

100 Ways to Pass the Chemistry Regents
Presentation
•
Chemistry
•
10th Grade
•
Medium
Pravesh Shiwnarain
Used 2+ times
FREE Resource
101 Slides • 134 Questions
1

2
Fill in the Blanks
Type answer...
3

4
Multiple Choice
Matter is classified as a
substance, only
substance or as a mixture of substances
homogenous mixture, only
homogenous mixture or as a heterogeneous mixture
5
Multiple Choice
A compound is a substance composed of two or more elements that are
physically mixed in a fixed proportion
physically mixed in a variable proportion
chemically combined in a fixed proportion
chemically combined in a variable proportion
6

7
Multiple Choice
In a chemical reaction, there is conservation of
energy, volume, and mass
energy, volume, and charge
mass, charge, and energy
mass, charge, and volume
8

9
Multiple Choice
Which statement describes a chemical property of iron?
Iron can be flattened into sheets.
Iron conducts electricity and heat.
Iron combines with oxygen to form rust.
Iron can be drawn into a wire.
10

11
Multiple Choice
Which substance can be broken down by chemical means?
ammonia
aluminum
antimony
argon
12

13
Multiple Choice
Equal amounts of ethanol and water are mixed at room temperature and at
101.3 kPa. Which process is used to separate ethanol from the mixture?
distillation
reduction
filtration
ionization
14
Multiple Choice
A beaker contains both alcohol and water. These liquids can be separated by distillation because the liquids have different
boiling points
densities
particle size
solubilities
15

16
Multiple Choice
At STP, solid carbon can exist as graphite or as diamond. These two forms of carbon have
the same properties and the same crystal structures
the same properties and different crystal structures
different properties and the same crystal structures
different properties and different crystal structures
17

18
Multiple Choice
According to the kinetic molecular theory for an ideal gas, all gas particles
are in random, constant, straight-line motion
are separated by very small distances relative to their sizes
have strong intermolecular forces
have collisions that decrease the total energy of the system
19
Multiple Choice
According to the kinetic molecular theory, which statement describes the particles of an ideal gas?
The gas particles are arranged in a regular pattern.
The force of attraction between the gas particles is strong.
The gas particles are hard spheres in continuous circular motion.
The collisions of the gas particles may result in the transfer of energy.
20

21
Multiple Choice
Under which conditions of temperature and pressure does carbon dioxide gas behave most like an ideal gas?
low temperature and low pressure
low temperature and high pressure
high temperature and low pressure
high temperature and high pressure
22
Multiple Choice
Under which conditions of temperature and pressure would a 1-liter sample of a real gas behave most like an ideal gas?
100 K and 0.1 atm
100 K and 10 atm
500 K and 0.1 atm
500 K and 10 atm
23

24
Multiple Choice
The amount of randomness of the atoms in a system is an indication of the
entropy of the system
polarity of the system
excited state of the atoms
ground state of atoms
25
Multiple Choice
Chemical systems in nature tend to undergo changes toward
lower energy and lower entropy
lower energy and higher entropy
higher energy and lower entropy
higher energy and higher entropy
26
Multiple Choice
The entropy of a sample of carbon dioxide increases as the carbon dioxide changes from
gas to liquid
gas to solid
liquid to solid
solid to gas
27

28
Multiple Choice
At the same temperature and pressure, 1.0 liter of CO(g) and 1.0 liter of NO(g) have
qual masses and the same number of molecules
different masses and a different number of molecules
equal volumes and the same number of molecules
different volumes and a different number of molecules
29

30
Multiple Choice
According to Reference Table H, what is the vapor pressure of propanone at 45°C?
22 kPa
33 kPa
70 kPa
98 kPa
31

32
Multiple Choice
The temperature of a sample of matter is a measure of the
average potential energy of the particles of the sample
average kinetic energy of the particles of the sample
total nuclear energy of the sample
total thermal energy of the sample
33

34
Multiple Choice
A few pieces of dry ice (solid carbon dioxide), at -78°C are placed in a flask that contains air at 21°C. The flask is sealed by placing an uninflated balloon over the mouth of the flask. As the balloon inflates, the dry ice disappears and no liquid is observed in the flask.
State the direction of heat flow that occurs between the dry ice and the air in the flask.
flask to the air
air to the flask
35

36
Fill in the Blanks
37

38
Multiple Choice
At standard pressure, which element has a melting point higher than standard temperature?
F2
Br2
Fe
Hg
39

40
Fill in the Blanks
41
Multiple Choice
In which process does a solid change directly into a vapor?
condensation
deposition
solidification
sublimation
42

43
Multiple Choice
According to the wave-mechanical model, an orbital is defined as the most probable location of
a proton
a neutron
a positron
an electron
44

45
Multiple Choice
Which statement describes the location of two types of subatomic particles in a helium atom?
Protons and neutrons are located in the nucleus.
Protons and neutrons are located outside the nucleus.
Protons and electrons are located in the nucleus.
Protons and electrons are located outside the nucleus.
46
Multiple Choice
What is the charge of the nucleus of a copper atom?
+1
+2
+29
+64
47
Multiple Choice
An atom that contains six protons, six neutrons, and six electrons has a mass of approximately
12 u
12 g
18 u
18 g
48

49
Multiple Choice
Positrons and beta particles have
the same charge and the same mass
the same charge and different masses
different charges and the same mass
different charges and different masses
50

51

52
Multiple Choice
An atom is electrically neutral because the
number of protons equals the number of electrons
number of protons equals the number of neutrons
ratio of the number of neutrons to the number of electrons is 1:1
ratio of the number of neutrons to the number of protons is 2:1
53

54
Multiple Choice
If two atoms are isotopes of the same element, the atoms must have
the same number of protons and the same number of neutrons
the same number of protons and a different number of neutrons
a different number of protons and the same number of neutrons
a different number of protons and a different number of neutrons
55

56
Multiple Choice
The atomic mass of an element is the weighted average of the
number of protons in the isotopes of that element
number of neutrons in the isotopes of that element
atomic numbers of the naturally occurring isotopes of that element
atomic masses of the naturally occurring isotopes of that element
57
Multiple Choice

The table below gives the atomic mass and the abundance of the two naturally occurring isotopes of bromine.
Which numerical setup can be used to calculate the atomic mass of the element bromine?
(78.92 u)(50.69) + (80.92 u)(49.31)
(78.92 u)(49.31) + (80.92 u)(50.69)
(78.92 u)(0.5069) + (80.92 u)(0.4931)
(78.92 u)(0.4931) + (80.92 u)(0.5069)
58

59
Multiple Choice
Which electron configuration represents the electrons in an atom of calcium in an excited state?
2–8–8
2–8–8–2
2–7–8–1
2–7–8–3
60
Multiple Choice
Which electron configuration represents the electrons of an atom in an excited state?
2-1
2-7-4
2-8-7
2-4
61
Multiple Choice
When an excited electron in an atom moves to the ground state, the electron
absorbs energy as it moves to a higher energy state
absorbs energy as it moves to a lower energy state
emits energy as it moves to a higher energy state
emits energy as it moves to a lower energy state
62
Multiple Choice
Which electron transition in an excited atom results in a release of energy?
first shell to the third shell
second shell to the fourth shell
third shell to the fourth shell
fourth shell to the second shell
63

64

65
Multiple Choice
A specific amount of energy is emitted when excited electrons in an atom in a sample of an element return to the ground state. This emitted energy can be used to determine the
mass of the sample
volume of the sample
identity of the element
number of moles of the element
66
Multiple Choice

Given the bright-line spectra of three elements and the spectrum of a mixture formed from at least two of these elements:
Which elements are present in this mixture?
E and D, only
E and G, only
D and G, only
D, E, and G
67

68
Multiple Choice
Which type of reaction occurs when a high energy particle collides with the nucleus of an atom, converting that atom to an atom of a different element?
addition
neutralization
substitution
transmutation
69
Multiple Choice

Which nuclear equation represents a natural transmutation?
Choice 1
Choice 2
Choice 3
Choice 4
70

71
Multiple Choice
Which radioisotopes have the same decay mode and have half-lives greater than 1 hour?
Au-198 and N-16
Ca-37 and Fe-53
I-131 and P-32
Tc-99 and U233
72

73
Multiple Choice
The amount of energy released from a fission reaction is much greater than the energy released from a chemical reaction because in a fission reaction
mass is converted into energy
energy is converted into mass
ionic bonds are broken
covalent bonds are broken
74
Multiple Choice
In which type of reaction do two lighter nuclei combine to form one heavier nucleus?
combustion
reduction
nuclear fission
nuclear fusion
75
Multiple Choice

Which balanced equation represents nuclear fusion?
Choice 1
Choice 2
Choice 3
Choice 4
76
Multiple Choice

Which type of nuclear reaction is represented by the equation?
fission
fusion
beta decay
alpha decay
77
Multiple Choice
A nuclear fission reaction and a nuclear fusion reaction are similar because both reactions
form heavy nuclides from light nuclides
form light nuclides from heavy nuclides
release a large amount of energy
absorb a large amount of energy
78

79

80
Multiple Choice
Which radioisotope is used to determine the age of once-living organisms?
carbon-14
cobalt-60
iodine-131
uranium-238
81
Multiple Choice
Which nuclide is used to investigate human thyroid gland disorders
carbon-14
potassium-37
cobalt-60
iodine-131
82
Multiple Choice
Which radioactive isotope is used in treating cancer?
carbon-14
cobalt-60
lead-208
uranium-239
83

84
Multiple Choice
One benefit of nuclear fission reactions is
nuclear reactor meltdowns
storage of waste materials
biological exposure
production of energy
85

86
Multiple Choice
On the present Periodic Table of the Elements, the elements are arranged according to increasing
number of oxidation states
number of neutrons
atomic mass
atomic number
87

88
Multiple Choice
What is a property of most metals?
hey tend to gain electrons easily when bonding.
They tend to lose electrons easily when bonding.
They are poor conductors of heat.
high electronegativities
89
Multiple Choice
The high electrical conductivity of metals is primarily due to
high ionization energies
filled energy levels
mobile electrons
high electronegativities
90

91
Multiple Choice
Which two characteristics are associated with metals?
low first ionization energy and low electronegativity
first ionization energy and high electronegativity
high first ionization energy and low electronegativity
high first ionization energy and high electronegativity
92
Multiple Choice
In the ground state, an atom of which element has seven valence electrons?
sodium
phosphorus
nitrogen
fluorine
93

94
Multiple Choice
Which list of elements consists of a metal, a metalloid, and a nonmetal?
Li, Na, Rb
Cr, Mo, W
Sn, Si, C
O, S, Te
95
Multiple Choice
Which list of elements contains a metal, a metalloid, a nonmetal, and a noble gas?
Be, Si, Cl, Kr
C, N, Ne, Ar
K, Fe, B, F
Na, Zn, As, Sb
96

97
Multiple Choice
Strontium and barium have similar chemical properties because atoms of these elements have the same number of
protons
neutrons
electron shells
valence electrons
98

99
Multiple Choice
When an atom of lithium loses an electron, the atom becomes a
negative ion with a radius smaller than the radius of the atom
negative ion with a radius larger than the radius of the atom
positive ion with a radius smaller than the radius of the atom
positive ion with a radius larger than the radius of the atom
100
Multiple Choice
What occurs when an atom of chlorine forms a chloride ion?
The chlorine atom gains an electron, and its radius becomes smaller
The chlorine atom gains an electron, and its radius becomes larger.
The chlorine atom loses an electron, and its radius becomes smaller.
The chlorine atom loses an electron, and its radius becomes larger
101

102
Multiple Choice
Which term indicates how strongly an atom attracts the electrons in a chemical bond?
alkalinity
atomic mass
electronegativity
activation energy
103

104
Multiple Choice
The amount of energy required to remove the outermost electron from a gaseous atom in the ground state is known as
first ionization energy
activation energy
conductivity
electronegativity
105

106

107
Multiple Choice
What occurs when an atom of chlorine and an atom of hydrogen become a molecule of hydrogen chloride?
A chemical bond is broken and energy is released.
A chemical bond is broken and energy is absorbed.
A chemical bond is formed and energy is released.
A chemical bond is formed and energy is absorbed.
108

109
Multiple Choice
What occurs when potassium reacts with chlorine to form potassium chloride?
Electrons are shared and the bonding is ionic.
Electrons are shared and the bonding is covalent.
Electrons are transferred and the bonding is ionic.
Electrons are transferred and the bonding is covalent.
110

111

112
Multiple Choice
Which elements can react to produce a molecular compound?
calcium and chlorine
hydrogen and sulfur
lithium and fluorine
magnesium and oxygen
113

114

115
Multiple Choice
Which type of bonding is found in all molecular substances?
covalent bonding
hydrogen bonding
ionic bonding
metallic bonding
116

117

118

119
Multiple Choice
Which statement explains why a C–O bond is more polar than a F–O bond?
At STP, carbon has a greater density than fluorine.
A carbon atom has more valence electrons than a fluorine atom.
The difference in electronegativity between carbon and oxygen is greater than that between fluorine and oxygen.
The difference in first ionization energy between carbon and oxygen is greater than that between fluorine and oxygen.
120

121
Multiple Choice

Which formula represents a nonpolar molecule containing polar covalent bonds?
Choice 1
Choice 2
Choice 3
Choice 4
122

123
Multiple Choice
The relatively high boiling point of water is due to water having
hydrogen bonding
metallic bonding
nonpolar covalent bonding
strong ionic bonding
124
Multiple Choice
Hydrogen bonding is a type of
strong covalent bond
weak ionic bond
strong intermolecular force
weak intermolecular force
125

126
Multiple Choice
Which compound has hydrogen bonding between its molecules?
CH4
CaH2
KH
NH3
127

128
Multiple Choice

Which type of bonding is present when valence electrons move within the sample?
metallic bonding
hydrogen bonding
covalent bonding
ionic bonding
129
Multiple Choice
Which list consists of types of chemical formulas?
atoms, ions, molecules
metals, nonmetals, metalloids
empirical, molecular, structural
synthesis, decomposition, neutralization
130
Multiple Choice
Which formula is an empirical formula?
CH4
C2H6
C3H6
C4H10
131
Fill in the Blanks
132
Fill in the Blanks
133

134
Multiple Choice

Which type of chemical reaction is represented by this equation?
synthesis
decomposition
single replacement
double replacement
combustion
135
Multiple Choice

Which types of chemical reactions are represented by these equations?
single replacement and decomposition
single replacement and double replacement
synthesis and decomposition
synthesis and double replacement
136
Multiple Choice

Which type of chemical reaction is represented by this equation?
synthesis
decomposition
single replacement
double replacement
combustion
137

138

139
Multiple Choice
What is the chemical formula for copper(II) hydroxide?
CuOH
CuOH2
Cu2(OH)
Cu(OH)2
140

141

142

143
Multiple Choice
At 298 K and 1 atm, which noble gas has the lowest density?
Ne
Kr
Xe
Rn
144
Multiple Choice
In a laboratory activity, the density of a sample of vanadium is determined to be 6.9 g/cm3 at room temperature. What is the percent error for the determined value?
0.15%
0.87%
13%
15%
145

146
Multiple Choice

If 46 grams of Na and 71 grams of Cl2 react completely, what is the total mass of NaCl produced?
58.5 g
117 g
163 g
234 g
147

148
Multiple Choice
The molar mass of Ba(OH)2
154.3 g
155.3 g
171.3 g
308.6 g
149
Fill in the Blanks
150

151
Math Response
Calculate the percent composition by mass of water in CaSO4 • 2H2O.
__%
152
Multiple Choice
A hydrate is a compound that includes water molecules within its crystal structure. During an experiment to determine the percent by mass of water in a hydrated crystal, a student found the mass of the hydrated crystal to be 4.10 grams. After heating to constant mass, the mass was 3.70 grams. What is the percent by mass of water in this crystal?
90%
11%
9.8%
0.40%
153

154

155

156
Multiple Choice

What is the total number of moles of O2(g) that must react completely with 5.00 moles of C4H10(g)?
10.0
20.0
26.5
32.5
157

158

159

160
Multiple Choice
What is the molarity of a solution of NaOH if 2 liters of the solution contains 4 moles of NaOH?
0.5 M
2 M
8 M
80 M
161

162

163
Multiple Choice
At standard pressure, how do the boiling point and the freezing point of NaCl(aq) compare to the boiling point and the freezing point of H2O(ℓ)?
Both the boiling point and the freezing point of NaCl(aq) are lower.
Both the boiling point and the freezing point of NaCl(aq) are higher.
The boiling point of NaCl(aq) is lower, and the freezing point of NaCl(aq) is higher.
The boiling point of NaCl(aq) is higher, and the freezing point of NaCl(aq) is lower.
164

165
Multiple Choice
A 5.0-gram sample of Fe(s) is to be placed in 100. milliliters of HCl(aq). Which changes will result in the fastest rate of reaction?
increasing the surface area of Fe(s) and increasing the concentration of HCl(aq)
increasing the surface area of Fe(s) and decreasing the concentration of HCl(aq
decreasing the surface area of Fe(s) and increasing the concentration of HCl(aq)
decreasing the surface area of Fe(s) and decreasing the concentration of HCl(aq)
166
Multiple Choice
A chemical reaction occurs when reactant particles
are separated by great distances
have no attractive forces between them
collide with proper energy and proper orientation
convert chemical energy into nuclear energy
167

168

169

170

171

172
Multiple Choice
Which term is defined as the difference between the potential energy of the products and the potential energy of the reactants in a chemical reaction?
activation energy
thermal energy
heat of fusion
heat of reaction
173

174
Multiple Choice
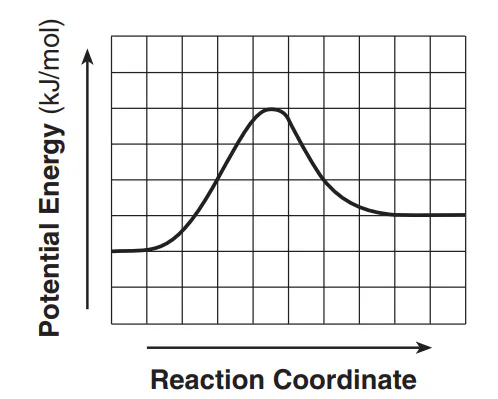
Each interval on the axis labeled “Potential Energy (kJ/mol)” represents 10. kilojoules per mole. What is the activation energy of the forward reaction?
10. kJ/mol
30 kJ/mol
40. kJ/mol
60. kJ/mol
175
Multiple Choice

Determine if the reaction is endothermic or exothermic.
endothermic
exothermic
176

177
Multiple Choice
Addition of a catalyst can speed up a reaction by providing an alternate reaction pathway that has a
lower activation energy
higher activation energy
lower heat of reaction
higher heat of reaction
178

179
Multiple Choice
Some solid KNO3 remains at the bottom of a stoppered flask containing a saturated KNO3(aq) solution at 22°C. Which statement explains why the contents of the flask are at equilibrium?
The rate of dissolving is equal to the rate of crystallization.
The rate of dissolving is greater than the rate of crystallization.
The concentration of the solid is equal to the concentration of the solution
The concentration of the solid is greater than the concentration of the solution.
180
Multiple Choice
Which statement must be true when solution equilibrium occurs?
The solution is at STP.
The solution is supersaturated.
The concentration of the solution remains constant.
The masses of the dissolved solute and the undissolved solute are equal.
181

182
Multiple Choice
Which type of substance yields hydrogen ions, H+, in an aqueous solution?
an Arrhenius acid
an Arrhenius base
183

184

185
Multiple Choice
Which statement describes one acid-base theory?
An acid is an H+ acceptor, and a base is an H+ donor.
An acid is a H+ donor, and a base is an H+ acceptor
186

187
Multiple Choice
Which type of reaction occurs when an Arrhenius acid reacts with an Arrhenius base to form a salt and water?
combustion
decomposition
neutralization
saponification
188

189
Fill in the Blanks
190

191
Multiple Choice
Which indicator is blue in a solution that has a pH of 5.6?
bromcresol green
bromthymol blue
methyl orange
thymol blue
192

193
Multiple Choice
Which process uses a volume of solution of known concentration to determine the concentration of another solution?
distillation
substitution
transmutation
titration
194
Fill in the Blanks
195

196
Multiple Choice
Which statement describes an electrolyte?
An electrolyte conducts an electric current as a solid and dissolves in water.
An electrolyte conducts an electric current as a solid and does not dissolve in water.
When an electrolyte dissolves in water, the resulting solution conducts an electric current.
When an electrolyte dissolves in water, the resulting solution does not conduct an electric current.
197

198
Multiple Choice
When a sample of Mg(s) reacts completely with O2(g), the Mg(s) loses 5.0 moles of electrons. How many moles of electrons are gained by the O2(g)?
1.0 M
2.5 M
5.0 M
10.0 M
199

200
Multiple Choice
Which metal reacts spontaneously with NiCl2(aq)?
Au(s)
Cu(s
Sn(s)
Zn(s)
201

202
Multiple Choice
Which energy change occurs in an operating voltaic cell?
chemical to electrical
electrical to chemical
chemical to nuclear
nuclear to chemical
203
Multiple Choice

What is the direction of electron flow in the external circuit in this operating cell.
Cu(s) electrode to Ag(s) electrode
Ag(s) electrode to Cu(s) electrode
204
Multiple Choice
Which statement describes the reactions in an electrochemical cell (voltaic and electrolytic)?
Oxidation occurs at the anode, and reduction occurs at the cathode.
Oxidation occurs at the cathode, and reduction occurs at the anode.
Oxidation and reduction both occur at the cathode.
Oxidation and reduction both occur at the anode.
205
Multiple Choice
An electrolytic cell differs from a voltaic cell because an electrolytic cell
generates its own energy from a spontaneous physical reaction
generates its own energy from a nonspontaneous physical reaction
requires an outside energy source for a spontaneous chemical reaction to occur
requires an outside energy source for a nonspontaneous chemical reaction to occur
206
Multiple Choice
When a voltaic cell operates, ions move through the
anode
cathode
salt bridge
external circuit (wire)
207

208

209

210
Multiple Choice
All organic compounds must contain the element
phosphorus
oxygen
carbon
nitrogen
211

212
Multiple Choice
The only two elements in alkenes and alkynes are
carbon and nitrogen
carbon and hydrogen
oxygen and nitrogen
oxygen and hydrogen
213
Multiple Choice

This compound is classified as an
aldehyde
alkene
alkyne
alcohol
214
Multiple Choice
Which formula represents an alkene?
C2H6
C3H6
C4H10
C5H12
215

216
Multiple Choice
Which formula represents an unsaturated hydrocarbon?
C2H4
C3H8
C4H10
C5H12
217
Multiple Choice
Which formula represents an unsaturated hydrocarbon?
218

219
Multiple Choice
An alcohol and an ether have the same molecular formula, C2H6O. These two compounds have
the same functional group and the same physical and chemical properties
the same functional group and different physical and chemical properties
different functional groups and the same physical and chemical properties
different functional groups and different physical and chemical properties
220
Multiple Choice
Which class of organic compounds has molecules that contain nitrogen atoms?
alcohol
amine
ether
ketone
221
Multiple Choice
The organic compound represented by the condensed structural formula CH3CH2CH2CHO is classified as an
alcohol
aldehyde
ester
ether
222
Multiple Choice

This compound is classified as an
amide
amine
aldehyde
alcohol
223
Multiple Choice
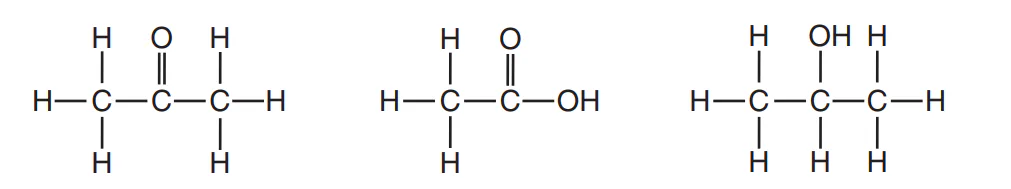
Which organic-compound classes are represented by these structural formulas, as shown from left to right?
ester, organic acid, ketone
ester, aldehyde, organic acid
ketone, aldehyde, alcohol
ketone, organic acid, alcohol
224
Multiple Choice
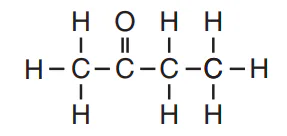
What is the IUPAC name of the compound with the following structural formula?
propanone
propanal
butanone
butanal
225

226
Multiple Choice
The isomers butane and methylpropane have
the same molecular formula and the same properties
the same molecular formula and different properties
different molecular formulas and the same properties
different molecular formulas and different properties
227
Multiple Choice
Which formulas represent compounds that are isomers of each other?
228

229
Multiple Choice

Which type of reaction is represented by the equation below?
esterification
fermentation
saponification
polymerization
230
Multiple Choice
A reaction between an alcohol and an organic acid is classified as
fermentation
esterification
saponification
substitution
231
Multiple Choice
Which reaction results in the production of soap?
esterification
fermentation
polymerization
saponification
232
Multiple Choice
Which type of organic reaction produces both water and carbon dioxide?
addition
combustion
esterification
fermentation
233
Multiple Choice

Which type of reaction is represented by this equation?
fermentation
addition
substitution
polymerization
234
Multiple Choice

This reaction is best described as
addition involving a saturated hydrocarbon
addition involving an unsaturated hydrocarbon
substitution involving a saturated hydrocarbon
substitution involving an unsaturated hydrocarbon
235


Show answer
Auto Play
Slide 1 / 235
SLIDE
Similar Resources on Wayground

219 questions
Advanced Livestock Nutrition
Presentation
•
9th - 12th Grade

217 questions
LECTURA CRÍTICA
Presentation
•
11th Grade

228 questions
DƯỢC LÝ 2
Presentation
•
University

216 questions
CHAPTER 10 : TRANSPORT SYSTEM
Presentation
•
12th Grade

215 questions
Round 3
Presentation
•
KG

231 questions
ASEAN
Presentation
•
9th Grade

230 questions
AVES CUYABENO
Presentation
•
KG - University

226 questions
Heterocyclic chemistry-Part-1 2022
Presentation
•
University
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade