

1.2 Groups and periods
Presentation
•
Science
•
7th Grade
•
Practice Problem
•
Easy
William Katumba
Used 5+ times
FREE Resource
4 Slides • 57 Questions
1
Recap & Review
What did we look at yesterday?
Complete these review questions to jog your memory about yesterday's class.
2
Fill in the Blanks
Type answer...
3
Fill in the Blanks
Type answer...
4
Multiple Choice
left
right
top
bottom
5
Multiple Choice
able to bend
will break easily
can be used for wire
is shiny
6
Multiple Choice
electricity flows easily
quacks like a duck
can be drawn into a wire
able to bend
7
Multiple Choice
left
right
top
bottom
8
Multiple Choice

metal
nonmetal
9
Multiple Choice
metal
nonmetal
10
Multiple Choice
metal
nonmetal
11
Multiple Choice
heat
electricity
both
neither
12
Multiple Choice
malleable
conductors
brittle
luster
13
Multiple Choice

Metal
Nonmetal
Metalloid
Metamorphic
14
Multiple Choice
Metal
Nonmetal
Metalloid
Pretty
15
Multiple Choice
can be pulled into long wires
dull surface
high density
malleable
16
Multiple Choice

Sample 1
Sample 2
Sample 3
Sample 4
17
Multiple Choice
What way of classifying elements did we look at yesterday?
Metals OR Nonmetals
Groups
Liquids or Gases
Metals or Metalloids
18
Multiple Choice
Approximately what percentage of the periodic table is comprised of metals?
20 %
50 %
80 %
99 %
19
Multiple Choice
Page 61;
When metals dissolve in water they form.........
acidic solutions
alkaline solutions
20
Multiple Select
Page 61;
Which of the following non metals dissolve in rain water to form acidic rain?
Sulfur dioxide
Nitrogen dioxide
Carbon dioxide
Sulfur monoxide
Nitrogen monoxide
21
Multiple Choice
Page 61;
When non metal oxides dissolve in water they form.........
acidic solutions
alkaline solutions
22
Multiple Choice
Page 61;
Chemical properties describe .........
things you can observe and measure
chemical reactions
23
Fill in the Blanks
Type answer...
24
Multiple Choice
Page 61;
When metals and non metals react with oxygen, the products are;
Oxides
Monoxides
Carbons
Oxygen & Hydrogen
25
Multiple Choice
Page 61; TRUE or FALSE
Most metal oxides are gases at 20°C
TRUE
FALSE
26
Multiple Choice
Page 61; TRUE or FALSE
Most non metal oxides are gases at 20°C
TRUE
FALSE
27
Fill in the Blanks
Type answer...
28
Fill in the Blanks
Type answer...
29
Multiple Choice
Acid rain is formed when ______ oxides dissolve in water
metal
non metal
30
Multiple Choice
If an element has a high melting point, its is most definitely a .....
metal
non metal
31
Multiple Choice
If an element has a low boiling point, its is most definitely a .....
metal
non metal
32
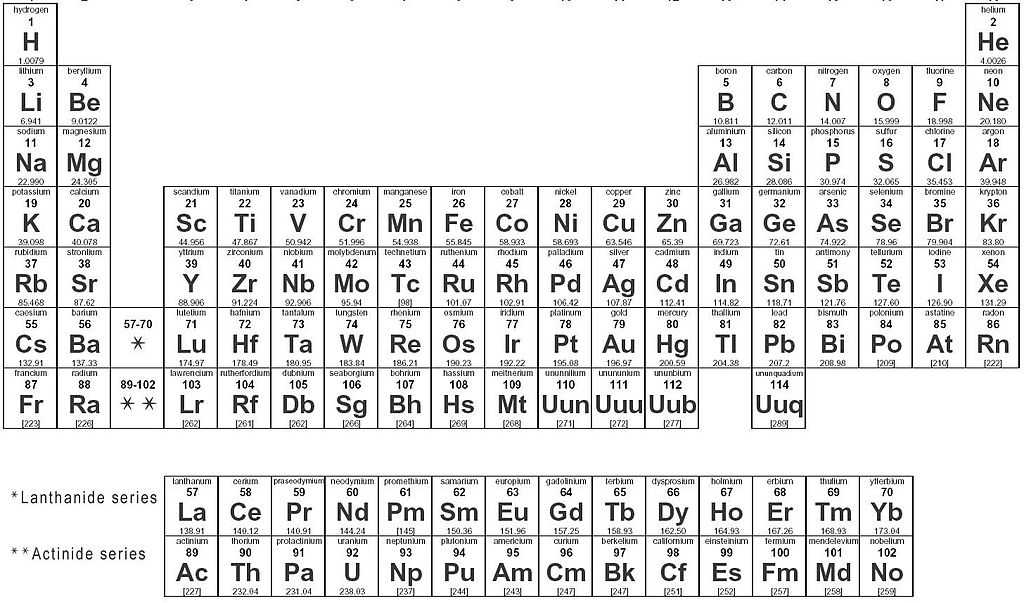
Metal or non-metal?
Carefully study the image below and help Gonza decide if the elements are metals or nonmetals

33
Multiple Choice

From the table, Element X is a ________
Metal
Non-metal
Metalloid
34
Multiple Choice

From the table, Element Y is a ________
Metal
Non-metal
Metalloid
35
Multiple Choice

From the table, Element Z is a ________
Metal
Non-metal
Metalloid
36
Lesson 1.2; Groups and periods
Lesson objectives;
Use patterns to predict properties of elements
Compare the patterns in properties in the groups and periods of the periodic table
Use pages 62-63 of your textbook to complete this worksheet.
37
Fill in the Blanks
Type answer...
38
Fill in the Blanks
Type answer...
39
Fill in the Blanks
Type answer...
40
Multiple Select
Page 62;
Going down the group, there is a pattern in the properties such as; (select 3 answers)
Melting point
Boiling point
Density
Volume
41
Multiple Choice
Recall;
What does 'melting point' mean?
The temperature at which a solid melts / turns to liquid
The temperature at which a liquid boils / turn to vapor
The amount of space occupied by an object
How much mass something has for its volume
42
Multiple Choice
Recall;
What does 'boiling point' mean?
The temperature at which a solid melts / turns to liquid
The temperature at which a liquid boils / turn to vapor
The amount of space occupied by an object
How much mass something has for its volume
43
Multiple Choice
Recall;
What does 'density' mean?
The temperature at which a solid melts / turns to liquid
The temperature at which a liquid boils / turn to vapor
The amount of space occupied by an object
How much mass something has for its volume
44
Multiple Choice
Recall;
What does 'volume' mean?
The temperature at which a solid melts / turns to liquid
The temperature at which a liquid boils / turn to vapor
The amount of space occupied by an object
How much mass something has for its volume
45
Multiple Choice
The name given to vertical columns in a periodic table is;
Groups
Periods
Compounds
Bases
46
Multiple Choice
Can you find Palladium (Pd) in the periodic table in your textbook? Which other elements are in the same group as Palladium?
Ni = Nickel
Pd = Palladium
Pt = Platinum
Rh = Rhodium
Pd = Palladium
Ag = Silver
Co = Cobalt
Pd = Palladium
Au = Gold
Ir = Iridium
Pd = Palladium
Cu = Copper
47
Multiple Choice
In the periodic table, the melting point increases as you go down the group (vertical column). Based on this information, which of the following is correct about the melting points of the Palladium group?
Nickel > Palladium > Platinum
Nickel < Palladium < Platinum
Nickel = Palladium = Platinum
48
Multiple Choice
In the periodic table, the melting point increases as you go down the group (vertical column).
Look at the copper group in your periodic table. Which metal in the group has the lowest melting point?
Copper
Silver
Gold
49
Multiple Choice
In the periodic table, the melting point increases as you go down the group (vertical column).
Look at the copper group in your periodic table. Which metal in the group has the highest melting point?
Copper
Silver
Gold
50
Multiple Choice
In the periodic table, the melting point increases as you go down the group (vertical column).
True of False?
Cobalt has a higher melting point than Iridium.
TRUE
FALSE
51
Fill in the Blanks
Type answer...
52
Fill in the Blanks
Type answer...
53
Multiple Choice
Page 63; Look at the bar charts;
Which of the following is the correct description of the pattern of melting points going across?
melting points increase then they decrease
melting points increase from left to right for the first four elements, the melting points of the other elements are low
melting points in the periods have a similar pattern to melting points in a group
melting points are all the same
54
Multiple Choice
What is the difference between groups and periods in the periodic table?
Groups are vertical columns, Periods are horizontal rows.
Periods are vertical columns, periods are Groups rows.
55
Multiple Choice
What can be said about the patterns of the melting points in groups VS periods.
Melting point in groups increases down the column, melting point in periods increases for first four elements only, then decreases
The pattern is the same for groups and columns, the melting point increases in both cases
In groups, the melting point increases left to right, in columns, it increases down the column
There are more elements in a period compared to a group.
56
Multiple Choice
One of the differences between groups and periods is;
Groups have more elements than periods
Groups have less elements than periods
Groups only have metals
Periods have both metals and non metals
57
Multiple Choice
Page
How many periods are in the periodic table?
4
5
6
7
58
Multiple Choice
How many elements are in the first period of the periodic table?
1
2
8
6
59
Multiple Choice
How many elements are in the first group of the periodic table?
1
2
4
6
60
Poll
I know this for sure! - so far..
Columns = Groups
Rows = Periods
Metals = alkaline solutions
Non metals = Acidic solutions
In the columns (groups), Melting point increases down the group
In the rows (periods), melting point increases for the first four elements
61
ALL DONE!
- BUT, THERE'S HW!!!!
GO TO GOOGLE CLASSROOM AND COMPLETE THE GRAPHING ACTIVITY FOR HW
Recap & Review
What did we look at yesterday?
Complete these review questions to jog your memory about yesterday's class.
Show answer
Auto Play
Slide 1 / 61
SLIDE
Similar Resources on Wayground

56 questions
Classification
Presentation
•
7th Grade

59 questions
Yo go verbs & Tener
Presentation
•
7th Grade

53 questions
Chapter 2 Section 1: Cell Structure
Presentation
•
7th Grade

55 questions
Chemistry STAAR review 2023
Presentation
•
8th Grade

56 questions
Weathering Part 1
Presentation
•
6th Grade

53 questions
Law of Conservation of Mass
Presentation
•
8th Grade

56 questions
Phase changes and States of Matter
Presentation
•
8th Grade

55 questions
Fertilization and Development PowerPoint Lesson
Presentation
•
7th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

20 questions
genetics, punnett squares, heredity
Quiz
•
7th Grade

20 questions
Food Webs + Energy Pyramids
Quiz
•
7th Grade

10 questions
Exploring Weather Influences and Map Reading
Interactive video
•
6th - 10th Grade

43 questions
Amplify Earth's Changing Climate Unit Review
Quiz
•
6th - 8th Grade

62 questions
Super Science Trivia
Quiz
•
6th - 8th Grade