

Bonding lesson 1: valence electrons
Presentation
•
Science
•
7th Grade
•
Medium
+11
Standards-aligned
STEPHANIE SLUSS
Used 6+ times
FREE Resource
11 Slides • 14 Questions
1

Mission
❏Define and explain the importance of valence electrons
❏ Learn how groups have similiar properties
2


How do atoms
bond together ?

3
Elements in Group 1 all have 1 valence electron, so they will have similiar properties.

4
Valence Electrons
are electrons that in the outermost energy level. They are important because they determine how atoms will act

5
Now go back to pg 36- we will add valence to your chart
6
Now, we will add valence to your colored table (pg 36)
Group 1 Alkali metals have 1 valence electron. Place a 1 on top of this column on your periodic table


7
Now, we will add valence to your colored table (pg 36)
Group 2 Alkaline Earth metals have 2 valence electron. Place a 2 on top of this column on your periodic table


8
Transition metals are group 3-12 . These have a wide variety of valence numbers and this cannot be known by just looking at the table

9
Now add valence for Groups 13-18. Use the chart below to help you....


10
Now, carefully check your work
Pg 36 Should look like this... check your #'s

11
Now check your notes carefully

12
Multiple Choice

What are electrons in the outermost level of atoms called?
valence electrons
alkali metals
alkaline earth metals
creepy electrons
13
Multiple Choice
4
3
14
16
14
Multiple Choice
7
6
4
1
15
Multiple Choice
What are valence electrons?
The total number of electrons in an atom
The number of electrons in the outermost shell
The number of electrons in the second shell
The number of protons in the outermost shell
16
Multiple Choice
Which elements have the most similar chemical properties?
boron and carbon
oxygen and sulfur
aluminum and bromine
argon and silicon
17
Multiple Choice
Electrons farthest away from the nucleus
Electrons closest to the nucleus
Electrons that just come and go - they don't stay with the atom
Electrons in the second shell
18
Multiple Choice
When trying to identify an unknown element, a scientist determines what other elements reacts with chemically. Which property of the unknown element determines the other element it reacts with?
The total number of neutrons in the unknown element
The total number of particles in the nucleus of the unknown element
The number of protons in the nucleus of the unknown element
The number of valence electrons in the unknown element
19
Multiple Choice
What groups are the most reactive metals?
2 and 3
1 and 18
1 and 2
16 and 17
20
Multiple Choice

2
8
3
13
21
Multiple Choice

2
3
5
22
Multiple Choice
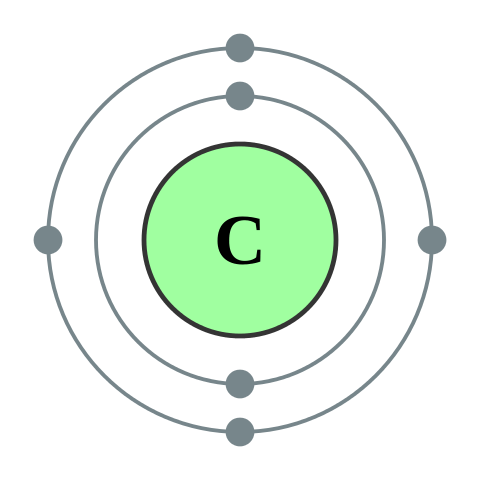
How many valence electrons does carbon have?
4
5
6
7
23
Multiple Choice

12
7
8
14
24
Multiple Choice

7
8
9
10
25
Multiple Choice
Innermost electrons
Middle electrons
Outermost electrons
Any electrons

Mission
❏Define and explain the importance of valence electrons
❏ Learn how groups have similiar properties
Show answer
Auto Play
Slide 1 / 25
SLIDE
Similar Resources on Wayground

21 questions
Plant Levels of Organization
Presentation
•
7th Grade

20 questions
Pemuaian
Presentation
•
7th Grade

20 questions
The Earth's Layers
Presentation
•
7th - 8th Grade

17 questions
Elements, Molecules and Compounds
Presentation
•
7th Grade

21 questions
The Water Cycle
Presentation
•
7th Grade

19 questions
Introduction to The Cell Cycle
Presentation
•
7th Grade

19 questions
Explain: Structure of Atoms
Presentation
•
7th Grade

21 questions
Rocks and The Rock Cycle
Presentation
•
7th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

20 questions
Moon Phases and Eclipses
Quiz
•
6th - 8th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

22 questions
6th & 8th Grade Science Material SOL Review
Quiz
•
6th - 8th Grade

20 questions
genetics, punnett squares, heredity
Quiz
•
7th Grade

17 questions
Human body systems
Quiz
•
7th Grade

20 questions
Food Webs + Energy Pyramids
Quiz
•
7th Grade

20 questions
Evolution and Natural Selection
Quiz
•
7th Grade