

Atoms Lesson
Presentation
•
Science
•
KG
•
Medium
Katherine Heter
Used 4+ times
FREE Resource
16 Slides • 27 Questions
1

2

3
Multiple Choice
Which of the following is NOT matter?
coffee
light
oxygen
computer
4

5

6

7
8

9
Multiple Choice
Who was the ancient Greek philosopher that first proposed the idea of atoms?
Aristotle
Democritus
John Dalton
J.J. Thompson
10
Multiple Choice
Who discovered the electron in 1897?
Aristotle
Democritus
John Dalton
J.J. Thompson
11
Multiple Choice
What did J.J. Thompson's model of the atom propose?
Atoms are made up of tiny particles surrounded by empty space
Atoms are uniformly packed spheres of positive matter filled with negatively charged electrons
Atoms consist largely of empty space with a dense, positively charged nucleus
Electrons orbit the nucleus at fixed energies and distances
12
Multiple Choice
What did Ernest Rutherford's gold foil experiment reveal about atoms?
Atoms are made up of tiny particles surrounded by empty space
Atoms are uniformly packed spheres of positive matter filled with negatively charged electrons
Atoms consist largely of empty space with a dense, positively charged nucleus
Electrons orbit the nucleus at fixed energies and distances
13
Multiple Choice
Who expanded on Rutherford's nuclear model and proposed that electrons orbit the nucleus at fixed energies and distances?
Aristotle
Democritus
Niels Bohr
Werner Heisenberg
14
Multiple Choice
According to the text, what did Democritus propose about the nature of atoms?
Atoms are made up of tiny particles surrounded by empty space
Atoms are uniformly packed spheres of positive matter filled with negatively charged electrons
Atoms consist largely of empty space with a dense, positively charged nucleus
Electrons orbit the nucleus at fixed energies and distances
15
16
Multiple Choice
What are the subatomic particles located in the nucleus of an atom?
Protons and electrons
Protons and neutrons
Neutrons and electrons
Protons, neutrons, and electrons
17
Multiple Choice
Which subatomic particle has a positive electrical charge?
Proton
Neutron
Electron
None of the above
18
Multiple Choice
How many electrons are required for stability in atoms with only one energy level or shell?
1
2
6
8
19
Multiple Choice
Which element is stable because it has 8 electrons in its outermost shell?
Carbon
Helium
Neon
Oxygen
20
Multiple Choice
What is the octet rule?
Atoms with fewer than 8 electrons in their outermost shells are stable
Atoms with 8 electrons in their outermost shells are stable
Atoms with more than 8 electrons in their outermost shells are stable
Atoms with 8 protons in their nucleus are stable
21

22
Multiple Choice

Which letter is pointing to the nucleus of the atom?
A
B
C
D
23

24

25

26
Multiple Choice

Which letter is pointing to an electron in the atom?
A
B
C
D
27

28
Multiple Choice
I am positively charged and located in the nucleus of an atom. What am I?
proton
neutron
electron
photon
29
Multiple Choice
Which of the following DOES NOT correctly match the sub-atomic particle with its charge?
proton; +1
neutron; -1
neutron; 0
electron; -1
30
Multiple Choice
Which of the following DOES NOT correctly pair each subatomic particle with its location?
proton, nucleus
neutron, electron cloud
neutron, nucleus
electron, electron cloud
31
Multiple Choice
Which of the following subatomic particles would be attracted to each other based on their charges?
proton, neutron
electron, neutron
proton, electron
32
Match
Match the following
Proton
Nuetron
Electron
Positive
Neutral
Negative
Positive
Neutral
Negative
33

34

35
Multiple Choice
36
Multiple Choice

What is the Atomic number of this atom?
2
4
6
37
Multiple Choice
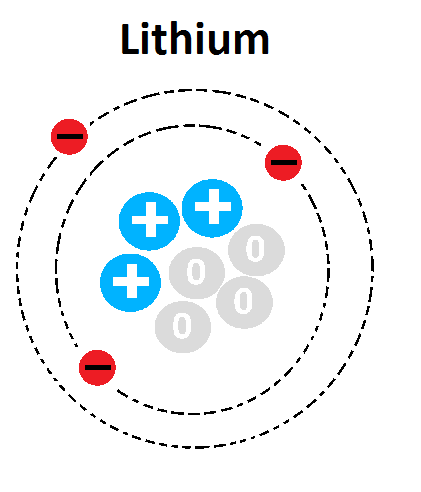
38

39
Multiple Choice
40
Multiple Choice

The fluorine atom has an atomic number of
19
9
41
Multiple Choice
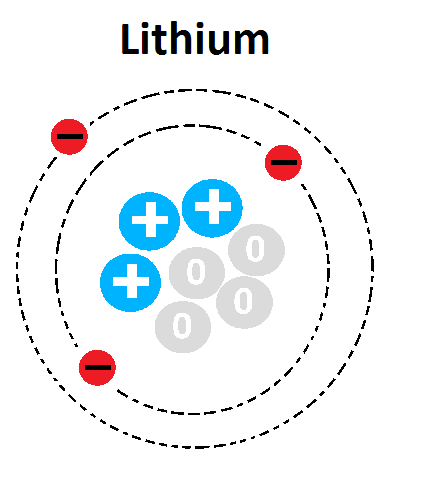
What is the mass number of this atom?
1
3
4
7
42
Multiple Choice

43
Multiple Choice

What is the atomic mass of this atom?
2
4
6

Show answer
Auto Play
Slide 1 / 43
SLIDE
Similar Resources on Wayground

40 questions
Scientific Method
Presentation
•
7th Grade

39 questions
Human Body Systems
Presentation
•
7th Grade

41 questions
Covalent Bonding
Presentation
•
8th - 12th Grade

38 questions
Life Cycle of a Star
Presentation
•
8th Grade

38 questions
Newton's Laws of Motion
Presentation
•
8th - 11th Grade

39 questions
S6E5g
Presentation
•
6th Grade

37 questions
Heating Metal Practice
Presentation
•
7th Grade

37 questions
Types of Plate Boundaries
Presentation
•
10th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade