
- Resource Library
- Science
- Chemistry
- Elements, Compounds, And Mixtures
- 1b Elements, Compounds And Mixtures

1b Elements, Compounds and Mixtures
Presentation
•
Science
•
8th Grade
•
Medium
+9
Standards-aligned
Michelle Zou
Used 12+ times
FREE Resource
17 Slides • 28 Questions
1

2

3
Multiple Choice
What is the best model for an element?
4
Multiple Choice

What kind of matter is represented in the model?
element
compound
mixture of elements
mixture of compounds
mixture of elements and compounds
5

6

7
Multiple Choice
What is the best model for a compound?
8
Multiple Choice

What kind of matter is represented in the model?
element
compound
mixture of elements
mixture of compounds
mixture of elements and compounds
9

10
Multiple Choice
What is the best model for a mixture of elements?
11
Multiple Choice
What is the best model for a mixture of compounds?
12
Multiple Choice
What is the best model for a mixture of elements and compound?
13
Mixtures can be easily separated by physical means
For example, by sieving, filtering or evaporating

Pure substance melt and boil at fixed temperature.
14
Fill in the Blanks
15
Drag and Drop
16
Separation of mixture
fitration
crystallisation
simple distillation
fractional distillation
paper chromatography

17
Multiple Choice
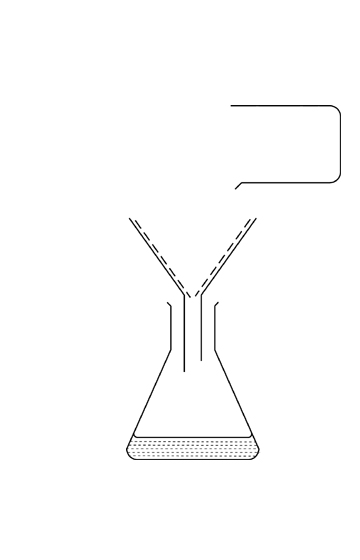
What is the name of this separation technique?
distillation
filtration
evaporation
chromatography
18
Filtration
Filtration is used to separate substances according to the size of their particles.
The substance left in the filter paper is called residue and the liquid that comes through is called filtrate.

19
Multiple Choice
During filtration, the solid that is trapped in the filter paper is called ..A.. and the liquid that has passed through the filter paper is called ..B..
A: residue B: filtrate
A: residue B: filterate
A: filtrate B: residue
A: filterate B: residue
20
Separation Flowchart
It is important to represent your flowchart of separation correctly in your answers. Indicating what substances you have and what is being separated out.
Use diamonds for a separation technique, and rectangles for a substance that is being separated out by that technique, or a substance that is being added.
E.g. Given a mixture of dry salt and salt, outline the procedure to obtain dry sand and dry salt.

21
Crystallisation
Crystallisation can be used to separate a solute from a solution.
e.g. separating NaCl from a NaCl solution.
Subject | Subject
Some text here about the topic of discussion

22
Multiple Choice
distillation
dissolving
evaporation
decanting
23
Multiple Choice

What is evaporation (or crystallisation) usually used for?
To get clean water from dirty water
To separate a soluble solid (like salt or minerals) from water
To separate hydrogen from water
To separate an insoluble solid (like sand) from water
24
Multiple Choice
What is the physical property on which fractional distillation depends?
different boiling points
different melting points
different solubilities
different densities
25
Distillation
Distillation uses evaporation (heating) and condensation (cooling) to separate the components of a liquid mixture according to their boiling points.
Subject | Subject
Some text here about the topic of discussion
26
Multiple Choice
It always requires heat
It cannot be used for insoluble solids
All of the solute is recovered
The solvent (liquid part) is not recovered
27
Fill in the Blanks
Type answer...
28
Multiple Choice
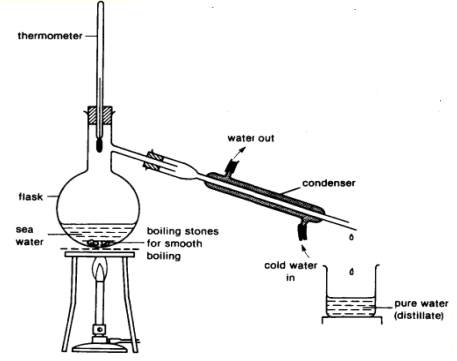
What is the name of this separation technique?
Simple distillation
Fractional distillation
Boiling
Evaporation
29
Fractional distillation
a method for separating a liquid from a mixture of two or more liquids.
the liquids in the mixture have different boiling points
When the mixture is heated, one liquid evaporates before the other

30
Multiple Choice

The separation technique shown in the image is called what?
Chromatography
Distillation
Centrifuging
Decanting
31
Chromatography
Chromatography is used to separate the substances in a mixture based on their solubility.
Subject | Subject
Some text here about the topic of discussion
32
Draw
Circle the most soluble substance in on the chromatography paper below.
33
Multiple Choice
Chromatography separates substances according to their...
Density
Solubility
Size
Weight
34

Example 1: To find out how many types of dyes are in an unknown sample.
35
Multiple Choice

Is the blue spot more soluble or less soluble than the yellow spot?
More soluble
Less soluble
36
Copy this down in Page 16 of Lesson Notes.
Example 1: To find out how many types of dyes are in an unknown sample.

37
Multiple Choice

There are two spots on the chromatogram. How many components are present in the green dye?
1
2
3
4
38
Multiple Select
Fractional distillation is a method for separating
a liquid from a mixture of solutions
a liquid from a mixture of solutes
a liquid from a mixture of solvents
a liquid from a mixture of liquids
39
Retention factor
how much the compound has moved compared to how much the solvent has moved
Rf = distance moved by compound ÷ distance moved by the solvent

40
Fill in the Blanks
Type answer...
41
Multiple Choice
What is the formula for the Rf value?
Rf value = distance travel of solvent / distance travel of solute or sample
Rf value = distance travel of solute or sample / distance travel of solvent
Rf value = distance travel of solid / distance travel of liquid
Rf value = distance travel of residue / distance travel of solution
42

Rf = distance travel of solute
distance travel of solvent front
Rf value formula
Key:
Usually measure to the middle of the ink spot.
43
Fill in the Blanks

Type answer...
44
Fill in the Blanks

Type answer...
45
Multiple Select

Which of the inks are pure?
Blue ink
Red ink
Yellow ink
Green ink
Black ink

Show answer
Auto Play
Slide 1 / 45
SLIDE
Similar Resources on Wayground

43 questions
Ecosystems (Biotic and Abiotic Factors)
Presentation
•
8th Grade

39 questions
Atomic Structure
Presentation
•
8th Grade

38 questions
Force and Motion
Presentation
•
8th Grade

40 questions
Sci 8 1.2 Scientific Method
Presentation
•
8th Grade

43 questions
Change over Time
Presentation
•
8th Grade

39 questions
Plate Tectonic Theory
Presentation
•
8th Grade

44 questions
The Carbon Cycle
Presentation
•
8th Grade

38 questions
Bacteria and Viruses
Presentation
•
8th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

12 questions
PS.8.1.2, PS.8.1.3, PS.8.1.5 Review
Quiz
•
8th Grade

20 questions
Moon Phases and Eclipses
Quiz
•
6th - 8th Grade

22 questions
6th & 8th Grade Science Material SOL Review
Quiz
•
6th - 8th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade

48 questions
8th Grade NC Science EOG Review 2
Quiz
•
8th Grade

19 questions
Renewable and Nonrenewable Resources (CK)
Quiz
•
8th Grade

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade