

G10_5.2: Classifying Chemical Reactions
Presentation
•
Chemistry
•
10th Grade
•
Easy
+5
Standards-aligned
Oyerohunke oyediran
Used 20+ times
FREE Resource
22 Slides • 33 Questions
1

Classifying Chemical Reactions
Pg 157 - 166
Lesson 2
2


Focus Question
What are the different types of chemical
reactions?
3


New Vocabulary (Quizlet Practice )
synthesis reaction
combustion reaction
decomposition reaction
single-replacement reaction
double-replacement reaction
precipitate
4


Review Vocabulary
metal: an element that is a solid at room temperature,
is a good conductor of heat and electricity, and is
generally shiny
5
Multiple Choice
a glob of gelatinous goo
smaller than an atom
filled with raisons
more than one atom bonded together
6
Multiple Choice
H2 + O → H2O
Reactants
Products
Yields
Chemical Equation
7
Multiple Choice
H2 + O → H2O
Reactants
Products
Yields
Chemical Equation
8
Multiple Choice

CH4 and CO2
CH4 and O2
CO2 and H2O
O2 and H2O
9
Multiple Choice
In this equation, CH4 is a
product
reactant
displacement
10
Multiple Choice
Symbol
Formula
Subscript
Reaction
11
Multiple Choice
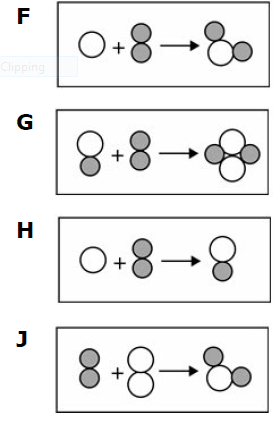
F
G
H
J
12


Chemical Reactions
Evidence of a Chemical Reaction
• A temperature change can indicate a chemical
reaction. Many reactions release energy in the
form of heat and light. Other chemical
reactions absorb heat.
• Color change can indicate a chemical reaction.
• Odor, gas bubbles, and the formation of a
solid are other indications of chemical change.
13
Multiple Select
Which of the following are evidence of a chemical reaction? (Pick more than one.)
Tearing paper.
Change in color.
Production of a gas.
Change in temperature.
Production of a precipitate.
14
Multiple Choice

A precipitate is a ___________ that forms when 2 liquids react together.
solid
liquid
gas
plasma
15
Multiple Choice

How many of the listed observations indicate a new substance was formed?
1
2
3
4
16


Types of Chemical Reactions
• Chemists distinguish among four chemical
reaction types: synthesis, combustion,
decomposition, and replacement reactions.
• By analyzing and comparing the reactants and
products of chemical reactions, you will begin
to notice patterns that will help you classify
them.
• Some reactions fit into more than one of the
types.
17



Synthesis or Combination Reactions
• In a synthesis reaction two or more substances react and combine to produce a single product.
• When two elements react, the reaction is
always a synthesis reaction.
• An example is shown below.
18
Multiple Choice
P4 + 3 O2 ----> 2 P2O3
Synthesis (or combination)
Decomposition
Single replacement
Double replacement
19
Multiple Choice
Decomposition
Combination
Combustion
Single Replacement
20
Multiple Choice
Synthesis (combination)
Decomposition
Single replacement
Double replacement
21



Combustion Reactions
• In a combustion reaction,oxygen combines
with a substance and releases energy in the
form of heat and light.
• Oxygen can combine in this way with many
substances. The following is an example.
22
Multiple Choice
Decomposition
Double replacement
Combustion
Single Replacement
23
Multiple Choice
Decomposition
Synthesis
Combustion
Single Replacement
24
Multiple Choice
What type of reaction is this?
2 CO + O2 → 2 CO2
Synthesis only
Combustion only
Single Displacement
Synthesis AND combustion
25


Decomposition Reactions
• In a decomposition reaction,a single compound
breaks down into two or more elements or new
compounds.
• To occur, these reactions often require an energy
source, such as heat, light, or electricity.
• The products of a decomposition reaction may
be elements, compounds, or one or more of
each.
26
Multiple Choice
Decomposition
Synthesis
Combustion
Single Replacement
27
Multiple Choice
CoCO3 → CoO + CO2
Synthesis
Decomposition
Single Replacement
Double Replacement
28
Multiple Choice
29


Replacement Reactions
Single-Replacement Reactions
• In a single-replacement reaction the atoms of
one element replace the atoms of another
element in a compound.
• The following generic equation can be used to
represent single-replacement reactions.
A + BX → AX + B
30
Multiple Choice
Synthesis
Combustion
Single Replacement
Double Replacement
31
Multiple Choice
Single Replacement
Decomposition
Double Replacement
Synthesis
32
Multiple Choice
Synthesis (combination)
Decomposition
Single replacement
Double replacement
33



Replacement Reactions
Double-Replacement Reactions
• A double-replacement reaction occurs when
ions exchange between two compounds.
• The figure shows a generic double replacement
equation.
34
Multiple Choice
Displacement
Synthesis
Decomposition
Oxidation
35
Multiple Choice

Synthesis
Decomposition
Single Replacement
Double Replacement
36
Multiple Choice
Synthesis (combination)
Decomposition
Single replacement
Double replacement
37


Replacement Reactions
• A solid product produced during a chemical
reaction in a solution is called a precipitate.
• All double replacement reactions produce either
water, a precipitate, or a gas.
38



Replacement Reactions
• Reactivity is the ability to react to
another substance. An activity
series (see right) can be used to
predict reactions.
• A metal will not always replace
another metal in a compound
dissolved in water because of
different reactivities.
• Halogens frequently replace other
halogens in reactions but have
differing reactivities and do not
always replace each other.
39
Multiple Choice

Which of the following is more reactive?
Calcium
Magnesium
Zinc
Magnesium
40
Multiple Choice

Will the following reaction take place?
Ni + NaCl ->
NO
YES
41
Multiple Choice
a more reactive metal displaces a less reactive metal from its compound.
A less reactive metal displaces a more reactive metal from its compound
Displacement only occurs when two of the same metals are reacted
Displacement reactions will only occur in metals above iron in the reactivity series
42


Think of the Quiz answers before moving
to the next slide
43

Quiz
double-replacement
D
synthesis
C
single-replacement
B
decomposition
A
A reaction involves two substances as reactants and
produces one compound. Which type of reaction is
it likely to be?
1.
44

Quiz
double-replacement
D
synthesis
C
single-replacement
B
decomposition
A
A reaction involves two substances as reactants and
produces one compound. Which type of reaction is
it likely to be?
1.
CORRECT
45

Quiz
decomposition
D
single-replacement
C
synthesis
B
double-replacement
A
What kind of reaction does the following
generic equation represent? A + BX → AX + B
2.
46

Quiz
decomposition
D
single-replacement
C
synthesis
B
double-replacement
A
What kind of reaction does the following
generic equation represent? A + BX → AX + B
2.
CORRECT
47

Quiz
gold
D
lithium
C
sodium
B
tin
A
According to the activity series, which of the metals
is most active?
3.
48

Quiz
gold
D
lithium
C
sodium
B
tin
A
According to the activity series, which of the metals
is most active?
3.
CORRECT
49

Quiz
Subscripts are changed
to make the number of
each element equal on
both sides.
D
Coefficients are changed
to make the number of
each element equal on
both sides.
C
Coefficients are written
in their lowest possible
ratio.
B
Subscripts should never
be changed to balance
the equation.
A
Which of the following is NOTtrue about balancing
chemical equations?
4.
50

Quiz
Subscripts are changed
to make the number of
each element equal on
both sides.
D
Coefficients are changed
to make the number of
each element equal on
both sides.
C
Coefficients are written
in their lowest possible
ratio.
B
Subscripts should never
be changed to balance
the equation.
A
Which of the following is NOTtrue about balancing
chemical equations?
4.
CORRECT
51
Match
combustion
synthesis
decomposition
single replacement
double replacement
combustion
synthesis
decomposition
single replacement
double replacement
52
Match
1 reactant breaks apart into 2 or more smaller products
2 or more reactants combine to make 1 product
O2 as a reactant & CO2 and water as products
1 element & 1 compound in products & reactants
2 compounds in reactants & products
decomposition
synthesis
combustion
single replacement
double replacement
decomposition
synthesis
combustion
single replacement
double replacement
53
Drag and Drop
54
Dropdown
55
Reorder
Put the equations in this order: decomposition, synthesis, combustion, single replacement, double replacement.

Classifying Chemical Reactions
Pg 157 - 166
Lesson 2
Show answer
Auto Play
Slide 1 / 55
SLIDE
Similar Resources on Wayground

46 questions
Lesson: Module 39 The von Thunen Model
Presentation
•
10th Grade

51 questions
Chemical Reactions
Presentation
•
10th Grade

50 questions
Chemical Binding
Presentation
•
9th Grade

50 questions
G10 Lesson 4.3: Molecular Structures
Presentation
•
10th Grade

50 questions
ESRT Astronomy
Presentation
•
10th Grade

51 questions
Virus Structure
Presentation
•
10th Grade

52 questions
Viruses (part-1)
Presentation
•
10th Grade

53 questions
SSA Earth Review All Topics
Presentation
•
9th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade