

Periodic Table Review Lesson
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
Standards-aligned
Christine Ruberti
Used 1+ times
FREE Resource
16 Slides • 15 Questions
1

2
3

4

5

6
Multiple Choice
The vertical (up and down) columns in the Periodic Table are called
groups
towers
periods
atomic numbers
7
Multiple Choice
Elements in a .................. have similar chemical properties.
period
group
row
8

9

10
Multiple Choice
Which group of the periodic table is composed of inert (not reactive) gases?
answer choices
alkali metals
alkaline earth metals
halogens
noble gases
11

12

13

14
Multiple Choice
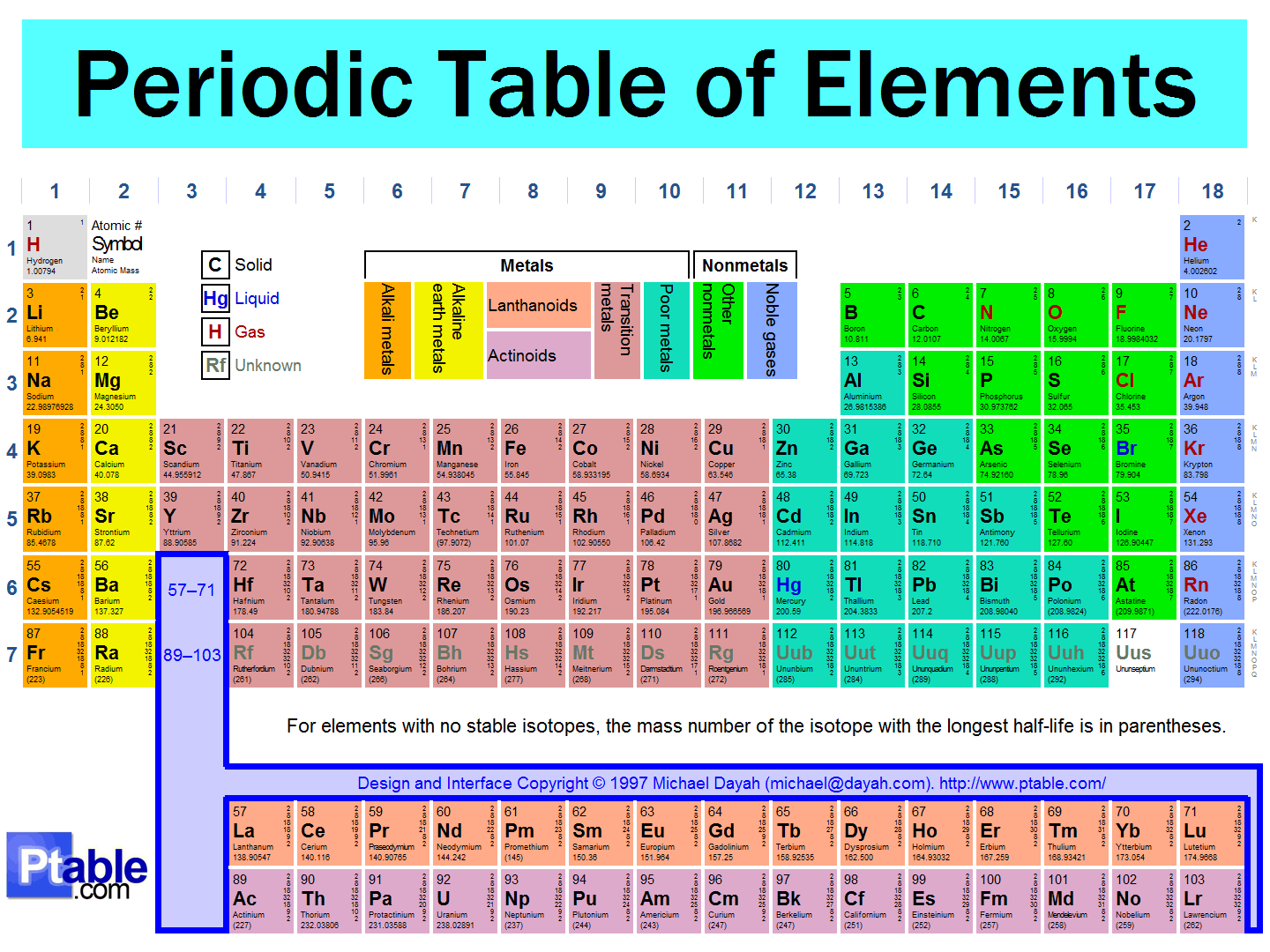
15
Multiple Choice

Hydrogen
atomic number
atomic mass
atomic explosion
16
Multiple Choice
Atomic Mass equals the combined number of
Protons and Electrons
Electrons and Neutrons
Protons and Neutrons
17
Multiple Choice
The number of valence electrons ________ by 1 as you move across the period.
increases
decreases
18

Parts of the Elemental Symbol
Atomic Number
# of protons and electrons
number on top of symbol
Atomic Mass
average # of protons + # of neutrons number below the symbol
Atomic Symbol
capital letter and lowercase letter that represents the element name
19
Multiple Choice

What is Neon's atomic mass?
10
20.18
20
Multiple Choice

What is Beryllium's atomic number?
4
5
9.01
21
Multiple Choice

The fluorine atom has an atomic number of
19
9
22

23

24

25
26
Multiple Choice

The atoms along the staircase are called
metals
nonmetals
metalloids
noble gases
27
Multiple Choice

The majority (most) of the elements are:
metals
nonmetals
metalloids
28
Multiple Choice

The elements on the right side of the periodic table of elements are mainly:
metals
nonmetals
metalloids
29
Multiple Choice
Why are noble gases not reactive
Too few electrons
Too many electrons
They have a full set of electrons
They need more electrons
30
the electrons in the outermost energy shell
number of valence electrons matches group number
(without transition metals)
electrons involved in bonding
ions created when valence electrons are gained or lost
Valence Electrons and Reactivity
31
Multiple Choice
Elements in a ________ have similar chemical properties.
period
group
row

Show answer
Auto Play
Slide 1 / 31
SLIDE
Similar Resources on Wayground

20 questions
Properties of Matter Notes
Presentation
•
9th - 12th Grade

21 questions
Naming Covalent (Molecular) Compounds
Presentation
•
10th - 12th Grade

23 questions
Chemical Equations & Balancing
Presentation
•
9th - 12th Grade

24 questions
Empirical, Molecular and Percent Comp.
Presentation
•
10th - 12th Grade

25 questions
Bohr Models
Presentation
•
10th - 12th Grade

21 questions
Scientific Method, Laws, and Theories
Presentation
•
9th - 12th Grade

24 questions
Significant Figures and Calculations - A/B
Presentation
•
9th - 12th Grade

22 questions
Writing and Naming Molecular Compounds
Presentation
•
8th - 12th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Unit 7 Chemical Reactions
Quiz
•
10th Grade

25 questions
Unit 8 Stoichiometry Review
Quiz
•
10th Grade

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

15 questions
Nuclear Chemistry: Balancing
Quiz
•
10th Grade

20 questions
Naming Acids and Bases
Quiz
•
10th Grade

10 questions
Exploring Stoichiometry in Chemistry
Interactive video
•
6th - 10th Grade