

U2L2 - Lecture Notes - How Atoms Differ
Presentation
•
Chemistry
•
12th Grade
•
Easy
Standards-aligned
Bryan Vaughan
Used 23+ times
FREE Resource
7 Slides • 10 Questions
1

Lesson 2
How Atoms Differ
Unit 2
The Structure of the Atom
2

The number of protons and the mass number define
the type of atom.
Essential Questions
• How is the atomic number used to determine
the identity of an atom?
• Why are atomic masses not whole numbers?
• Given the mass number and atomic number,
how are the number of electrons, protons, and
neutrons in an atom calculated?
3

Atomic Number
•Each element contains a unique positive charge in their nucleus.
•The number of protons in the nucleus of an atom identifies the element
and is known as the element’s atomic number.
•Because all atoms are neutral, the number of protons and electrons in an
atom must be equal.
Atomic number = number of protons
= number of electrons
4
Multiple Choice
What is the atomic number?
the number of protons
the number of protons and neutrons
the number of neutrons
the number of atoms
5
Multiple Choice
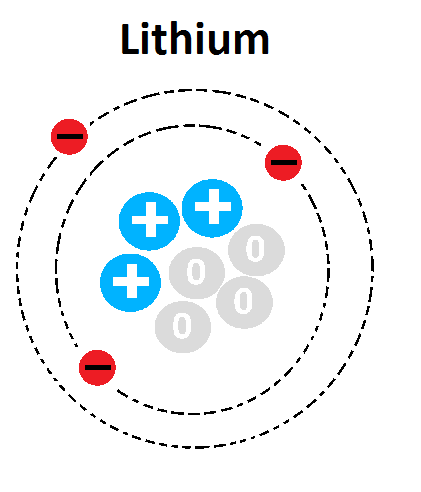
1
3
4
7
6
Multiple Choice

The fluorine atom has an atomic number of
19
9
7

Isotopes and Mass Number
•All atoms of a particular element have the same number of protons and
electrons but the number of neutrons in the nucleus can differ.
•Atoms with the same number of protons but different numbers of neutrons
are called isotopes.
•In nature, most elements are found as mixtures of isotopes. Usually, the
relative abundance of each isotope is constant.
–Example: In a banana, 93.26% is potassium-39, 6.73% is potassium-41 and
0.01% is potassium-40. In another banana or in a different source of
potassium, the percentage composition of the potassium isotopes will still be
the same.
•Isotopes containing more neutrons have a greater mass.
•Isotopes of an atom have the same chemical behavior.
How Atoms Differ
Copyright © McGraw-Hill Education
8
Multiple Choice

A charged atom
An atom with different amounts of neutrons
An atom with different amounts of protons
A neutral atom
9
Multiple Select
Which of the following can vary for atoms of the same element?
number of protons
number of electrons
number of neutrons
10

Isotopes and Mass Number
•The mass number is the sum of the protons and neutrons in the nucleus.
Mass number = atomic number + number of neutrons
11
Multiple Choice
the number of protons
the number of protons and neutrons
the number of neutrons
the number of protons and electrons
12
Multiple Choice
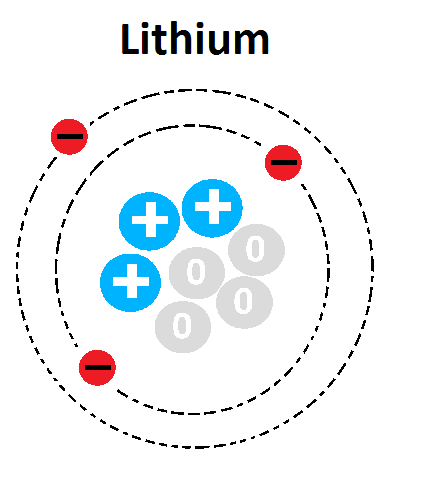
What is the mass number of this atom?
1
3
4
7
13
Multiple Choice

16
15.99
8
Doesn't contain any neutrons
14

Mass of Atoms
•One atomic mass unit (amu) is defined as 1/12th the mass of a carbon-12
atom.
•One amu is nearly, but not exactly, equal to one proton and one neutron.
How Atoms Differ
Copyright © McGraw-Hill Education
15
Multiple Select
Which of the three sub-atomic particles is the lightest?
the proton
the electron
the neutron
16

Mass of Atoms
•The atomic mass of an element is the weighted average mass of the
isotopes of that element.
•Because isotopes have different masses, the weighted average is not a
whole number.
17
Fill in the Blanks

Lesson 2
How Atoms Differ
Unit 2
The Structure of the Atom
Show answer
Auto Play
Slide 1 / 17
SLIDE
Similar Resources on Wayground

12 questions
FFA Emblem
Presentation
•
12th Grade

14 questions
CAPTION TEXT
Presentation
•
12th Grade

12 questions
SECOND CONDITIONAL
Presentation
•
12th Grade

12 questions
Living Things
Presentation
•
KG

11 questions
Algebra - Lesson Negative Exponents
Presentation
•
12th Grade

11 questions
Teenager 5 Session 24: Pronunciation & Speaking
Presentation
•
KG

13 questions
Mixed Conditional
Presentation
•
12th Grade

12 questions
Intro to Thermochemistry
Presentation
•
10th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

15 questions
Calculating pH and pOH
Quiz
•
10th - 12th Grade

20 questions
Le Chatelier's Principle
Quiz
•
9th - 12th Grade

40 questions
AP Chem Review: Units 6-9
Quiz
•
12th Grade

20 questions
Uranium: Twisting the Dragon's Tail
Quiz
•
10th - 12th Grade

13 questions
Nuclear Chemistry
Presentation
•
9th - 12th Grade

5 questions
DOL: Ideal Gas Law
Quiz
•
9th - 12th Grade

20 questions
pH/pOH Calculations
Quiz
•
12th Grade