

AY 2526 JC1 Atomic Structure
Presentation
•
Chemistry
•
11th Grade
•
Easy
Yosefina Rahayu
Used 5+ times
FREE Resource
3 Slides • 20 Questions
1
ATOMIC STRUCTURE
2



3

4
Match
Match the following
behavior of subatomic particles in electric field
proton
electron
neutron
bend towards negative charged plate
bend towards positive charged plate
not deflected
bend towards negative charged plate
bend towards positive charged plate
not deflected
5
Match
Match the following
subatomic particle and its relative charge and relative mass
proton (p)
electron (e)
neutron (n)
charge +1
mass 1
charge -1
mass 1/1836
charge 0
mass 1
charge +1
mass 1
charge -1
mass 1/1836
charge 0
mass 1
6
Multiple Select
describe correct term of atomic number
the answer can be more than one
number of proton in the nucleus of an atom
number of proton and neutron in an atom
proton number
number of proton and electron in an atom
7
Multiple Select
describe correct term of nucleon number
the answer can be more than one
number of proton and electron in an atom
number of proton and neutron in an atom
proton number
mass number
8
Match

Match the following
the atom and its respective proton
nickel
krypton
gallium
barium
potassium
28
36
31
56
19
28
36
31
56
19
9
Match

Match the following
the species and its proton number
calcium ion, Ca2+
chromium ion, Cr3+
bromide ion, Br-
sulfide ion, S2-
copper ion, Cu2+
20
24
35
16
29
20
24
35
16
29
10
Match

Match the following
the species and its neutron number
manganese atom
cobalt(II) ion, Co2+
bromide ion, Br-
germanium atom
calcium atom
30
32
45
41
20
30
32
45
41
20
11
Match

Match the following
the species and its number of electron
calcium ion, Ca2+
bromide ion, Br-
oxide ion, O2-
18
36
10
18
36
10
12
Multiple Choice
An atom has 10 protons, 15 neutrons and 10 electrons what is its mass number.
20
10
35
25
13
Multiple Choice

9
10
18
19
14
Reorder

Using a periodic table (you can use the one in the textbook) order the following elements by mass number, least to greatest.
lithium
manganese
Gallium
yttrium
Radon
15
Match
Match the following:
Electrons
Protons
Neutrons
Nucleus
Energy Level
negatively charged particle
positively charged particle
particles with no charge
center of atom
location of where negative charges exist
negatively charged particle
positively charged particle
particles with no charge
center of atom
location of where negative charges exist
16
Multiple Choice
Which model was created after the discovery of the nucleus?
Dalton's
Rutherford's
Quantum Mechanical
Plum Pudding
17
Multiple Choice
An atom has 9 protons, 10 neutrons, and 9 electrons.
What is the charge on this atom?
-1
0
+1
19
18
18
Multiple Choice
An atom has 8 protons, 8 neutrons, and 10 electrons.
What is the mass of this atom?
8
16
18
-2
19
Multiple Select
which descriptor are correctly applied to term "isotopes"
same atom of the same element
different atoms of the same element
same proton and electron number
same neutron number
different neutron or nucleon number
20
Match
Match each definition with the correct term
An atom that has lost or gained an electron
An atom of an element with a different amount of neutrons
A positively charged atom
A negatively charged atom
Ion
Isotope
Cation
Anion
Ion
Isotope
Cation
Anion
21
Match

Match the following parts of the symbol to the correct term
27
13
3+
14
protons
mass number
atomic number
ion charge
neutrons
13
mass number
atomic number
ion charge
neutrons
13
22
Multiple Choice
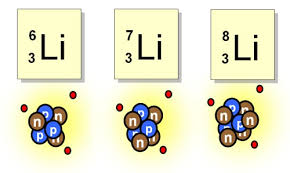
which atom has 5 neutrons?
Li-6
Li-7
Li-8
they have the same # of neutrons
23
Multiple Choice
If X is the symbol for an element, which of the following two symbols represent isotopes of the same element? I. 7735X II. 7835X III. 8137X IV. 8136 X
I and II
III and IV
I and IV
I and III
ATOMIC STRUCTURE
Show answer
Auto Play
Slide 1 / 23
SLIDE
Similar Resources on Wayground

18 questions
Presentasi HIDROKARBON DAN MINYAK BUMI
Presentation
•
11th Grade

21 questions
Intro to mole
Presentation
•
10th Grade

17 questions
Precipitation reactions
Presentation
•
11th Grade

20 questions
Electron Configuration
Presentation
•
10th - 12th Grade

20 questions
Gas Laws
Presentation
•
10th - 12th Grade

19 questions
PPT Asam Basa
Presentation
•
11th Grade

18 questions
BRANCHED ALKANE
Presentation
•
11th Grade

19 questions
Atomic Structure
Presentation
•
10th - 11th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Redox Reactions
Quiz
•
10th - 12th Grade

20 questions
pH and pOH Calculations
Quiz
•
10th - 11th Grade

36 questions
Unit 4 (Part 2) Solutions
Quiz
•
10th - 12th Grade

30 questions
Final Exam Prep S22: 2.1.A.1 Isotopes
Quiz
•
9th - 12th Grade

25 questions
Stoichiometry Mass to Mass
Quiz
•
10th - 11th Grade

15 questions
Calculating pH and pOH
Quiz
•
10th - 12th Grade

15 questions
Balancing Chemical Equations
Quiz
•
10th - 12th Grade