

REVIEW Grade 10_Thermal Properties
Presentation
•
Physics
•
10th Grade
•
Medium
Irvani Prasanti
Used 21+ times
FREE Resource
5 Slides • 57 Questions
1
REVIEW TIME!
Grade 10

Thermal Expansion, Specific heat capacity, Evaporation and Boiling, Transfer heat.
2
THERMAL EXPANSION
3
Multiple Choice
it gains heat from the hot water and expands.
loses heat from the hot water and contracts.
the surrounding air is too warm.
the liquid is boiling.
4
Multiple Choice
Particles move faster and expand
Particles move faster and become closer together.
Particles move slower and the air expands
Particles move faster become further apart
5
Multiple Choice
Jack placed his wooden baseball bat in the sun. The bat cracked open along the center. What is the most likely reason for this happening?
The wooden bat cools in the sun.
The wooden bat evaporates in the sun.
The sun’s heat causes the wooden bat to expand.
The sun’s heat causes the wooden bat to contract.
6
Multiple Choice
Which statement best describes how winter weather can damage roads?
Snow and ice can cause car accidents, which damage roads.
Rain, ice, and snow can cause the painted lines on roads to fade.
Cold temperatures can cause road materials to expand, creating potholes.
Water can get into cracks in roads, then freeze and expand, making the cracks worse.
7
Multiple Choice

The ball and the ring shown below are made up of metal. The diameter of the ball is larger than the diameter of the ring. Which of the following processes makes the ball pass through the ring?
Heat the ball
Heat the ring
Heat both of them
Cool both of them down
8
Multiple Choice

When water in the flask was heated, the water level rose because ______________.
the water gained heat and expanded
the water lost heat and expanded
the water gained heat and contracted
the water lost heat and contracted
9
Multiple Choice

When water in the flask was cooled down, the water level dropped because ______________.
the water gained heat and expanded
the water lost heat and expanded
the water gained heat and contracted
the water lost heat and contracted
10
Multiple Choice

A bottle of air is connected to a balloon. When the bottle is placed in cold water, ______________.
the air in the balloon gained heat and the balloon expanded
the air in the balloon lost heat and the balloon expanded
the air in the balloon gained heat and the balloon contracted
the air in the balloon lost heat and the balloon contracted
11
Multiple Choice

A bottle of air is connected to a balloon. When the bottle is placed in hot water, ______________.
the air in the balloon gained heat and the balloon expanded
the air in the balloon lost heat and the balloon expanded
the air in the balloon gained heat and the balloon contracted
the air in the balloon lost heat and the balloon contracted
12
Multiple Choice

When the metal ball is placed in fire, ______________.
the metal ball gained heat and expanded
the metal ball gained heat and contracted
the metal ball lost heat and contracted
the metal ball lost heat and expanded
13
Multiple Choice

When the metal ball is placed in a bowl of ice, ______________.
the metal ball gained heat and expanded
the metal ball gained heat and contracted
the metal ball lost heat and contracted
the metal ball lost heat and expanded
14
Multiple Choice

The metal plate ______________(lost / gained) heat from the heat source and _____________ (contracted / expanded).
gained, expanded
gained, contracted
lost, expanded
lost, contracted
15
Multiple Choice

The metal plate ______________(lost / gained) heat to the ice cube and _____________ (contracted / expanded).
gained, expanded
gained, contracted
lost, expanded
lost, contracted
16
Multiple Choice

Which statement is correct?
Block P gained heat and expanded.
Block P gained heat and contracted.
Block P lost heat and expanded.
Block P lost heat and contracted.
17
BOILING VS EVAPORATION
18
Multiple Choice
What do you call the temperature at which the liquid boils?
boiling point
melting point
evaporating point
freezing point
19
Multiple Choice
What is the boiling point of water?
100°C
10°C
101°C
110°C
20
Multiple Choice
When water gains heat, its temperature ..............................
increases
decreases
remains the same
21
Multiple Choice
When the liquid reaches its boiling point, the temperature ..............................
increases
decreases
remains constant
22
Multiple Choice
At which altitude does the water boil at the lowest temperature?
At sea level
At 4,400 metres above sea level
At 8,800 metres above sea level
At 1,800 metres above sea level
23
Multiple Choice

Which letter shows the steam?
A
B
C
24
Multiple Choice
.............................. occurs throughout the liquid.
Boiling
Evaporation
25
Multiple Choice
.............................. occurs all the time and at any temperature.
Boiling
Evaporation
26
Multiple Choice
.............................. occurs only on the surface of the liquid.
Boiling
Evaporation
27
Multiple Choice
.............................. takes place at a fixed temperature.
Boiling
Evaporation
28
Multiple Choice
The process of boiling occurs .............................. than evaporation.
faster
slower
29
SPECIFIC HEAT CAPACITY
30
Multiple Choice
The specific heat of platinum is 0.133 J/g°C. How much heat(Q) is released when a 10 g piece of platinum cools from 100°C to 50°C?
66.5 J
665 J
0.0266 J
0.665 J
31
Multiple Choice
The specific heat(c) of copper is 0.39 J/g °C. What is the temperature change(∆t) when 100 Joules of heat(Q) is added to 20 grams?
12.82 °C
24.12°C
1.95 °C
5128 °C
32
Multiple Choice
It heats up quickly with energy added
It requires more energy to change temperature
33
Multiple Choice
c
Q
m
t
34
Multiple Choice
The amount of thermal energy required to increase the temperature of 1kg of a material by 1°C.
The amount of radiant energy required to increase the temperature of 1kgof a material by 1°C
The amount of energy required to increase the temperature of 1kgof a material by 1°C.
The amount of friction required to increase the temperature of 1kgof a material by 1°C.
35
Multiple Choice

Which material heats up quickest?
Aluminium
Concrete
Diamond
Glass
Water
36
Multiple Choice

Some 1kg of water and 1kg of diamond are heated to the same temperature. Which is true?
They both require the same amount of energy
The diamond needs around 8 times more energy
The water needs about 8 times more energy
The diamond heats up slower than the water
The water ends up colder
37
Multiple Choice

10 kg of aluminium is heated from 120C to 220C. How much energy is needed?
8 970 000 J
897 J
8970 J
89 700 J
897 000J
38
Multiple Choice
The temperature of a 6.0 g sample of glass changed from 20.0°C to 45.0°C when it absorbed 550 J of heat. What is the specific heat of this glass?
0.27 J/g °C
3.7 J/g °C
130 J/g °C
12 J/g °C
39
Multiple Choice
Which of the following best explains why the sand at the beach is hotter than the water?
Sand has a higher specific heat than water.
Sand has a lower specific heat than water.
There is more water than sand at the beach.
There is more sand than water at the beach.
40
Multiple Choice
Object A, it has a lower spefici heat
Object B, it has a lower specific heat
Object A, it has a higher specific heat
Object B, it has a higher specific heat
41
Multiple Choice
Given that 10 g of copper block and 10 g of clay block is heated with the same amount of heat. It is observed that the 10 g copper block is hotter than 10 g of clay block. Which of the following is accurate?
heat capacity of 10 g of copper block is lower than 10 g of clay block
specific heat capacity of 10 g copper block is lower than 10 g clay block
heat capacity of 10 g of copper block is higher than 10 g of clay block
specific heat capacity of 10 g copper block is higher than specific heat capacity of 10 g clay block
42
HEAT TRANSFER
43
Multiple Choice

conduction
convection
radiation
44
Multiple Choice
45
Multiple Choice

convection
conduction
radiation
46
Multiple Choice
convection
conduction
radiation
47
Multiple Choice
convection currents in fluids due to changing masses
convection currents in fluids due to changing densities
heat transfer by conduction
convection currents in fluids due to constant temperatures
48
Multiple Choice

Hot coffee is stirred with a spoon, the spoon gets hot due to
Convection
Conduction
Radiation
49
Multiple Choice

A chair is placed several feet from a fire in a fireplace. The fireplace has a glass screen. The side of the chair facing the fireplace gets warm because of
Convection
Conduction
Radiation
50
Multiple Choice

A certain type of decorative lamp contains colored liquids. These liquids form globs that break off and rise to the top of the liquid. The globs rise due to
Convection
Conduction
Radiation
51
Multiple Choice
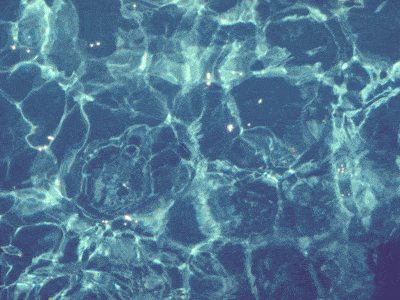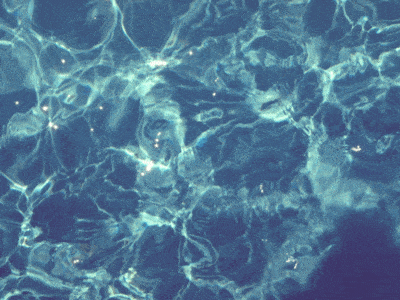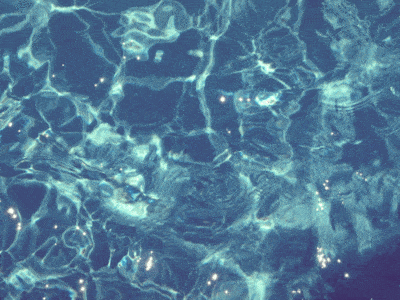
In a swimming pool, the water near the surface is slightly warmer. The warm water rises because of
Convection
Conduction
Radiation
52
Multiple Choice

As you hold pieces of ice, they begi to melt from the warmth of you hand on them. This is because of
Convection
Conduction
Radiation
53
Multiple Choice

A group of friends gather around a fire to stay warm.
conduction
convection
radiation
54
Multiple Choice
Radiation
Convection
Conduction
Heat
55
Multiple Choice
Electromagnetic waves
Convection
Conduction
Heat
56
Multiple Choice
Radiation
Convection
Conduction
Heat
57
Multiple Choice
Convection
Electromagnetic waves
Radiation
Conduction
58
Multiple Choice
Conductor
Insulator
Convector
Radiator
59
Multiple Choice
Conductor
Insulator
Convector
Radiator
60
Multiple Choice
In what state does the conduction occur the best?
Solid
Liquid
Gas
61
Multiple Choice
A wooden block
A ceramic mug
A glass cup
A metal bowl
62
Multiple Choice
Plastic
Ceramic
Styrofoam
Glass
REVIEW TIME!
Grade 10

Thermal Expansion, Specific heat capacity, Evaporation and Boiling, Transfer heat.
Show answer
Auto Play
Slide 1 / 62
SLIDE
Similar Resources on Wayground

53 questions
Ecology Overview
Presentation
•
9th - 10th Grade

56 questions
Thermal Energy Transfer
Presentation
•
10th Grade

57 questions
Author's Purpose
Presentation
•
10th Grade

55 questions
BOYLE'S LAW
Presentation
•
9th - 10th Grade

60 questions
Unit 6 Building Solutions Lesson A
Presentation
•
10th Grade

60 questions
The Constitution
Presentation
•
9th - 10th Grade

56 questions
PREPARE POULTRY AND GAME DISHES
Presentation
•
10th Grade

56 questions
Measurement, Motion & Density
Presentation
•
9th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Physics

11 questions
Electricity Explained
Interactive video
•
9th - 12th Grade

15 questions
waves and wave properties
Quiz
•
9th - 12th Grade

35 questions
Electricity and Magnetism SPS10
Quiz
•
8th - 10th Grade

18 questions
Unit 1 and 2 Concepts Review
Quiz
•
9th - 12th Grade

15 questions
Motion Graphs 101
Quiz
•
8th - 11th Grade

12 questions
EOY DAY 1 ELECTRICITY
Presentation
•
10th - 12th Grade

10 questions
EOY REVIEW 2- Electrostatics & Momentum
Presentation
•
10th - 12th Grade

6 questions
EOY REVIEW 3 (Momentum/Collisions/Impulse)
Presentation
•
10th - 12th Grade