

Chemistry Semester 1 Review
Presentation
•
Chemistry
•
10th Grade
•
Medium
+6
Standards-aligned
Chika Okolo
Used 7+ times
FREE Resource
22 Slides • 59 Questions
1

2
Multiple Choice
The measure of the pull of gravity.
Weight
Gravity
Volume
Density
3
Multiple Choice
The amount of matter in an object; measured by grams, milligrams, or kilograms.
Matter
Mass
Weight
Volume
4
Multiple Choice

Which cup of water has a bigger volume?
Left Cup
Right Cup
5
Multiple Choice

6
Multiple Choice
Anything that has mass and takes up space.
Mass
Matter
Weight
Volume
7
Multiple Choice
How much space an object takes up.
Mass
Weight
Volume
Matter
8
Multiple Choice
Tool used to measure MASS
Metric Ruler
Triple Beam Balance
Graduated Cylinder
Thermometer
9
Multiple Choice

Find the mass.
550.4 g
559.4 g
559.6 g
550.6 g
10
Multiple Choice

50 mL of water was added to the graduated cylinder. After we drop in the object, the new water level goes up to 60 mL. What is the volume of the blocks? (Hint: new volume- old volume)
8 mL
10 mL
60 mL
5 mL
11

Density, mass and volume
12
Multiple Choice
A crayon has a mass of 10g and volume of 5 ml. What is the density?
30 g/ml
15 g/ml
1 g/ml
2 g/ml
13
Multiple Choice
14
Multiple Choice

15
Multiple Choice

The mass and an unknown property are graphed for five samples of the same solid material.
Density
Energy
Volume
Weight
16


Chemical vs Physical Properties
17
Multiple Choice
18
Multiple Choice
19
Multiple Choice
20
Physical change:
1. Temporary change (e.g. change of state of matter)
2. Same substance
Chemical change
1. Permanent change - must go through chemical change to reverse
2. New substance formed.

Chemical vs Physical Changes
Examples
21
Multiple Choice

22
Multiple Choice

23
Multiple Choice

A balanced particle-diagram equation is shown.
Which statement describes the type of change and the chemical properties of the product and reactants?
The equation represents a chemical change, with the product and reactants having identical chemical properties.
The equation represents a physical change, with the product and reactants having identical chemical properties.
The equation represents a chemical change, with the product and reactants having different chemical properties.
The equation represents a physical change, with the product and reactants having different chemical properties.
24
Multiple Choice

Chemical property
Physical property
25
Multiple Choice

Oxygen combines with substance to form rust
bubbles forming
heat, light, or temperature change
All the above
26
Multiple Choice

Which statement describes a chemical property of cobalt?
can be drawn into a wire.
conducts electricity and heat.
combines with oxygen to form rust.
can be flattened into sheets
27
Multiple Choice

28


29
Multiple Choice
Properties that DO depend on the amount of matter present.
Mass
Hardness
Extensive
Intensive
30
Multiple Choice
Properties that do NOT depend on the amount of a substance.
Mass
Hardness
Extensive
Intensive
31
Multiple Choice
Which of the following is an extensive property?
Color
Mass
Odor
Luster
32
Multiple Choice

Which of these observations describes an extensive property of gold?
Shiny, bright appearance
1,943 degrees Fahrenheit melting temperature.
19.3 g/cm3
100 grams
33
Multiple Choice
Which of the following is an extensive property?
Hardness
Boiling Point
Density
Weight
34
Multiple Choice
Which of the following is an intensive property?
Color
Mass
Volume
Weight
35
Multiple Choice
What kind of physical property is this? Density
intensive
extensive
36
Multiple Choice

Olivia measured mass and another property, X, of three pure samples of the same compound. She recorded her data in the table.
Which of the following best describes property X?
X is an intensive property because it varies directly with the size of the sample.
X is an extensive property because it does not vary with the size of the sample.
X is an intensive property because it does not vary with the size of the sample.
X is an extensive property because it varies directly with the size of the sample.
37
Groups of the Periodic Table
Names of the columns

38
Elements in groups behave similarly.
Many of the important groups have names.
39
Alkali Metals
All have 1 valence electron

40
All VERY reactive!
The get more reactive as you go down the column.

41
Group 2
Alkaline-Earth Metals

42
Alkaline-earth Metals
All have 2 valence electrons

43
Group 17
Halogens

44
Halogens
All have 7 valence electrons

45
Halogens
Also VERY reactive!

46
Group 18
Noble Gases

47
Noble Gases
All have 8 valence electrons
(except for He, it has 2!)

48
Noble Gases
Very UNreactive!

49
Groups 3-12
Transition Metals
Called transition metals because their charges change, depending on what they're bonded to. I.e. they do not have a set # of valence e-.

50
Multiple Choice

51
Multiple Select
Elements in the same group have : (2 correct answers)
Similar chemical properties
Similar names
Same number of outer (valence) electrons)
The same number of protons
52
Multiple Select

Which two groups are the most reactive?
halogens
alkaline earth metals
noble gases
alkali metals
salts
53
Multiple Choice

Which group of elements is the most snooty and doesn't want to play with any of the other groups?
Halogens
Lanthanides
Alkali Earth Metals
Noble Gases
Alkali Metals
54
Multiple Choice
Which group does Bromine belong to?
Alkali Metals
Alkaline-Earth Metals
Halogens
Noble Gases
Transition Metals
55
Multiple Choice
Which group does sodium belong to?
Alkali Metals
Alkaline-Earth Metals
Halogens
Noble Gases
Transition Metals
56
Multiple Choice

Which of the following elements is most likely to have similar properties to those of sodium (Na)?
Magnesium (Mg)
Sulfur (S)
Francium (Fr)
Titanium (Ti)
57
Multiple Choice
Name this group: These metals contain familiar metals such as gold, iron, and copper.
58
Multiple Choice
Which two elements have the most similar chemical properties?
Ca and F
B and Ga
Li and Ag
He and O
59
Multiple Choice
Which of the following will have similar properties?
O, S, Se
O, F, Ne
N, Ne, Na
H, K, P
60
Multiple Choice
Find an element with similar chemical properties to Barium (Ba).
Ca an Ra, because they are in the same group and have similar chemical properties.
Cs and La, because they are in the same period number.
Ca and Y, because they are 90 degree angle and have similar properties.
61

62
Multiple Choice

How many valence electrons are found in atoms of group 1?
7
6
4
1
63
Multiple Choice

USE THE PERIODIC TABLE
How many valence electrons does Phosphorus have?
31
5
15
4
64
Multiple Choice
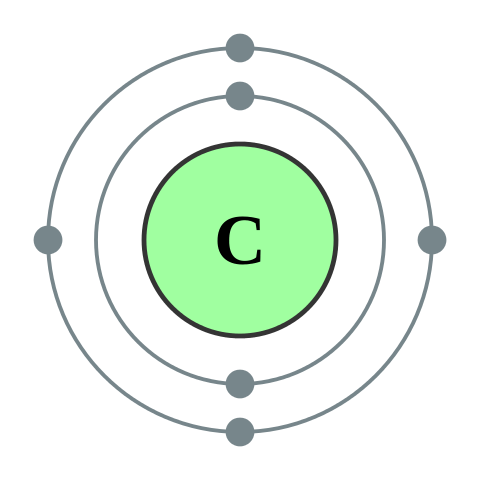
How many valence electrons does carbon have?
4
5
6
7
65
Multiple Choice

How many TOTAL electrons does Phosphorus have?
15
11
20
5
66

67
Multiple Choice
How did Dmitri predict elements?
Time traveled
Predicted the properties by the atomic numbers left
Guess and checked
Calculated the atomic mass and properties
68
Multiple Choice
Dmitri Mendeleev arranged the 63 known elements at the time by __________________.
atomic number
atomic weight
neutron number
none of the above
69
Multiple Choice
What was observed that led to arrange sixty-three already-known elements by increasing atomic weights in the periodic table?
A repetition occurs in chemical and physical properties.
An atomic weight corresponds to the existing abundance.
The number of neutrons and protons increased by one.
The chemical compounds formed by the elements increased by one.
70
Multiple Choice
How are the elements arranged on the periodic table today?
By atomic mass
By number of electrons
By atomic number
By alphabetic order
71
Multiple Choice
In 1869, Mendeleev created a periodic table in which elements were ordered by atomic weight and placed in groups based on their chemical properties. Mendeleev's decision to design the periodic table in this way allowed immediate advances in chemistry by —
providing a framework for models of electron configurations.
discovering the inert gas family, the noble gases
providing an explanation for the reactive properties of the alkali metals.
predicting the physical/chemical properties of yet undiscovered elements
72

Two Types of Substances
1. Element: simplest form
of matter from which
more complex
substances are made
(listed on the Periodic
Table)
– All the atoms that make up
an element are alike
– Ex. Helium
73
Multiple Select
Choose all the items that are elements.
NaCl (table salt)
Sulfur
O2
Water
Air
74
Multiple Choice
Which of the following will NOT break down due to a chemical change?
NaCl
Ni
H2O
FeO2
75

Two Types of Substances
2.
Compound: two or more elements chemically combined
in a fixed proportion
– Can be broken down, but only by chemical reactions
– Have properties different elements that make them up
Ex. H2O
(Water)
Ex. C6H12O6
(Sugar)
Ex. NaCl
(Table Salt)
76
Multiple Choice

Mercury is (Hg)
Element
Compound
77
Multiple Choice

Water is (H20)
Element
Compound
78
Multiple Choice

Oxygen is (O2)
Element
Compound
79
Multiple Choice

Carbon Dioxide (CO2)
Element
Compound
80
Multiple Select
Choose all of the items that are compounds.
NaCl (table salt)
Sulfur
O2
Water
81
Multiple Choice

A suspension is a mixture of a solid and a liquid. The solid can be seen evenly throughout the liquid. After a long period of time, the solid will settle to the bottom of the mixture. Is a suspension a compound?
No it is not, because the solid can only be separated through chemical processes.
Yes it is, because the solid can only be separated through chemical processes.
No it is not, because the solid can be separated through physical processes.
Yes it is, because the solid can be separated through physical processes.

Show answer
Auto Play
Slide 1 / 81
SLIDE
Similar Resources on Wayground

76 questions
Grade 9 General Reading Revision Tri 1
Presentation
•
9th Grade

76 questions
CP Bio : Cell Structure and Function Notes
Presentation
•
10th Grade

75 questions
11.1: Spain's Empire and European Absolutism
Presentation
•
10th Grade

75 questions
OPTICS INTRODUCTION lessons
Presentation
•
10th Grade

74 questions
9.1: The Issue of Slavery
Presentation
•
10th Grade

73 questions
Macromolecules Part 1
Presentation
•
10th Grade

78 questions
Part 2 EOC Review
Presentation
•
10th Grade

79 questions
Classification
Presentation
•
10th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade