 protons and 6 neutrons.
189
Multiple Choice
Which of the following equation is balanced?
Na + O2 > Na2O
4Na + 2O2 > Na2O
Na + 4O2 > 2Na2O
4Na + O2 > 2Na2O
190
Multiple Choice
Which of the following equation is balance?
N2 + H3 > NH3
N2 + 3H2 > 2NH3
3N2 + 2H3 > NH3
2N2 + H3 > 3NH3
191
Multiple Choice
Which of the following equation is balance?
Ca + O2 > CaO
N2 + O2 > NH3
H2O + NaCl > HCl + NaOH
K + Cl2 > KCl
192
Multiple Choice
Which of the following compounds is expected to have a pH of 12?
HCl
H2O
LiOH
H2SO4
193
Multiple Choice
Which of the following is an acid?
NaCl
CH4
KOH
HCl
194
Multiple Choice
Which group on the periodic table is composed of all gases?
1
3
16
18
195
Multiple Choice
Atomic number of an element is equal to:
the number of electrons in the nucleus
the number of protons in the nucleus
the number of neutrons in the nucleus
the number of electron shells
196
Multiple Choice
Which would be the expected pH level for a strong acid?
pH level of 14
pH level of 10
pH level of 7
pH level of 2
197
Multiple Choice

Using the periodic table, how many Protons does one Carbon atom have?
2
4
6
12
198
Multiple Choice

Which element has 11 protons in its nucleus?
Cu
B
Na
C
199
Multiple Choice

How many electrons does an atom of Chlorine have?
1
3
17
35
200
Multiple Choice
Which of the following compounds contain an ionic bond?
HCl
NH3
CH4
NaCl
201
Multiple Choice
Which of the following compound contain a covalent bond?
Li2O
NaCl
NH3
MgCl2
202
Multiple Choice

The model represents which of the following element?
Nitrogen
Silicon
Argon
Chlorine
203
Multiple Choice
Which of the following is true for Covalent Bonds?
Electrons are transferred from one atom to another.
Electrons are shared between atoms.
They are the bonds that form between metals and non-metals.
There is electrostatic attraction.
204
Multiple Choice
What type of bonds form between metals and a non-metal?
Covalent bonds
Ionic bonds
Nuclear bonds
Polar bonds
205
Multiple Choice

How many valence electrons does Oxygen have?
2
4
6
8
206
Multiple Choice

Which of the following elements have 5 valence electrons?
Boron
Carbon
Iodine
Nitrogen
207
Multiple Choice

Which of the following element could this Dot diagram represent?
Carbon
Iodine
Sodium
Nitrogen
208
Multiple Choice

Kool-Aid can be used to tie dye shirts by just mixing it with hot water to make a hot Kool-Aid solution.
In the Kool-Aid solution, the Kool-Aid is the___ and the water is the ___.
element / compound
atom / molecule
reactant / product
solute / solvent
209
Multiple Choice

This salad dressing is a ___ mixture. So it ___ a solution.
homogeneous / is not
heterogeneous / is not
heterogeneous / is
homogeneous / is
210
Multiple Choice

This tea, without the lemon, is a ___ mixture so it
___ a solution.
homogeneous / is
heterogeneous / is
homogeneous / is not
heterogeneous / is not
211
Multiple Choice

The solubility of a solute tells us ___.
how much solute dissolves in a given amount of solvent
how much solvent dissolves in a given amount of solute
how fast a solvent dissolves in a given amount of solute
how fast a solute dissolves in a given amount of solvent
212
Multiple Choice

The solubility of sodium chloride (NaCl) in 100 mL of H2O at 20⁰C is about ___.
43 g/100 mL
50 g/ 100 mL
25 g/100 mL
37 g/100/mL
213
Multiple Choice

What is the solubility of sugar in 100 mL of water at 80⁰C?
40 g/100 mL
350 g/100 mL
214
Multiple Choice

If you wanted to dissolve 250 g of sugar in 100 mL of water, the water would have to be at least
___.
100C
250C
350C
450C
215
Multiple Choice

Which salt is the most soluble in 100 mL of water at 1000C?
Ce2(SO4)3
cerium sulfate
NaCl
sodium chloride
KClO3
potassium chlorate
KCl
potassium chloride
K2Cr2O7
potassium dichromate
216
Multiple Choice

Which salt is the least soluble in 100 mL of water at 1000C?
Ce2(SO4)3
cerium sulfate
NaCl
sodium chloride
KClO3
potassium chlorate
KCl
potassium chloride
K2Cr2O7
potassium dichlorate
217
Multiple Choice

Which salt is the most soluble in 100 mL of water at all temperatures?
CaCl2
calcium chloride
NaNO3
sodium nitrate
KNO3
potassium nitrate
KCl
potassium chloride
K2Cr2O7
potassium dichromate
218
Multiple Choice

If you wanted to dissolve about 45 g of potassium chloride in 100 mL of water, the water would have to be at least ___ 0C.
20
30
40
50
219
Multiple Choice

How much potassium nitrate, KNO3, will dissolve in 200 mL of water at 100C?
20
30
40
50
220
Multiple Choice

When the solvent in a solution CAN dissolve more solute, the solution is ___.
unsaturated
saturated
supersaturated
221
Multiple Choice

When the solvent in a solution CANNOT dissolve more solute and some solute settles on the bottom of the container, the solution is ___.
unsaturated
saturated
supersaturated
222
Multiple Choice

This solution of potassium nitrate and water is a(n)
___ solution because there is no KNO3 on the bottom.
saturated
unsaturated
supersaturated
223
Multiple Choice

This solution of potassium nitrate and water is a(n)
___ solution because there is KNO3 on the bottom.
saturated
unsaturated
supersaturated
224
Multiple Choice

This solution of sucrose (table sugar) and water is a(n)
___ solution because there are crystals in the solution. This is how rock candy is made!
saturated
unsaturated
supersaturated
225
Multiple Choice

The pH of a solution depends on the amount H+/H3O+ ___ in the solution.
atoms
ions
molecules
elements
226
Multiple Choice

The the lower the pH of a substance, the ___ H+/H3O+ ions it has in it and the ___ OH- ions it has in it.
more/less
less/more
227
Multiple Choice

The the higher the pH of a substance, the ___ H+/H3O+ ions it has in it and the ___ OH- it has in it.
more/less
less/more
228
Multiple Choice

The the pH scale goes from ___ to ___.
0 - 20
5 - 25
0 - 14
1 - 50
229
Multiple Choice

Bases have a pH of ___.
0 to less than 7
5 to 14
more than 7 to 14
1 to 14
230
Multiple Choice

Acids have a pH of ___.
0 to less than 7
5 to 14
more than 7 to 14
1 to 14
231
Multiple Choice

Substances with a of 7 like water are neutral meaning that they have ___.
more H+ ions than OH- ions
less H+ ions than OH- ions
equal amounts of H+ ions and OH- ions
232
Multiple Choice
H3O+ is called the ___ ion.
hydrogen
hydroxide
hydronium
hydro
233
Multiple Choice
OH- is called the ___ ion.
hydrogen
hydroxide
hydronium
hydro
234
Multiple Choice

Blue litmus paper turns ___ in acids.
green
red
purple
pink
235
Multiple Choice

Red litmus paper turns ___ in bases.
green
brown
blue
pink
Group 18
Group 1
Group 2
Groups 3-12
Group 17
Noble Gases
Alkali Metals
Alkaline Earth Metals
Transition metals
Halogens
Noble Gases
Alkali Metals
Alkaline Earth Metals
Transition metals
Halogens
Show answer
Auto Play
Slide 1 / 235
MATCH
Similar Resources on Wayground

229 questions
ALL Bruno Sight Words
Presentation
•
KG

234 questions
math oe part 7
Presentation
•
KG - 1st Grade

240 questions
ELLevate Lv.1 (Module 1 - What Makes You Special ?)
Presentation
•
8th Grade

236 questions
Untitled presentation
Presentation
•
KG

236 questions
Untitled Presentation
Presentation
•
University

237 questions
Ignition systems ppt
Presentation
•
KG - University

242 questions
Mid-Unit Review | CALM and LS 15
Presentation
•
9th - 12th Grade

241 questions
Homework Unit 5 P2,4,6
Presentation
•
6th - 8th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for

20 questions
Cartoon Characters!
Quiz
•
KG - 5th Grade

10 questions
Movie Trivia
Quiz
•
KG - 2nd Grade

15 questions
Memorial Day Trivia
Quiz
•
KG - 12th Grade

12 questions
Name that Candy
Quiz
•
KG - 12th Grade

20 questions
Guess The App
Quiz
•
KG - Professional Dev...

20 questions
Guess that Disney
Quiz
•
KG - 5th Grade

16 questions
cartoons
Quiz
•
KG - 6th Grade

16 questions
Fun Fun Fun Fun!!!!!!!!!!!!!!
Quiz
•
KG - 5th Grade