

M3 periodic table review
Presentation
•
Science
•
9th - 12th Grade
•
Easy
+8
Standards-aligned
William Greene
Used 3+ times
FREE Resource
13 Slides • 23 Questions
1


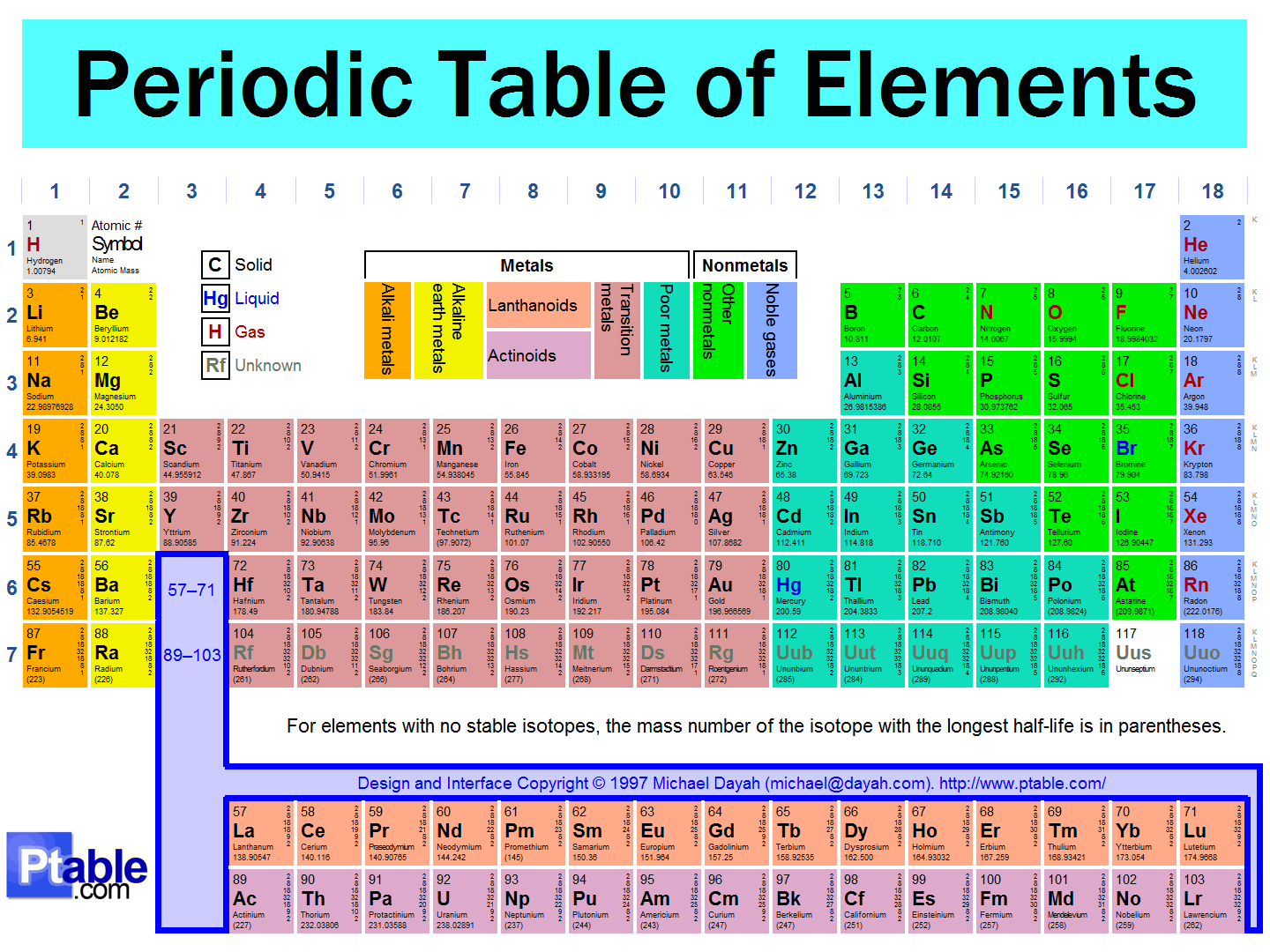
Atoms and the Periodic Table
2

u Chemists used the properties of elements to sort
the elements into groups.
u Mendeleev used increasing atomic mass to construct his
table.
u He knew some elements had similar chemical and
physical properties!
u He put each element on a card with its melting point
(MP) density, color, atomic mass and the # of bonds it
would form.
u He was able to predict undiscovered elements and their
properties after he arranged the cards by atomic mass!!
u Modern periodic table has elements arranged in
increasing atomic number (we needed to identify subatomic
particles before we could use atomic #)
3

When Mendeleev arranged his cards of elements
he noticed that the pattern was not quite right,
but when he moved a few cards over and left
spaces the pattern worked. He concluded that
the spaces were “yet undiscovered” elements.
He was able to predict the properties of these
elements!
4

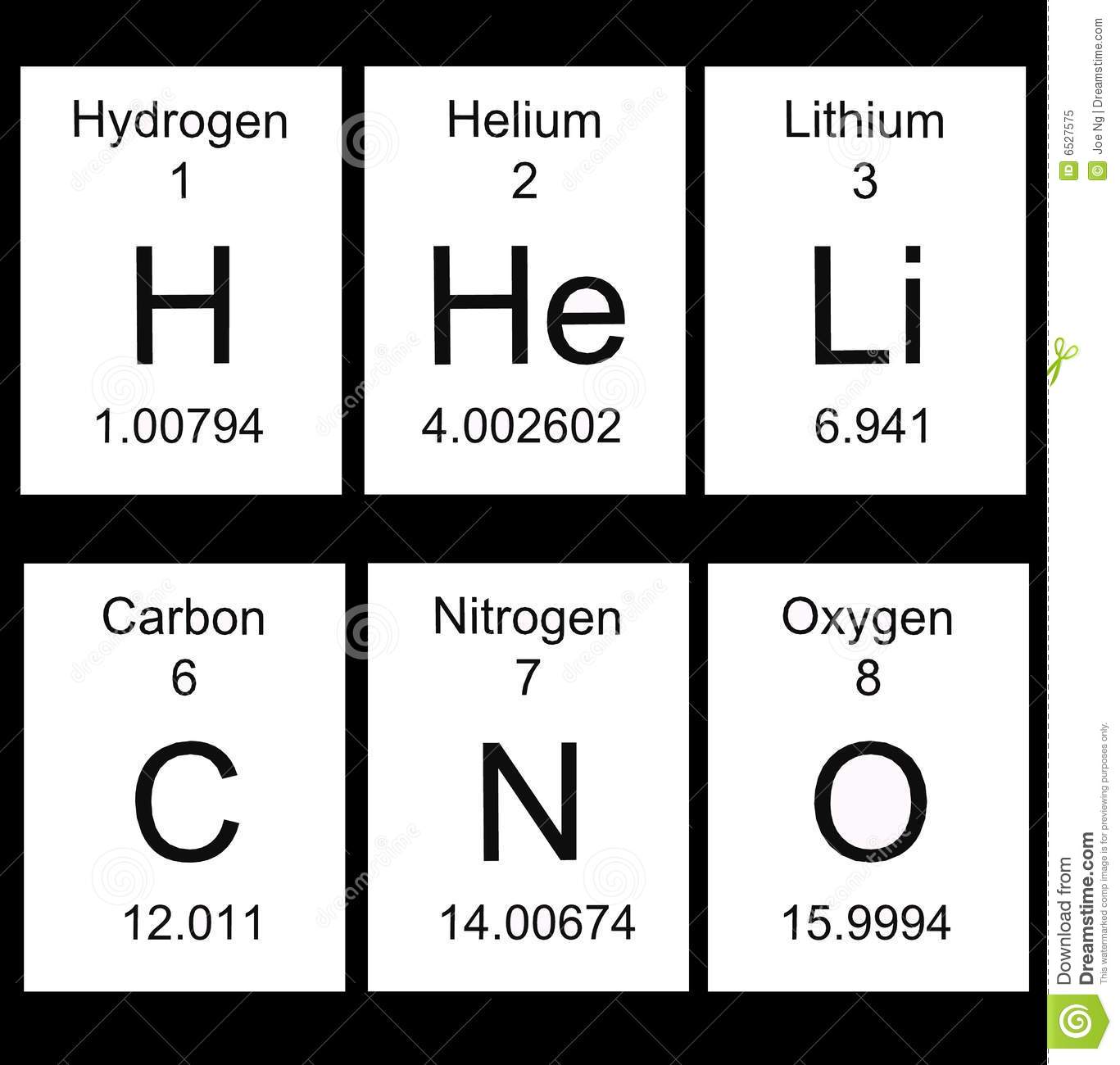
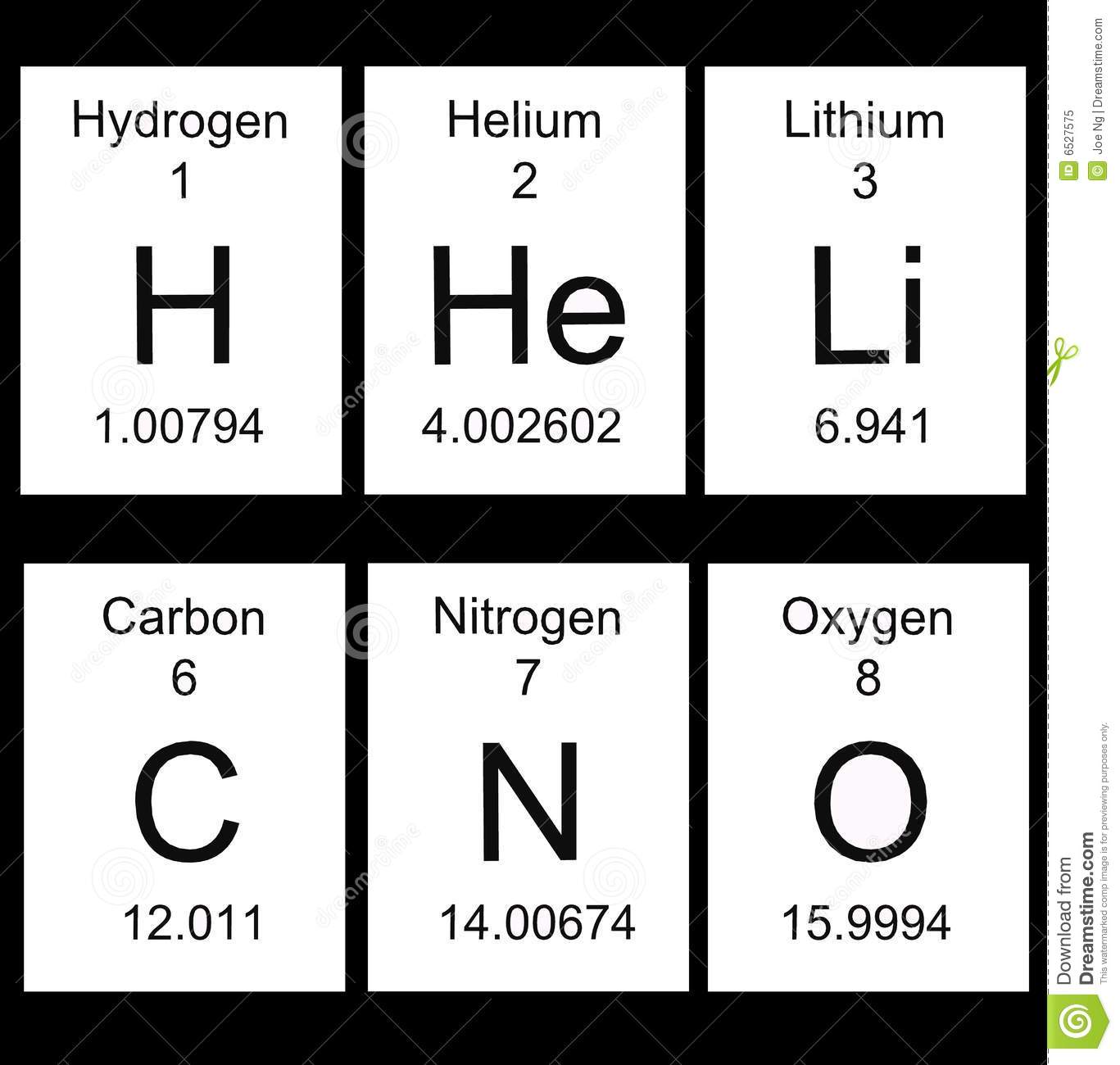
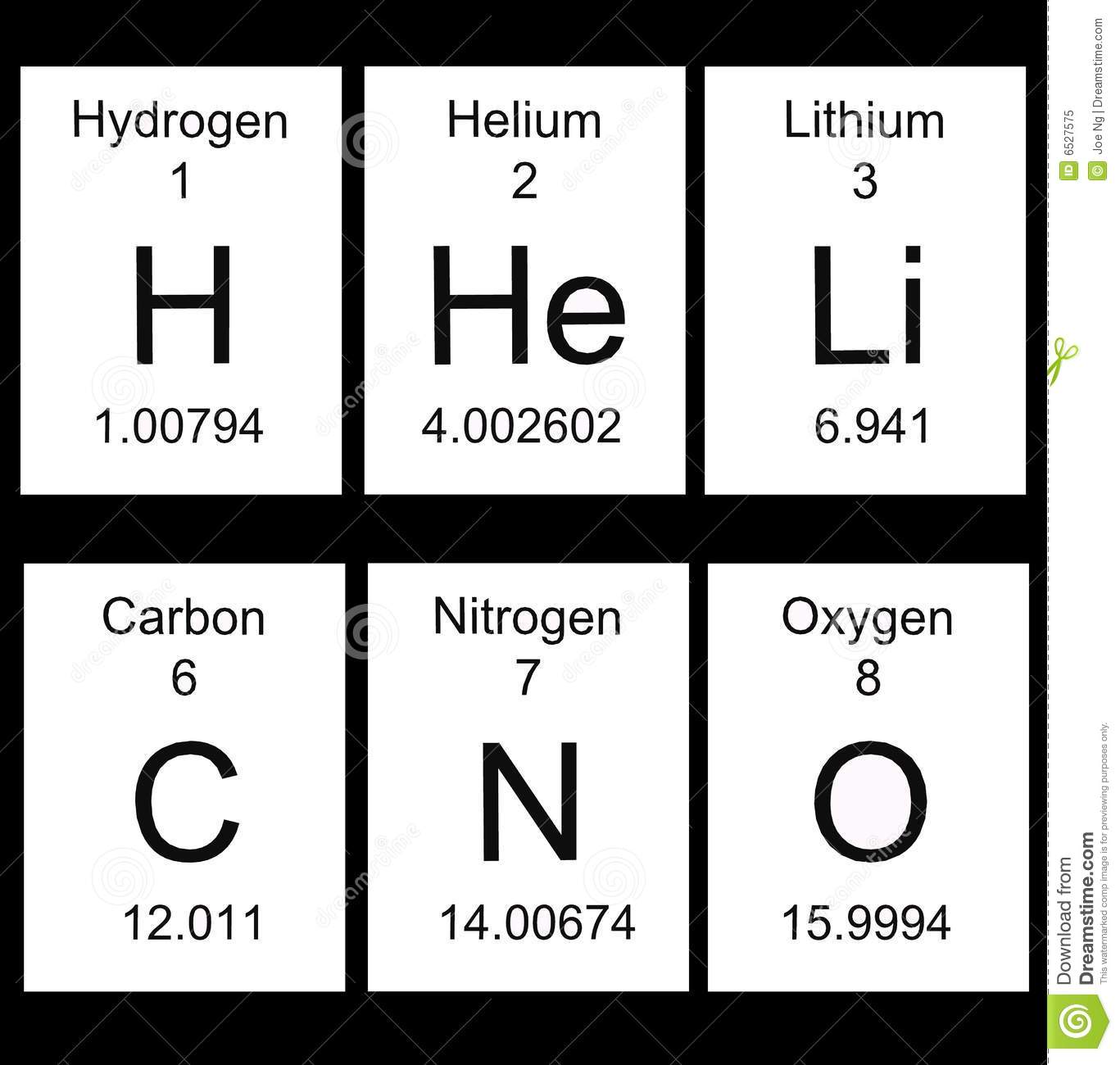
6
C
Carbon
12.011
Atomic number.
Atomic symbol
Element name
Average atomic mass -
Review:
*To find the number of protons, see the atomic number.
*To find the number of electrons (see the atomic number)
*To find the number of neutrons, subtract the atomic number from the
atomic mass. Use whole number. 6 protons
6 neutrons
12 atomic mass
5
Multiple Choice

6
Multiple Choice
7
Multiple Choice
8
Multiple Choice
9

V. How Do The Structures of the Atoms Differ?
A. Atomic Number - # of protons in the nucleus
1. Also tells the number of electrons in a neutral
atom
2. Each atom has a unique atomic number; never
changes
3. It identifies the atom
4. Usually the smallest number, always a whole #
B. Mass Number – sum of the protons + neutrons
1. Usually the biggest number, often a decimal
C. Ion – an atom or group of atoms that has lost or gained 1
or more electrons; therefore has a net (overall) charge
D. Isotope – atoms of the element with different numbers of
neutrons Ex: C-12 and C-14
E. Average Atomic Mass – weighed average mass of a
elements isotopes; why the mass is usually not a whole
number
10

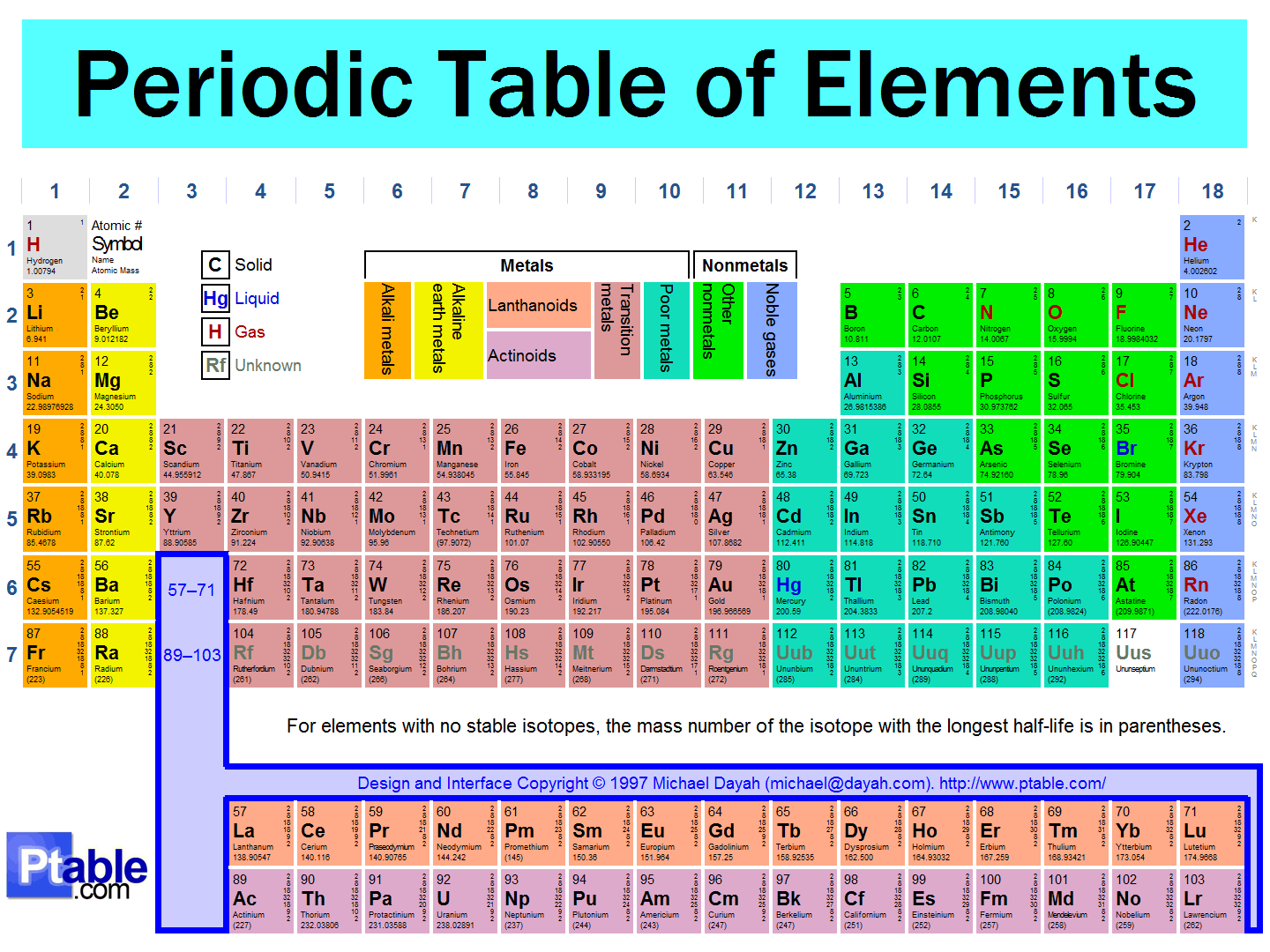
uI: Organization of the Periodic Table
u A. Elements are arranged based upon atomic number
(# of protons). Usually the smallest of the 2 numbers
u Periodic Law – when elements are arranged the way there are
because of similarities in properties and it occurs in a regular
pattern.
u B. Period (series) – horizontal rows
u Number of protons and electrons increase by 1 as you move
across a period.
u C. Group (family) – vertical columns
u Elements in these groups have the same number of valence
electrons and have similar properties
11

Groups
Or
families
Periods
(when we write a sentence, we go ACROSS the paper and put a PERIOD at the end.)
Because the first
shell is full at 2 it
is at the end of
the period,
instead of
in the
second
group with
the other
elements
with 2
valence
electrons
12

Group 1
+1
Looses 1 -e
Group 2
+2
Looses 2 -e
Group
4
Group 5
-3
Gains 3 -e
Group 3
+3
Looses 3 -e
Group
6
Gains 2 -e
Group 7
-1
Gains 1 -e
Group
8
No charge
Transition metals
Variable charges
Less
(electrons) is
more (+) of a
charge
If you gain an
electron e- you
increase your
(-) charge
13

How Are Elements Classified
u Metals are usually shiny
solids that can be
stretched and shaped
They are ductile (put into
thin wire) and malleable(
hammered into sheets).
They are also good
conductors of heat and
electricity.
Most of the elements are
metals.
They are on the left side of
the periodic table
Form (+) ions
u Nonmetals (except H) are
found on the right side of
the chart. They may be
solid, liquid or gas…solid
ones are usually dull,brittle
(shatter if hit with a hamer)
Form (-) ions.
They are poor conductors of
electricity except for C
14


Alkali Metals - Group 1
u Soft, shiny metals
that react violently
with water and other
stuff.
u 1 valence electron in
this group=very
reactive. Will give up
an electron.
u Form 1+ ion
u Ex.: Sodium reacts
quickly with Cl to give
up an electron to it
and become NaCl,
salt.
u Must store in oil
15


Halogens - Group 7
u 7 valence electrons and
have a (-1) charge
u Very reactive; especially
with group 1
u Ex: Bromine (Br) – only
liquid nonmetal at room
temp
u Ex: Fluorine (Fl) – used in
toothpaste
u Ex: Chlorine (Cl) – used in
swimming pools
Diatomic (bond together)
Boiling and melting points
decrease as you move
down.
Go from gas, liquid, solid as you move down.
16


Nobel gases - Group 8
u All gases. Different from most
elements, because they are very
unreactive.
u 8 valence electrons, so they won’t
react with any other elements.
u Also known as inert.
u Ex: Neon (Ne) - used in neon signs
(mixed with other elements gives
other colors).
u Ex: Helium (He) - is very light
(used in balloons).
u Ex: Argon (Ar) - used in light bulbs
to prevent it from burning up
17
Multiple Choice
18
Multiple Choice
19
Multiple Choice

20
Multiple Choice

21
Multiple Choice

22
Multiple Choice

23
Multiple Choice

24
Multiple Choice
25
Multiple Choice
26
Multiple Choice
27
Multiple Choice
28
Multiple Choice
29
Multiple Choice

30
Multiple Choice

31
Multiple Choice

32
Multiple Choice

33
Multiple Choice

34
Multiple Choice
35
Multiple Choice
36

2
He
10
Ne
18
Ar
36
Kr
54
Xe
86
Rn
This group, the Noble gases (group18)
is usually very unreactive. This is
because they all have full valence
electron shells. Helium (He) has 2e in
the 1st shell (holds 2), Neon (Ne) has
2e in the lst shell, and 8e in the 2nd
shell (full).


Atoms and the Periodic Table
Show answer
Auto Play
Slide 1 / 36
SLIDE
Similar Resources on Wayground

28 questions
Reading a Triple Beam Balance
Presentation
•
9th - 12th Grade

32 questions
Waves Part 2
Presentation
•
9th - 11th Grade

25 questions
Solar and Wind Energy
Presentation
•
9th - 12th Grade

26 questions
Properties of Life LESSON
Presentation
•
10th - 12th Grade

29 questions
Parts of the Ear
Presentation
•
10th - 12th Grade

32 questions
SI Units and measurement
Presentation
•
KG

31 questions
Doppler Effect
Presentation
•
9th - 12th Grade

32 questions
Types of Chemical Reactions
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Science

20 questions
Earth Day Trivia
Quiz
•
9th - 12th Grade

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

20 questions
Earth Day
Quiz
•
3rd - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

10 questions
Exploring Natural Selection Concepts
Interactive video
•
6th - 10th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

10 questions
Exploring Air Masses and Weather Fronts
Interactive video
•
6th - 10th Grade