

Density Review
Presentation
•
Science
•
6th Grade
•
Hard
+5
Standards-aligned
Simone Bedoya
Used 1+ times
FREE Resource
10 Slides • 43 Questions
1

2
Multiple Choice

3
Multiple Choice
The density of an object is equal to
Mass plus volume
volume divided by mass
volume times mass
Mass divided by volume
4
Multiple Choice
Select an image with the least amount of density
5
Multiple Choice
6
Multiple Choice

7
Multiple Choice

Which liquid is less dense than dish detergent?
DIsh detergent
Shampoo
Antifreeze
Maple syrup
8
Multiple Choice
Warm air is _____ dense than cool air.
more
less
the same
9

10

11

12

13

14
Multiple Choice
When the density of a substance is lower than the density of water the substance the substance does what in water?
sink
float
15
Multiple Choice
16
Multiple Choice
If a volcanic rock floats in water, what can you conclude about the rock?
The rock's density is more than the density of water.
The rock has a density less than water's density.
The rock's atoms and molecules are tightly packed together.
The rock's atoms and molecules are buoyant.
17

18

19
Multiple Choice

20
Multiple Choice
21
Multiple Choice

What is the mass shown in the picture?
202002 g
222 g
202 g
200 g
22

23

When the object has regular sides, you need to measure the sides and use the formula
V= L x W x H
Regular Objects
When the object does not have sides that you can easily measure, you need to use the water displacement method.
Irregular Object

24
Multiple Choice
25
Multiple Choice

26
Multiple Choice

27
Multiple Choice

What is the volume of the regular object?
10 cm3
15 cm3
20 cm3
40 cm3
28
Multiple Choice

18.0 ml
6.0 ml
38.0 ml
29
Multiple Choice
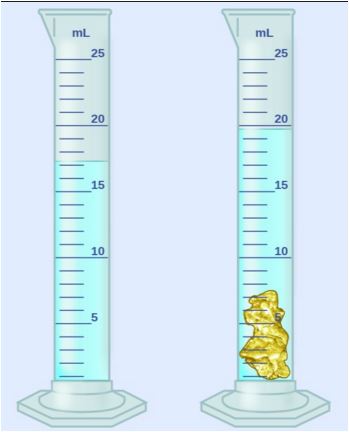
30
Multiple Choice
An object is is placed into 45 mL of water. The water rises to 48 mL. What is the volume of the object?
3 mL
93mL
8 mL
83 mL
31
Multiple Select
Students were asked to calculate the density of a cube. Which measurements do the students need to collect in order to calculate the density of the cube?
Select Two answers.
distance
mass
force
volume
32
Multiple Choice
A 30kg sample of an unknown material has a volume of 15m3. What is its density in kg/m3?
45
2
450
1.5
33
Multiple Choice
Billy is trying to figure out what type of wood to use for building a model boat. He has a block of wood with a volume of 8.0 cubic centimeters (cm3) and a mass of 4.0 grams (g). What is the density of the block of wood in g/cm3 to the nearest tenth?
0.5
2.0
4.0
32.0
34
Multiple Choice
35
Multiple Choice
An object has a mass of 20 g and a volume of 10 mL. What is the density of the object?
200 g/mL
20 g/mL
2 g/mL
0.2 g/mL
36
Multiple Choice
37
Multiple Choice
38
Multiple Choice
39
Multiple Choice
40
Multiple Choice
41
Multiple Choice
What happens to air molecules when they are cooled?
They spread apart
they become compacted
they stay the same
they move quickly
42
Multiple Choice
What does cooler, denser material do?
float
sink
nothing
43
Multiple Choice
What happens to an object that is denser (more dense) than the liquid it is in?
floats
sinks
44
Multiple Choice
45
Multiple Choice

46
Multiple Choice
47
Multiple Choice

48
Multiple Choice
49
Multiple Choice

50
Multiple Choice
When you put an object in a graduated cylinder of water and measure how much the water level changed what is this process called?
Relative Density
Displacement
Density
51
Multiple Select

Which ones can be units for density?
(Multiple responses)
g/ mL
g
cm3g
cm4
52
Multiple Choice

What is the mass of a 25 cm3 liquid if it has a density of 5 g/cm3?
125 g
5 g
cm35g
0.2 cm3
53
Multiple Choice
Find the density of a substance with a mass of 45 grams and a volume of 9 milliliters.
5 kg/L
5 g/ml
45g
36 g/ml

Show answer
Auto Play
Slide 1 / 53
SLIDE
Similar Resources on Wayground

51 questions
Catastrophic Events
Presentation
•
7th Grade

45 questions
Evidence of Evolution Notes
Presentation
•
7th Grade

49 questions
10-3 PART 1 Lesson
Presentation
•
6th Grade

50 questions
Energy Transformation Lesson
Presentation
•
6th Grade

47 questions
Energy Types Review
Presentation
•
6th Grade

47 questions
Energy & waves
Presentation
•
6th Grade

47 questions
Unit 2 Test: Earth's Structure Reteach
Presentation
•
6th Grade

46 questions
Lesson 2.4 - Microbiome
Presentation
•
6th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

20 questions
Rocks and The Rock Cycle
Quiz
•
6th Grade

20 questions
Moon Phases and Eclipses
Quiz
•
6th - 8th Grade

20 questions
Human Body Systems
Quiz
•
6th Grade

22 questions
6th & 8th Grade Science Material SOL Review
Quiz
•
6th - 8th Grade

15 questions
Punnett Squares
Quiz
•
6th Grade

20 questions
Cells! Cell Theory and Characteristics of Eukaryotes/Prokaryotes
Quiz
•
6th Grade

35 questions
Amplify Weather Patterns Unit Review
Quiz
•
6th Grade