

EOT G12 Biology Revision Term 1
Presentation
•
Biology
•
12th Grade
•
Medium
Meeta Goklani
Used 3+ times
FREE Resource
0 Slides • 36 Questions
1
Multiple Choice
2
Multiple Choice
3
Multiple Choice
The __________________ is a property of a system in which two points have opposite characteristics, such as charges (positive/negative) or magnetic poles.
polarity
opposites
cohesion
adhesion
4
Multiple Choice
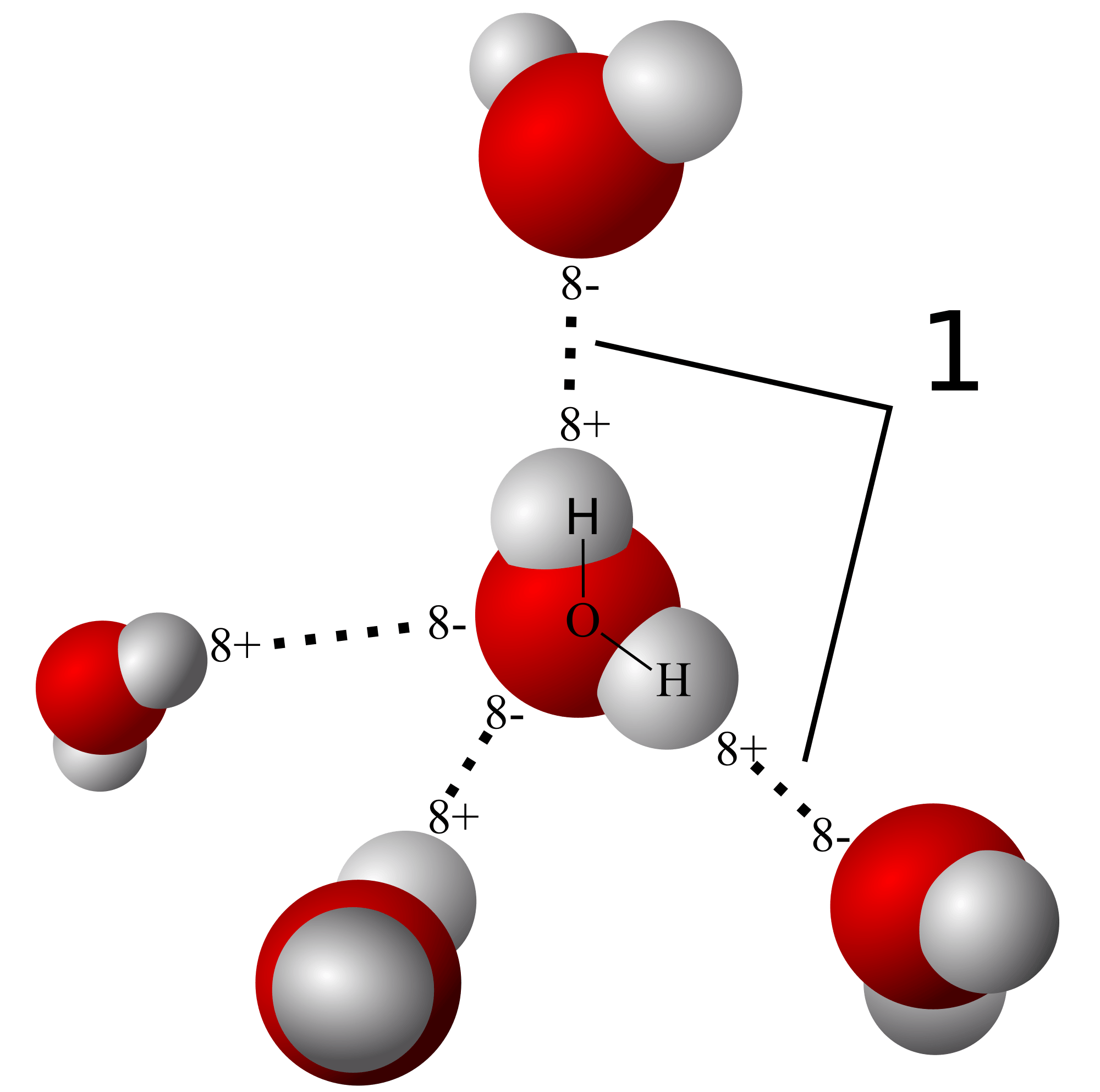
Water molecules are polar molecules. What is meant by the term polar molecule?
a molecule that has no charge on one end and either an overall negative or positive charge on the other end
a molecule that has an overall positive charge on one end, as well as an overall positive charge on the other end
a molecule that has an overall negative charge on one end, as well as an overall negative charge on the other end
a molecule that has an overall positive charge on one end and an overall negative charge on the other end
5
Multiple Choice
A water molecule contains two hydrogen atoms and one oxygen atom. Which of the following statements about water molecules is true?
The oxygen atom is negatively charged and the hydrogen atoms are positively charged.
The oxygen atom is positively charged and the hydrogen atoms are negatively charged.
All of the atoms have a negative charge.
All of the atoms have a positive charge.
6
Multiple Choice

7
Multiple Choice

8
Multiple Choice

9
Multiple Choice
6. Your science teacher showed in class a bowl of fruit and vegetables
salad. Your classmate examined the mixture and was able to
distinguish fruits from vegetables. What type of mixture was presented
by your teacher?
A. colloid
B. heterogeneous (Non-Uniform)
C. homogeneous (Uniform)
D. solution
10
Multiple Choice

Oil and water
Homogeneous Mixture
Heterogeneous Mixture
11
Multiple Choice
Saliva is
Homogenous
mixture
Heterogeneous
mixture
12
Multiple Choice
Air is a solution of gases so it is
Homogeneous
mixture
Heterogeneous
mixture
13
Multiple Select
Which is a heterogenous mixture
sand and water
drink mix in water
14
Multiple Choice
The pH scale is used to know what?
Your temperature
Know one knows
The colors of acids and bases
The strength of acids and bases
15
Multiple Choice
Acids have a pH of _______________.
below 7
above 7
exactly 7
16
Multiple Choice
Bases have a pH of _______________.
below 7
above 7
exactly 7
17
Multiple Choice
Neutral solutions have a pH of _______________.
below 7
above 7
exactly 7
18
Multiple Choice
What ions are there more of in acidic solutions?
H+ (H3O+)
OH-
19
Multiple Choice
What ions are there more of in basic solutions?
H+ (H3O+)
OH-
20
Multiple Choice
Substance with Hydrogen ion is
acidic solution
Basic solution
21
Multiple Choice
Substance with Hydroxyl ion is
acidic solution
basic solution
22
Multiple Select

which of the following is a physical change?
A
B
C
D
23
Multiple Choice

what is shown in the image?
amino acid
nucleotide
glucose
protein
24
Multiple Choice
In an endothermic reaction, heat is ...
taken in
given out
25
Multiple Choice
26
Multiple Choice
27
Multiple Choice

28
Multiple Choice

29
Multiple Choice
O2(g) + 2 H2 (g) → 2 H2O (g) + Energy
endothermic
exothermic
30
Multiple Choice

What does this image shows?
Chemical reaction
isotopes
elements
polar moleules
31
Multiple Choice
In which of the following are bonds broken and new bonds are formed?
chemical reactions
isotopes
elements
polar molecules
32
Multiple Choice
A chemical reaction is a process by which atoms or groups of atoms in substances are
Reorganized into different substances
Dissolved in other substances
Mixed together with atoms in other substances
Ionized by the loss of protons
33
Multiple Choice

What does this image show?
covalent bond
physical property
Van der waals forces
chemical reaction
34
Multiple Choice
35
Multiple Choice
Energy needed to get a reaction started is called
activation energy.
Power Energy
Nuclear Energy
None
36
Multiple Choice

Look at the figure. Determine what the upward curve represents.
Activation energy
reactants
products
enzymes

Show answer
Auto Play
Slide 1 / 36
MULTIPLE CHOICE
Similar Resources on Wayground

29 questions
water cycle
Presentation
•
12th Grade

32 questions
Applied Bio 27.4
Presentation
•
12th Grade

28 questions
milankovich Lesson
Presentation
•
12th Grade

29 questions
Covalent Bonding and Molecular Compounds
Presentation
•
11th - 12th Grade

28 questions
Насекомые - вредители растений
Presentation
•
KG

30 questions
GDP
Presentation
•
12th Grade

32 questions
Reproduction of Representative Animals
Presentation
•
11th - 12th Grade

29 questions
Lesson: Mutations
Presentation
•
12th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Biology

20 questions
Food Chains and Food Webs
Quiz
•
7th - 12th Grade

50 questions
NC EOC Biology Review part 1
Quiz
•
9th - 12th Grade

15 questions
Evidence of Evolution
Quiz
•
8th - 12th Grade

20 questions
NC EOC Biology Review part 1
Quiz
•
9th - 12th Grade

22 questions
Classification
Quiz
•
9th - 12th Grade

50 questions
2026 Ecology TEST Review
Quiz
•
9th - 12th Grade

57 questions
HBS EOC Review
Quiz
•
9th - 12th Grade

14 questions
NEW Ecology Vocab
Quiz
•
9th - 12th Grade