

States of Matter & Phase Changes
Presentation
•
Science
•
12th Grade
•
Easy
+15
Standards-aligned
Sarah Trinidad
Used 3+ times
FREE Resource
19 Slides • 13 Questions
1
States of Matter

2
Main Concepts
Matter is anything that has mass, or takes up space.
All matter is comprised of particles.
Particles are constantly moving.
Every substance can be a solid, liquid or gas, but at different temperatures.

3
State of Matter is a Physical Property.
When the state of matter changes, the actual substance doesn't change, only the form changes.
Water and ice are made up of the same molecules, it just changes form.

4
Each State of Matter is defined by how their particles behave.
How close the molecules are to one another, and how quickly they move determine what state of matter a substance is in.
5
There are 4 states of matter.
solid
liquid
gas
plasma

6
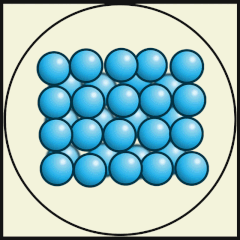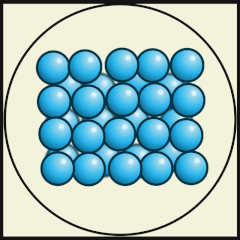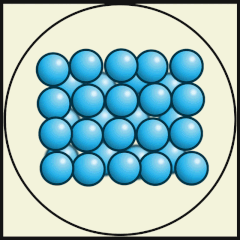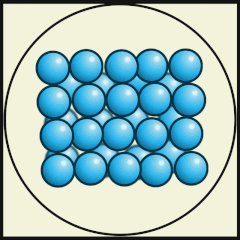
Solid
Particles packed tightly together (dense)
Atoms vibrate in a fixed position.
Have a definite shape, as well as mass and volume
Particles have low kinetic energy (energy of movement)
7
Draw
Draw an example of a solid.
8
Draw
Circle the solid.
9
Liquid
Particles are more loosely packed than a solid
Not easily compressible.
Particles flow around each other
Indefinite shape
Definite Volume

10
Draw
Circle the liquid.
11
Gas
Particles have lots of space between them and high kinetic energy
Easily compressed
Indefinite Shape & Indefinite Volume

12
Draw
Circle the gas.
13
Gas

14
Liquid on the other hand...
In a liquid, the particles are already close together, so it is not easily compressed.

15
Gas
A gas has no definite shape or volume. If unconfined, the particles of a gas will spread out indefinitely. However, if confined, the gas will expand to fill its container.

16
Plasma
Uncommon on Earth (lightning)
most common state of matter in the universe. (Stars)
Highly charged gas particles
High kinetic energy
Think of plasma as gas, with an electrical charge.

17
Multiple Choice
Which state of matter contains particles that flow freely past one another, always touching?
Solid
Liquid
Gas
Plasma
18
Multiple Choice
Which state of matter has a fixed (definite) volume and mass?
Solid
Liquid
Gas
Plasma
19
Multiple Choice
Which state of matter spreads out and completely fills any container it's placed in?
Solid
Liquid
Gas
20
Multiple Choice
Which state of matter contains particles that slowly vibrate, but in a fixed position?
Solid
Liquid
Gas
Plasma
21
Multiple Choice
Which state of matter does not contain easily flowing particles?
Solid
Liquid
Gas
Plasma
22
Multiple Choice
Which state of matter has the ability for its particles to be easily compressed?
Solid
Liquid
Gas
23
Multiple Choice
Which state of matter is like a gas, but with particles that are charged?
Solid
Liquid
Gas
Plasma
24
Multiple Choice
Which state of matter has a definite shape?
Solid
Liquid
Gas
Plasma
25
Reorder
Reorder the following in order from MOST energy to LEAST energy.
26

27

28

29

30

31

32

States of Matter

Show answer
Auto Play
Slide 1 / 32
SLIDE
Similar Resources on Wayground

22 questions
Intro to Oceans Challenge
Presentation
•
12th Grade

25 questions
Measurement Review
Presentation
•
KG

23 questions
Lymphatic system
Presentation
•
12th Grade

25 questions
2 Notes: Post Mortem Stages & Decomposition
Presentation
•
11th Grade

22 questions
Groundwater Notes
Presentation
•
12th Grade

27 questions
Terrestrial Biomes Part Two Lessons
Presentation
•
12th Grade

27 questions
Terrestrial Biome Vocab
Presentation
•
12th Grade

26 questions
Electric Charge (6.1 PS)
Presentation
•
11th - 12th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade

49 questions
AP Environmental Science Final Exam Review
Quiz
•
10th Grade - University

16 questions
Taxonomy and Classification
Quiz
•
6th - 12th Grade

50 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

28 questions
Greenhouse effect
Quiz
•
9th - 12th Grade