

Density
Presentation
•
Physics
•
7th Grade
•
Medium
Edu Tools
Used 2+ times
FREE Resource
28 Slides • 16 Questions
1
Warming UP
2
Multiple Choice
Kilograms
newtons
pounds
grams
3
Multiple Choice

degree fahrenheit (° F)
degree celsius (° C)
kelvin (K)
degree centigrade (°C)
4
Multiple Choice
What SI unit is used to measure length?
meters
liters
centimeters
kilograms
5
Multiple Choice

The SI unit for time is the __?__.
hours (h)
liter (L)
gram (g)
second (s)
6
Multiple Choice

How many unit cube are there in each solid?
5 unit cubes
6 unit cubes
7 unit cubes
8 unit cubes
7

How to know the pure gold in jewelry?

8
9

PHYSICS – Density
10

LEARNING
OBJECTIVES
1.4 Density
Core
• Recall and use the equation ρ =
m/V
• Describe an experiment to
determine the density of a liquid
and of a regularly shaped solid and
make the necessary calculation
• Describe the determination of
the density of an irregularly
shaped solid by the method of
displacement
• Predict whether an object will
float based on density data
11

SI units
(Le Systeme International d’Unites)
12

SI units
(Le Systeme International d’Unites)
Volume → cubic metre (m3)
1m
1m
1m
13

SI units
(Le Systeme International d’Unites)
Volume → cubic metre (m3)
1m
1m
1m
1 cubic metre = 1m x 1m x 1m
14

SI units
(Le Systeme International d’Unites)
Volume → cubic metre (m3)
1m
1m
1m
1 cubic metre = 1m x 1m x 1m
1 cubic metre (1m3)
= 1000 litres (l)
15
Multiple Choice

410 cm3
420 cm3
402 cm3
401 cm3
16

1 litre bottle = 1l
Same as 1 cubic
decimetre (dm3)
17

1 litre bottle = 1l
Same as 1 cubic
decimetre (dm3)
1 litre = 1000 cubic centimetres (cm3)
= 1000 millilitres (ml)
18

1 litre bottle = 1l
Same as 1 cubic
decimetre (dm3)
1 litre = 1000 cubic centimetres (cm3)
= 1000 millilitres (ml)
1 cubic centimetre (cm3) = 1 millilitre (ml)
1 cm
1 cm
1 cm
19

1 litre bottle = 1l
Same as 1 cubic
decimetre (dm3)
1 litre = 1000 cubic centimetres (cm3)
= 1000 millilitres (ml)
1 cubic centimetre (cm3) = 1 millilitre (ml)
1 cm
1 cm
1 cm
Aluminium
Lead
For any given volume (eg. 1cm3) lead is
heavier than aluminium. It has a higher
DENSITY
20
Poll
See the video, in your opinion if we put Balsa wood and metal pin, what happen to them?
Balsa wood will sink
Metal pin will sink
Metal pin will float
Both of them will sink
21
Poll
See the video! In your opinion, why its happen?
Because they have same density
Because they have different density
22
Video Response
What is density?

23

DENSITY = Mass
Volume
24


DENSITY = Mass
Volume
Water has a
density of
1 g/cm3
1000 cm3of water (1 litre) therefore
has a mass of 1000g (1 kilogram)
25


DENSITY = Mass
Volume
Water has a
density of
1 g/cm3
1000 cm3of water (1 litre) therefore
has a mass of 1000g (1 kilogram)
Substance
Density (g/cm3)
Wood
0.75
Ice
0.92
Water
1.00
Aluminium
2.70
Copper
8.90
Lead
11.4
26



DENSITY = Mass
Volume
Water has a
density of
1 g/cm3
1000 cm3of water (1 litre) therefore
has a mass of 1000g (1 kilogram)
Substance
Density (g/cm3)
Wood
0.75
Ice
0.92
Water
1.00
Aluminium
2.70
Copper
8.90
Lead
11.4
THINK!
A solid object with a density lower
than that of water will float.
So why should a battleship made of
aluminium not sink?
27


Calculating density
In the density equation,
density is shown by the
Greek letter ‘rho’ =
DENSITY = Mass
Volume
= m
V
28


Calculating density
In the density equation,
density is shown by the
Greek letter ‘rho’ =
DENSITY = Mass
Volume
= m
V
m
V
V = m
m = V x
29





Calculating density
m
V
Eg. What is the density of a block of
steel if it has a mass of 9,360g and a
volume of 1,200cm3?
30





Calculating density
m
V
Eg. What is the density of a block of
steel if it has a mass of 9,360g and a
volume of 1,200cm3?
= 9 360
1 200
= 7.8 g/cm3
31
Multiple Choice
density = mass x volume
density = mass / volume
density = mass + volume
density = mass - volume
32
Multiple Choice
Mass and Volume
Temperature and Mass
Grams and Centimeters
Volume and Weight
33
Multiple Choice
3 g/cm3
1/3 g/cm3
27 g/cm3
39 g/cm3
34
Multiple Choice
g
cm3
g/cm3
cm3/g
35





Calculating density
m
V
Eg. What is the density of a block of
steel if it has a mass of 9,360g and a
volume of 1,200cm3?
= 9 360
1 200
= 7.8 g/cm3
Eg. What is the mass of a block of gold if
it has a density of 19.3 g/cm3and a
volume of 540 cm3?
m = 540 x 19.3 = 10,422g
36


Volume and Density
Volume of a liquid
Measuring cylinder
Read off the level on the scale to
get the volume of liquid
37

Volume and Density
Volume of a
regular solid
Length
Height
Width
Volume of rectangular block = length x width x height
38

Volume and Density
Volume of a
regular solid
Length
Height
Width
Volume of rectangular block = length x width x height
Height
Radius
Volume of a cylinder= π x radius2x height
39


Volume and Density
Volume of an
irregular solid
Read off the level on the scale
before adding the irregular solid,
and again after the solid has been
put into the measuring cylinder.
Volume of irregular solid =
Volume after – volume before
Eg. 235 – 155 = 80cm3
40


Volume and Density
Volume of an
irregular solid
Using a Eureka (displacement) can
Fill the Eureka can until it stops
over-flowing.
Gently lower the irregular solid
into the can and measure how
much displaced liquid is collected
in the measuring cylinder.
41


Volume and Density
Volume of an
irregular solid
Using a Eureka (displacement) can
Mass of irregular solid = 350g
Volume of irregular solid = 80cm3
Density = mass / volume
Density = 350 / 80 = 4.375g/cm3
42
Multiple Choice
8 g/cm3
2 g/cm3
1/2 g/cm3
24 g/cm3
43
Multiple Choice
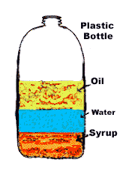
oil
water
syrup
plastic bottle
44
Multiple Choice

10.0 cm3
15.0 cm3
20.0 cm3
25.0 cm3
Warming UP
Show answer
Auto Play
Slide 1 / 44
SLIDE
Similar Resources on Wayground

39 questions
Course 2/3 sol retake
Presentation
•
7th Grade

37 questions
Genetic Engineering
Presentation
•
6th - 7th Grade

40 questions
Pressure
Presentation
•
8th Grade

36 questions
Pressure
Presentation
•
7th Grade

37 questions
Triangles and MAAP Mixed Review
Presentation
•
7th Grade

41 questions
Repaso de ondas
Presentation
•
6th - 8th Grade

38 questions
(Un)Balanced Forces
Presentation
•
8th Grade

37 questions
Scientific Method Notes
Presentation
•
8th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Physics

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade

75 questions
Georgia K-12 ELA Standards – 7th Grade Questions
Quiz
•
7th Grade

14 questions
Volume of rectangular prisms
Quiz
•
7th Grade

17 questions
Georgia Milestones 7th Grade Math Review- Units 1 and 2: Integers, Rational Numbers, Expressions, Equations, Inequalities
Quiz
•
7th Grade

22 questions
Simple Probability
Quiz
•
7th Grade

20 questions
Graphing Inequalities on a Number Line
Quiz
•
6th - 9th Grade

20 questions
genetics vocabulary
Quiz
•
7th Grade