

Chemistry Unit 5 Review
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
+6
Standards-aligned
Subha Vidyadharan
Used 10+ times
FREE Resource
9 Slides • 33 Questions
1
Multiple Choice
The specific heat of aluminum is 0.900 J/goC . How much heat is required to
raise the temperature of a 30.0 g block of aluminum from 25.0°C to 75.0°C?
1670 J
1.50 J
0.540 J
1350 J
2
Collision theory-
Rate constant k=Zfp
k = rate constant (how fast the reaction occurs)
Z = collision frequency (how often molecules collide)
f = fraction of collisions with enough energy to react (activation energy requirement)
p = probability that molecules collide in the correct orientation
Collide with each other
Collide with enough energy
Collide in the correct orientation
For Reactions to happen, particles must
3
Multiple Choice
What is the rate of reaction?
The time it takes for a reaction to occur.
How much energy is needed for a reaction to occur.
The energy required to break a bond.
Collision Theory
4

5
Multiple Choice
A 10.0 L sealed container holds 3.00 moles of H2, 4.00 moles of He, 3.00 moles of CO2
The total pressure of the gases in the container is 2.50 atm
Find: partial pressure of CO₂.
10 atm
0.30 atm
0.75 atm
3.0 atm
6
Multiple Choice
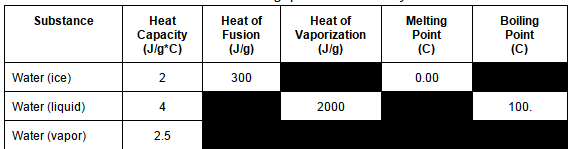
How many Joules of energy are required to change 10 gram of ice at -2 C to water at 20 C?
440 J
880 J
3840 J
66,000 J
7
Multiple Choice

8
Multiple Choice

9
Multiple Choice
PV=nRT
10
Multiple Choice

11
Multiple Choice
A gas tank carrying 55L of Cl2 has a pressure of 2.6atm and a temperature of 289K. How many moles of chlorine gas are in the tank?
6.03 mol
0.55 mol
1.45 mol
0.75 mol
12
Multiple Choice

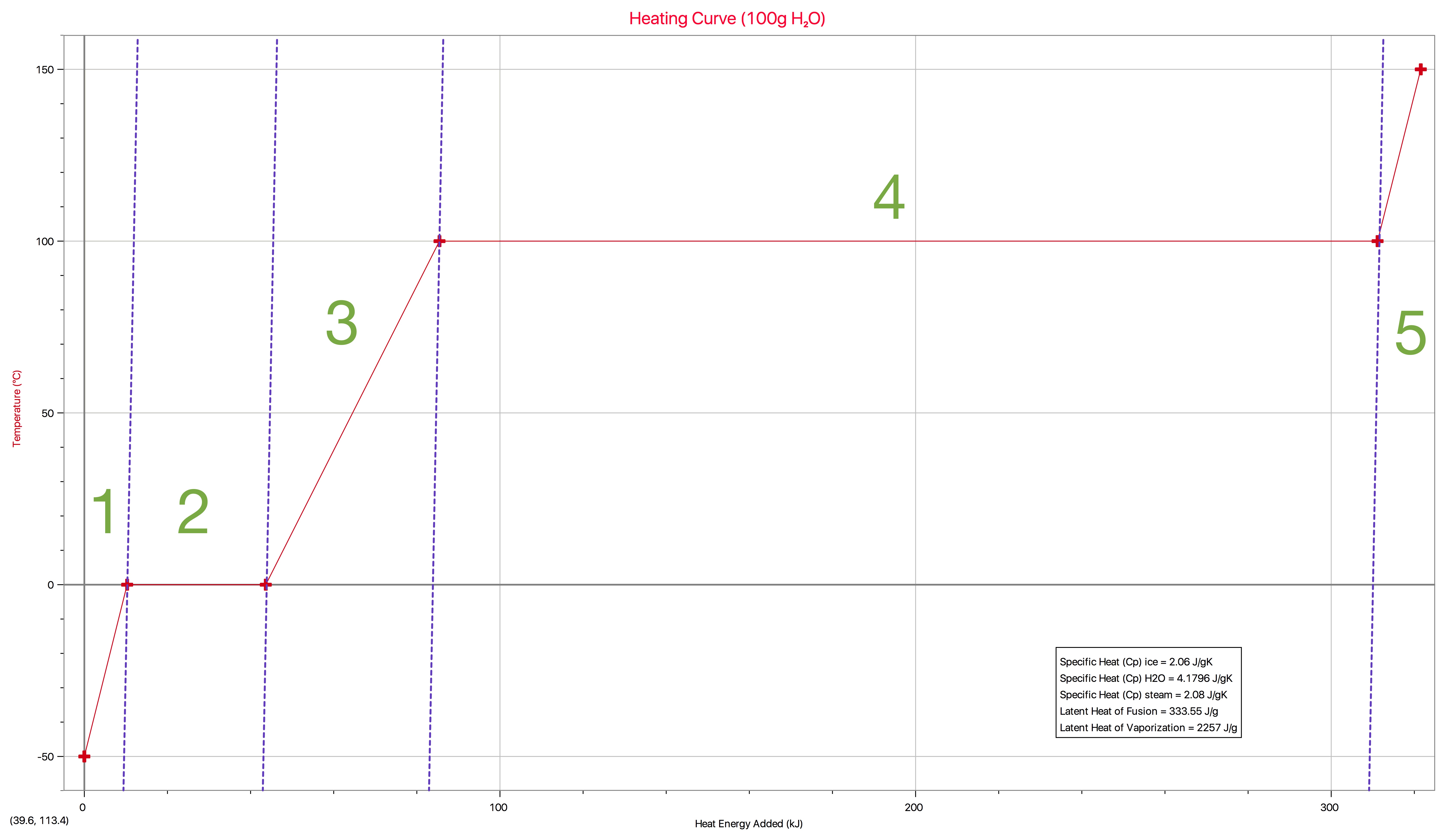
The phase change identified in the image as #2 is called _____ and is _____.
evaporating, endothermic
condensing, exothermic
melting, endothermic
melting, exothermic
13
Multiple Choice

14
Multiple Choice
15
Multiple Choice

16
Some text here about the topic of discussion

17
Multiple Choice

Which container will have lower pressure?
First container
second container
18

19
Multiple Choice
Charles' Law States...
As Volume goes up temperature goes up
As Pressure goes down volume goes down
As Pressure goes up volume goes down
As Pressure goes up temperature goes up
20

21
Multiple Choice
The Ideal Gas Law is understood as: PV=nRT. How does the Ideal Gas Law explain how a hot air balloon rises in the sky?
P is a constant. So as T increases as the air is heated at constant n, volume of the balloon increases causing it to rise.
P and V are kept constant, so as T increases, n must decrease causing the balloon to rise.
V is constant, so as T increases at a constant n, pressure increases causing the balloon to rise.
None of these
22
Multiple Choice
How many moles of helium are contained in a 5.00L canister at 101kPa and 30.0oC?
2.02 moles
0.201 moles
0.494 moles
4.99 moles
23
Multiple Choice
If I have 5.6 liters of gas in a piston at a pressure of 1.5 atm and compress the gas until its volume is 4.8 L, what will the new pressure inside the piston be?
6.3 ATM
2.4 ATM
1.8 ATM
8.6 ATM
24
Multiple Choice
Removing O2(g) will
25
Multiple Choice

26
Multiple Choice
During equilibrium, the amount of the products and the amount of the reactants are __________.
equal
constant (unchanging)
constantly increasing
constantly decreasing
27
Multiple Choice
Use the following reaction to answer the following questions.
Reaction: N2(g) + 3H2(g) ⇌ 2NH3(g)
What will be the result if the volume of the reaction vessel is decreased (aka increase in pressure) for the reaction above?
The equilibrium shifts to the left.
The equilibrium shifts to the right.
The equilibrium does not change.
The equilibrium constant changes.
28
Multiple Choice
CO(g) + H2O(g) ⇌ CO2(g) + H2(g)
This is the expression for the equilibrium constant for the reaction shown above.
29
Multiple Choice
What is the energy required to get the reaction started?
Inhibitor Energy
Catalyst Energy
Activation Energy
Surface Energy
30

31

32
Multiple Choice
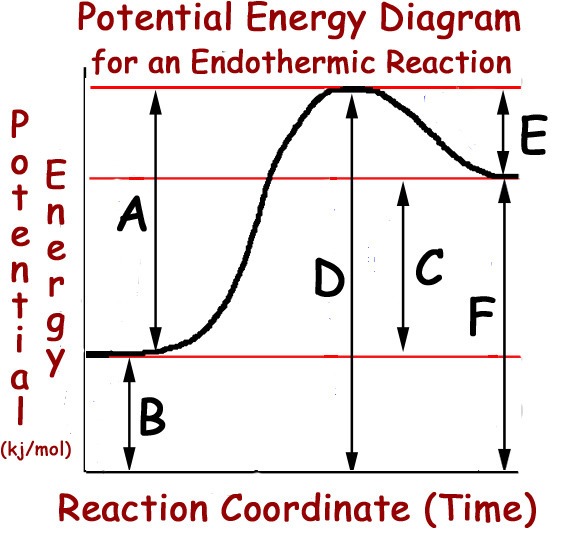
A
B
C
D
33

34

35
Multiple Choice
What factors effect rate of reaction?
Temperature, Concentration, Pressure and Energy
Surface Area, Concentration, Energy and Pressure
Temperature, Pressure, Concentration and Surface area
Pressure, Surface area, Density and Energy
36
Multiple Choice
Slow collision frequency
Allow less effective collision between the particles
Cause particles to lose speed
Increase collision between the particles thus increasing the rate.
37
Multiple Choice
it increases the amount of reactants
it lowers the activation energy
it increases the energy of particle collisions
it increases the frequency of particle collisions because there are more collision sites
38
Multiple Choice
How does a catalyst work in speeding up a reaction?
By lowering the activation energy or reaction
by giving them more energy
by making them more available
39
Multiple Choice

40
Multiple Choice
41
Multiple Choice
42
Multiple Choice
N2 (g) + 3 H2 (g) <=> 2 NH3 (g)
If the pressure in the system is increased, which substance(s) will increase in concentration?
The specific heat of aluminum is 0.900 J/goC . How much heat is required to
raise the temperature of a 30.0 g block of aluminum from 25.0°C to 75.0°C?
1670 J
1.50 J
0.540 J
1350 J
Show answer
Auto Play
Slide 1 / 42
MULTIPLE CHOICE
Similar Resources on Wayground

38 questions
RNA and Protein Synthesis Lesson
Presentation
•
9th - 12th Grade

36 questions
SAVOIR vs Connaitre
Presentation
•
9th - 12th Grade

38 questions
Classification terms part 1
Presentation
•
9th - 12th Grade

36 questions
Introduction to Chemical Reactions (Spring 2023)
Presentation
•
9th - 12th Grade

35 questions
Balancing Chemical Equations
Presentation
•
9th - 12th Grade

35 questions
Chemistry Review
Presentation
•
9th - 12th Grade

37 questions
East Asia History & Culture
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Chemistry

25 questions
Waygrounds 5/20 PS.8.1.2
Quiz
•
9th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

15 questions
Types of chemical reactions
Quiz
•
9th - 12th Grade

32 questions
Acids/Bases, Electrochem, Orgo
Quiz
•
9th - 12th Grade