

Chemistry STAAR review 2023
Presentation
•
Science
•
8th Grade
•
Medium
+1
Standards-aligned
Joseph F Mcguinness
Used 23+ times
FREE Resource
10 Slides • 45 Questions
1
Do now:
→ Join quizizz
→ Read warm-up questions below:
What is an element?
Where do we find information on the elements?

2
Multiple Choice

What is an element?
A substance that is made up of one type of atom and can't be reduced to simpler substances.
A substance made of two or more different atoms chemically bonded to one another. They can only be destroyed by chemical processes.
A material containing two or more elements or compounds that are in close contact and are mixed in any proportion. They can be separated by physical means.
3
Multiple Choice

Where can you find examples of Elements?
Dictionary
Periodic Table of Compounds
Periodic Table of Elements
Your Neighbor
4


ELEMENTS AND THE PERIODIC TABLE ARE BROKEN UP INTO THREE TYPES
5
Multiple Choice

The blue elements (left side) are called
metals
nonmetals
metalloids
noble gases
6
Multiple Choice

The yellow elements (right side) are called
metals
nonmetals
metalloids
noble gases
7
Multiple Choice

metals
nonmetals
metalloids
noble gases
8
Physical Properties of Metals
Luster - Shines, reflects light
Great Conductivity - How well electricity and heat travels through the element
Malleable - Can be beaten into different shapes without breaking
Ductile - Turned into wires
Solid at room temperature - Except for Mercury

9
Multiple Choice

Luster
how an object reflects light
can be hammered into a shape
transfers heat and/or electricity
10
Multiple Choice

Malleability
how an object reflects light
can be hammered into a shape
transfers heat and/or electricity
11
Multiple Select

Check off all properties of metal you can observe in this image of US coins.
Luster
Malleable
Conductivity
Ductile
Solid at room temperature
12
Multiple Choice

Conductivity
how an object reflects light
can be hammered into a shape
transfers heat and/or electricity
13
Multiple Choice

Manganese is shiny, conducts heat and electricity, and can be hammered into thin sheets. What is is?
Nonmetal
Metal
Metalloid
None of the above
14
Properties of Nonmetals
Essentially, they are the opposite of metals
Dull, not shiny
Brittle when solid
Some are gasses
NOT malleable, conductive, or ductile

15
Multiple Choice

Neon is colorless, a poor conductor of heat and is not able to be stretched. How would it be classified?
Nonmetal
Metalloid
Metal
None of the above
16
Multiple Select

Check off all of the nonmetal physical properties you can observe in this image of sulfur.
Dull appearance
Brittle solid
Gaseous
Not malleable
17
Properties of Metalloids
Properties are between metals and nonmetals
Can have a metallic luster, or be dull
Conductive, but less than metals
Solid at room temperature, but more brittle than metals
Usually malleable and ductile

18
Multiple Choice

Metal
Nonmetal
Metalloid
19
Multiple Choice

Titanium
Selenium
Potassium
Polonium
20

THE PERIODIC TABLE IS ALSO ORGANIZED BY GROUPS, PERIODS, AND MORE
WATCH THE VIDEO TO REMEMBER
21
Multiple Choice

period
group
cluster
unit
22
Multiple Choice

Horizontal Rows
Vertical Columns
23
Multiple Choice

Horizontal Rows
Vertical Columns
24
Multiple Select

Elements in the same group have : (2 correct answers)
Similar chemical properties
Similar names
Same number of outer (valence) electrons)
The same number of protons
25
Multiple Choice

How many periods are on the periodic table?
5
6
7
8
26
Multiple Choice

Same group
Same period
27
Multiple Choice

Compound formulas have ____ capital letter(s).
1
2
3
more than 1
28
Multiple Choice

Element symbols have _____ capital letter(s).
1
2
3
4
29
The electron configuration of Group 8 elements completely fill the valence shell
The filled valence shell makes Group 8 element very stable (NON-REACTIVE). They are called "noble gases".
All elements want to have Noble Gas Electron Configuration.

30
Multiple Choice

Helium
Argon
Nitrogen
Krypton
31
Multiple Choice

The reason that Noble Gases do not react is because
Their outer electron shell is filled
Their inner electron shell is filled
They are able to bond to fill their outer shell
None of the above, Noble Gases are highly reactive
32
Multiple Select

select the (ONE) property of noble gases that apply
non reactive
low ionization energy
high boiling point
can never conduct electricity
33

Elements are Made of Atoms
34
Draw
Draw where the three subatomic particles go
35
Multiple Choice

protons, neutrons, and isotopes
neutrons, isotopes, and electrons
positives, negatives, and electrons
protons, neutrons, and electrons
36
Multiple Choice

Neutrons only
Electrons only
Protons and Neutrons
Protons and Electrons
37
Multiple Choice

What two particles are in the nucleus of an atom?
protons and electrons
electrons and neutrons
protons and neutrons
38
Multiple Choice

nucleus
electron cloud
core
center
39
Multiple Choice
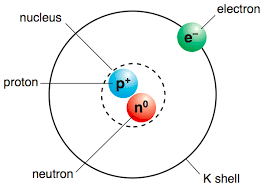
Protons, Neutrons, Electrons
Protons only
Protons and Electrons
Protons and Neutrons
40
Multiple Choice

Proton
Ion
Neutron
Electron
41
Multiple Choice

electrons
positrons
neutrons
protons
42

The periodic table provides the number of Protons, Electrons, and Neutrons for each element
43
Multiple Choice

The atomic number is the number of ____ in the nucleus. It's the element's "identification"!
electrons
neutrons
protons
44
Multiple Choice

What particle(s) make up the mass number?
electrons
neutrons
protons
protons and neutrons
45
Multiple Choice

What is the mass number of Phosphorous (P)?
15
30
31
16
46
Dropdown

E=
N=
47
Draw
Draw the Bohr Model for Lithium
48




VALENCE ELECTRONS
49
Multiple Choice

This is a Bohr Diagram of Aluminum. How many valence electrons does Aluminum have?
3
4
5
50
Multiple Choice

How many valence electrons does Lithium (Li) have?
8
1
5
2
51
Multiple Choice

How many valence electrons does Tin (Sn) have?
8
7
5
4
52
Multiple Choice

How many valence electrons does Bromine (Br) have?
5
6
7
8
53
Multiple Choice

an electron that is found in the outermost shell of an atom.
an electron found in the innermost shell of an atom.
an electron found in the middle shell.
54
Multiple Choice

Which element is this?
Nickel
Neon
Sodium
Nitrogen
55
Multiple Choice

How many valence electrons?
2
3
4
5
Do now:
→ Join quizizz
→ Read warm-up questions below:
What is an element?
Where do we find information on the elements?

Show answer
Auto Play
Slide 1 / 55
SLIDE
Similar Resources on Wayground

51 questions
Respiratory System 6.2
Presentation
•
8th Grade

49 questions
8th Grade 1-6 Scientific Measurement & Math
Presentation
•
8th Grade

49 questions
8.5C Periodic Table (Identifying Elements)
Presentation
•
8th Grade

49 questions
STAAR Review Day 4
Presentation
•
8th Grade

53 questions
Cellular Respiration
Presentation
•
9th Grade

49 questions
Chemical Reaction ( Change, Properties , Law COM) -EOG 2022
Presentation
•
8th Grade

51 questions
7.10B Volcanoes & Hot Spots Research Notes Lesson
Presentation
•
8th Grade

50 questions
Natural Selection, Variation, and Adaptation
Presentation
•
8th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

30 questions
GMAS Physical Science Review
Quiz
•
8th Grade

10 questions
Exploring the Rock Cycle
Interactive video
•
6th - 8th Grade

18 questions
Carbon Cycle
Quiz
•
8th Grade

10 questions
Exploring Earth's Seasons and Their Causes
Interactive video
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

10 questions
Evidence of Evolution
Presentation
•
6th - 8th Grade

20 questions
8th Grade Science NC EOG Review
Quiz
•
8th Grade