

Unit 2 Review
Presentation
•
Science
•
9th Grade
•
Medium
+4
Standards-aligned

Victoria Francis
Used 3+ times
FREE Resource
23 Slides • 52 Questions
1
Matter

2
Matter
Anything that has
mass
Takes up space (volume)
3
Open Ended
What is matter?
4
Multiple Choice
Mass measures _____________
The amount of space an object takes up
The amount of "stuff" in an object ________________
How tall the object is
5
Multiple Choice
density
volume
mass
matter
6

7
Multiple Choice
solid, volume, mass
density, volume, mass
solid, liquid, gas
density, gas, volume
8
Multiple Choice

has a definite shape and a definite volume
has a definite volume but no definite shape
has no definite shape and no definite volume
9
Multiple Choice

A liquid
has a definite shape and a definite volume
has a definite volume but no definite shape
has no definite shape and no definite volume
10
Multiple Choice

A solid
has a definite shape and a definite volume
has a definite volume but no definite shape
has no definite shape and no definite volume
11

12

13
Multiple Choice

What happens to the average kinetic energy of matter when it is heated?
It increases
It stays the same
It decreases
It cannot be determined
14
Multiple Choice
Which of the following states of matter has the LOWEST level of energy?
Solid
Liquid
Gas
Plasma
15
Multiple Choice
If the atoms of an object are moving fast, the temperature would be...
High
Low
16
Multiple Choice
What is the measure of the average kinetic energy of the particles in an object?
Heat
Motion
Thermal Energy
Temperature
17

Energy
0o C
100o C
18
Multiple Choice
At what temperature does matter change phases from a solid to a liquid?
Melting Point
Boiling Point
Condensation Point
0 degrees Celsius
19
Multiple Choice
What is the phase change called that occurs when a liquid turns into a gas?
Condensation
Freezing
Melting
Vaporization
20
Multiple Choice
Phase change going from Liquid to Solid...
Melting
Freezing
Condensation
Sublimation
21
Multiple Choice

evaporation
precipitation
condensation
sublimation
22

23

24
Multiple Choice

What is it?
Pure Substance
Mixture
25
Multiple Choice

What is it?
Pure Substance
Mixture
26

27

28

29

30
Multiple Choice

homogeneous
hetergeneous
pure substance
element
31
Multiple Choice
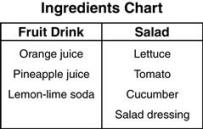
Using the information in the chart, which statement is the BEST description of the drink and salad?
The salad and fruit drink are both mixtures.
The salad and the fruit drink are both solutions.
The fruit drink is a mixture because the ingredients can't be separated.
The salad is a solution because the ingredients can't be separated.
32
Multiple Choice
A ______________ mixture is one that does not have a uniform composition.
heterogeneous
homogenous
pure substance
33
Multiple Choice
A _________________ mixture is one where the substances are evenly spread throughout.
heterogeneous
homogeneous
pure substance
34

35
Multiple Choice
True or False: elements AND compounds are pure substances.
True
False
36
Multiple Choice

What type of mixture is this
heteregenous
homogeneous
37

38

39

40
Multiple Choice
Identify the change:
Glass breaking
Physical
Chemical
41
Multiple Choice
Identify the change:
Wood burning
Physical
Chemical
42
Multiple Choice
Boiling Water
Physical Change
Chemical Change
43
Multiple Choice
Dissolving Sugar
Physical Change
Chemical Change
44
Multiple Choice
Nail Rusting
Physical Change
Chemical Change
45
Multiple Choice
Egg Cooking
Physical Change
Chemical Change
46
Multiple Choice
Which observations prove a chemical change occurred ?
Dissolving and expected color change
Mixing and dissolving
State of matter change and boiling water bubbles
Unexpected color change and temperature change
47
Multiple Choice
When mixing milk and vinegar, a white, chunky, solid substance is formed. What has occurred?
Physical change: state of matter
Chemical change: precipitate (forming a solid)
Physical change: freezing
Chemical change: new gas
48
Multiple Choice
Milk turns yellow when it spoils.
Dough becomes darker when it is baked into cake.
A red candy dissolves and turns water pink.
A match turns black when it is burned.
49
Multiple Choice
Which of these is NOT an indicator of a chemical change:
Unexpected temperature change after combining two or more substances
Two combined substances cause fizzing
Bubbles and steam rise from a pot of boiling salt water
Precipitates form in a solution of two or more substances
50

51
Multiple Choice
Mass and Volume
Temperature and Mass
Grams and Centimeters
Volume and Weight
52
Multiple Choice
7 mL
7 g/cm3
28 g/cm3
1/7 g/cm3
53
Multiple Choice

Calculate the density of the cube.
Hint: Volume of the Cube is LxWxH
2 g/cm3
4 g/cm3
6 g/cm3
8 g/cm3
54
Multiple Choice
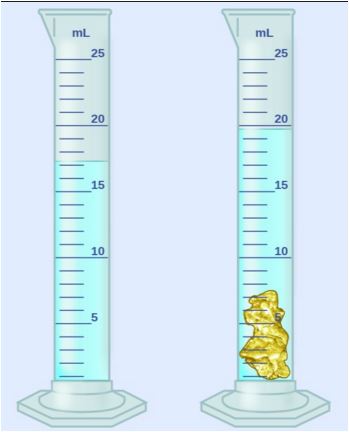
9 g/mL
10.5 g/mL
6.5 g/mL
8 g/mL
55

56
Multiple Choice
Which of these pH values represent an acid?
4
8
10
12
57
Multiple Choice
Which of the following are properties of acids?
They conduct electricity when dissolved in water
They taste sour
They react with metals to produce hydrogen gas
All of the answer choices are correct
58
Multiple Choice
Which of the following is a property of a base?
bitter taste
reacts with metals
salty
sour taste
59
Multiple Choice
Which of the following pH values represents a base?
2.1
4.6
6.8
8.1
60
Multiple Choice
If the substance is neutral, what would the pH be?
3
5
7
9
11
61

The Law of Conservation of Mass
1 + 1 = 2
62

10g + 5g -------> 15g
63
Multiple Choice
If 20g of sugar is stirred into a beaker containing 100g of water. What would the mass of the dissolved solution be after the sugar is dissolved fully?
20g
100g
80g
120g
64

65

66
Multiple Choice
How many atoms of aluminum are on each side of the following equation?
4Al + 3O2 --> 2Al2 O3
2
6
4
1
67

68
Labelling
How many atoms of each are in the reactants?
69
Multiple Choice
a number in front of a chemical formula that indicates how many molecules or compounds of each reactant and product are involved in a reaction
denominator
coefficient
numerator
subscript
70
Multiple Choice
a substance that forms in a chemical reaction
coefficient
subscript
reactant
product
71
Multiple Choice
a total of 40g of products
a total of 10g of products
a total of 80 g of products
a total of 20g of products
72
Multiple Choice

Which of the models of a chemical reaction best represents the Law of Conservation of Mass?
A
B
C
D
73
Multiple Choice
Based on the Law of Conservation of Matter, describe the relationship between the mass of the reactants and the mass of the products.
The mass of the products is always greater than the mass of the reactants.
The mass of the reactants is less than the mass of the products.
The mass of the reactants equals the mass of the products.
The mass of the reactants is greater than the mass of the products.
74
Multiple Choice
What happens to atoms in a chemical reaction?
they are destroyed
they are created
they are rearranged
75
Multiple Choice

Which section represents the reactants?
A
B
C
Matter

Show answer
Auto Play
Slide 1 / 75
SLIDE
Similar Resources on Wayground

69 questions
Mitosis
Presentation
•
9th - 10th Grade

68 questions
Forces and Motion Test Review
Presentation
•
8th Grade

66 questions
Chapter 7
Presentation
•
9th Grade

70 questions
9. Kl. Unit 1 Gaming Vocabulary
Presentation
•
9th Grade

65 questions
8 - Earthquakes
Presentation
•
9th Grade

71 questions
D. useful or wasted energy exchanged in physical systems
Presentation
•
KG

73 questions
PhySci Final Review 2: Mechanical Energy
Presentation
•
9th Grade

68 questions
8th Grade Science CBA Review
Presentation
•
8th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Science

46 questions
Unit 4 Geosphere Test Review
Quiz
•
9th - 12th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

16 questions
Ecosystem Stability
Quiz
•
9th Grade

20 questions
Mendelian Genetics Review
Quiz
•
9th Grade

12 questions
Unit 6 Quiz #3 (Life Cycles of Stars)
Quiz
•
9th Grade

34 questions
Human Impact on Ecosystems Quiz
Quiz
•
9th Grade

10 questions
Exploring Alfred Wegener's Continental Drift Theory
Interactive video
•
6th - 10th Grade