

Final M4 Chapter III (Chemical Bonding)
Presentation
•
Chemistry
•
10th Grade
•
Practice Problem
•
Medium
Lady Alias
Used 4+ times
FREE Resource
46 Slides • 36 Questions
1
Chemical Bonding
2
Learning Objectives
Describe the formation of ions by electron loss/gain in order to obtain the electronic configuration of a noble gas.
3
What are ions?
Ions are formed when atoms gain or lose electrons.
4
Why do atoms form ions???
Question

5


3
6


Characteristics
• Belong to Group 18 of the periodic table.
• Stable and unreactive
• Exist as single atoms
4
7


5
Why are noble gases
unreactive?
8


6
What do you notice about the
valence shell?
2 2,8 2,8,8 2,8,8,8 2,8,8,8,8
9
Open Ended

What do you notice about the valence shell?
10


6
What do you notice about the
valence shell?
2 2,8 2,8,8 2,8,8,8 2,8,8,8,8
Ans: They have a full valence shell
🡪 noble gas configuration
11





7
How about other elements?
2,1
2,5



2,2
2,3
2,4
2,6
2,7
12

7
How about other elements?
Sadly, they do not have a full valence shell...

13
So how can other elements become like noble gases?

14
Multiple Choice
How can other elements become like noble gases?
They gain or lose protons to form ions.
They gain or lose electrons to form ions.
They gain or lose neutrons to form ions.
They gain or lose nucleons to form ions.
15


8
They form ions by gaining or losing electrons
They form ions by gaining or losing electrons
in order to achieve noble gas configuration.
16
Multiple Choice
Can noble gases form ions?
Yes
No
17
Multiple Choice
How do we know if an atom has a noble gas configuration?
They have a charge.
They gain or lose electrons to form an ion.
They are neutral.
They have a full valence electron shell
18


10
19
Ions
An ion is an atom that has gained or lost an electron or electrons.
An atom that loses an electron or electrons will have more protons than electrons, it will become an ion with a positive charge.
An atom that gains an electron or electrons will have more electrons than protons, it will become an ion with a negative charge.
Atoms that have a charge can only gain or lose electrons they cannot gain or lose protons.
20
Multiple Choice
An ion is an atom that has gained or lost _______.
protons
neutrons
electrons
21
Multiple Choice
An atom that gains electrons will have a ___ charge.
negative
positive
22
Multiple Choice
An atom that loses electrons has a ____ charge.
negative
positive
23
Cations and Anions
Cations- pronounced cat-ion- is an ion with a positive (+) charge (it will be an ion that lost electrons). It has more protons than electrons.
Anions- pronounced an-ion- is an ion with a negative (-) charge (it will be an ion that gained electrons). It has more electrons than protons.
24
Multiple Choice
Cations have a positive charge, which means they have _____ electrons.
gained
lost
25
Multiple Choice
An ion that has negative charge has gained electrons, it is called a________.
anion
cation
26
Valence Electrons and Ions
Atoms gain or lose electrons from the highest energy level.
The atoms in the highest energy level are called valence electrons.
Elements in group 1 like Lithium [Li] have 1 valence electron.
Elements in group 2 like Berylium [Be] have 2 valence electrons.
Elements in group 13 like Boron [B] have 3 valence electrons.
Elements in group 14 like Carbon [C] have 4 valence electrons.
Elements in group 15 like Nitrogen [N] have 5 valence electrons.
Elements in group 16 like Oxygen [O] have 6 valence electrons.
Elements in group 17 like Flourine [F] have 7 valence electrons.
27
Multiple Choice
Valence electrons are atoms of the outer most energy level, there is a maximum number of eight.
True
False
28
Multiple Choice
How many valence electrons does an atom of silicon have?
3
4
5
6
29
Multiple Choice
Be, Mg, and Ca have ___ valence electrons.
1
2
3
4
30
Octet Rule
The octet rule determines if an atom will gain or lose electrons.
The outer level of an atom has eight electrons.
An atom will gain or lose electrons to have a full outer energy level.
Atoms of metals tend to lose their valence electrons.
Nonmetals tend to gain or share electrons to achieve a full octet.
31
Multiple Choice
Atoms will gain or lose electrons in order to have a full outer energy level this is called the _____ rule.
valence
cation
octet
anion
32
Will an atom gain or lose an electron?
Atoms in group 1, 2, and 13 lose electrons. Remember the atom want a full outer energy level.
For atoms that lose electrons, they empty the outer most energy level.
For groups 1, 2 and 13 - atoms lose electrons forming a positive ion.
For groups 15, 16 and 17 - atoms gain electrons forming a negative ion.
For group 14- atoms can gain or lose electrons forming a positive or negative ion.
33
Multiple Choice
Group 2 will _____ electrons, forming an ion with a positive charge.
gain
lose
34
Multiple Choice
Group 17 elements _____ electrons, forming a negative charge.
gain
lose
35
Elements that lose electrons form CATION
Atoms that lose electrons have more protons than electrons, the ion that forms has a positive charge and is a cation.
They are part of representative groups 1 , 2 , 13 and 14 in the periodic table.
For example sodium [Na] atom is in group 1, it will lose 1 electron and will have the +1 charge to make it a sodium ion [Na+].
For example magnesium [Mg] atom is in group 2, it will lose 2 electron and will have the +2 charge to make it a magnesium ion [Mg+2].
CONTINUE COPYING THE EXAMPLES IN THE WHITEBOARD
36
Multiple Choice
What is the charge on an ion of sodium (Na)?
+1
+2
+3
+4
37
Multiple Choice
Aluminum forms an ion with +3 charge, what group is it in?
1
2
13
14
38
Elements that gain electrons form ANION
Nonmetals tend to gain or share electrons forming negative ions or anions.
When an atom gains an electron or electrons it forms an ion with more electrons than protons. It forms a negative ion (an anion).
To determine the charge of a negative ion (anion) subtract 8 (to obey with the octet rule)
Example- Flourine atom is in group 17. Group 17 has 7 valence electrons subtract 8 (to obey with the octet rule) so that would be (7-8= -1) . That makes a Flourine ion [F-].
CONTINUE COPYING THE EXAMPLES IN THE WHITEBOARD
39
Multiple Choice
An atom of Br will form an anion. What will the charge of the anion be?
+1
+2
-1
-2
40
Multiple Choice
Phosphorous forms an anion with a charge of -3 what representative group is it part of?
13
14
15
16
41

42
Ionic, covalent and metallic bonds
43

44

45

46

47

48

49
Multiple Choice
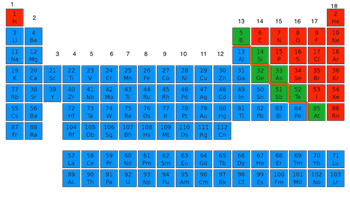
Where are metals located on the periodic table?
Blue
Green
Red
50
Multiple Choice
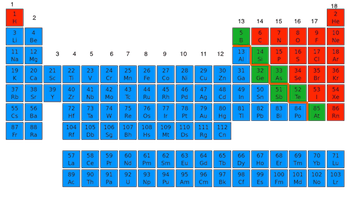
Where are the nonmetals located on the periodic table?
Blue
Red
Green
51

52

53

54

55

56
Multiple Choice
A bond between a metal and a nonmetal is called a __________.
covalent bond
ionic bond
metallic bond
transfer bond
57

58

59

60

61

62

63

64

65

66
Multiple Choice
67

68

69

70
Multiple Choice

NO2
Ionic Bond
Covalent Bond
Both bonds
Neither bond
71
Multiple Choice

MgBr2
Ionic Bond
Covalent Bond
Both bonds
Neither bond
72
Multiple Choice

Predict the bond that will form between Be and F.
Ionic
Covalent
73
Multiple Choice
What type of bond involves the sharing of electrons between atoms?
ionic bond
covalent bond
metallic bond
74
Multiple Choice

Identify the following compound as ionic or covalent: Na2SO4
ionic
covalent
75
Multiple Choice
A bond between a nonmetal and a nonmetal is called a
ionic bond
covalent bond
metallic bond
76
Multiple Choice
A bond between a metal and a nonmetal is called
ionic bond
covalent bond
metallic bond
77
Multiple Choice

Identify the following compound as ionic or covalent: CF4
ionic bond
covalent bond
metallic bond
78
Multiple Choice

Identify the following compound as ionic or covalent: MgO
ionic bond
covalent bond
metallic bond
79
Multiple Choice

Identify the following compound as ionic or covalent: SO2
ionic bond
covalent bond
metallic bond
80
Multiple Choice

Identify the following compound as ionic or covalent: Na2SO4
ionic bond
covalent bond
metallic bond
81
Multiple Choice

H2O, Ionic or Covalent?
ionic bond
covalent bond
metallic bond
82
Multiple Choice

NaBr, Ionic or Covalent?
ionic bond
covalent bond
metallic bond
Chemical Bonding
Show answer
Auto Play
Slide 1 / 82
SLIDE
Similar Resources on Wayground

82 questions
Cell Membrane and Cell Transport Review
Presentation
•
9th - 10th Grade

73 questions
Unit 3 Objectives
Presentation
•
11th - 12th Grade

73 questions
BL Ch 9 Molecular Shape and Bonding
Presentation
•
9th - 12th Grade

74 questions
Chemistry 1st Quarter Review: Eve
Presentation
•
9th - 12th Grade

74 questions
Starting electrolysis
Presentation
•
9th Grade

73 questions
kelas tambahan
Presentation
•
10th Grade

73 questions
Periodic Trends; Atomic Radius
Presentation
•
11th Grade

77 questions
Introduction to Biochemistry Lesson
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade