

Y8 U2 - Solubility Practical - Pre-Lab
Presentation
•
Science
•
7th Grade
•
Practice Problem
•
Medium
+15
Standards-aligned
Christopher Terry
Used 10+ times
FREE Resource
15 Slides • 25 Questions
1
Y8 - Unit 2 Solubility
Practical

Pre-Practical Assessment.
2
Poll
What class are you in?
Mr. Aidan
Ms. Lene
Ms. Sahbia
Mr. Rob
3
This Quizizz is a pre-lab assessment!
This is graded.
This Quizizz aims to:
- Inform students about variables.
- Inform students on an example of a solubility experiment.
- Prepare students with hazards, safety and practical method for this practical.
4

The next slide is for students who would like to review "SOLUBILITY".
If you are confident in solubility, you can move to the next slide after the video.
5
6

The next slide is for students who would like to review "VARIABLES".
If you are confident in variables, you can move to the next slide after the video.
7
8
Match
Vocabulary and Definition - Match the following:
Variable
Control Variables
Independent Variable
Dependent Variable
Procedure
Things that can change in an experiment.
What is kept the same
What is changed on purpose.
What is measured for each change as data
The steps or method that is followed.
Things that can change in an experiment.
What is kept the same
What is changed on purpose.
What is measured for each change as data
The steps or method that is followed.
9
Match
Importance of each part of a practical experiment - Match the following:
Control Variables (CV)
Independent Variable (IV)
Dependent Variable (DV)
Procedure
To make it a fair experiment.
Only the change will affect results.
Get data to see if IV had any effect.
To perform the experiment.
To make it a fair experiment.
Only the change will affect results.
Get data to see if IV had any effect.
To perform the experiment.
10
Introduction:
During this practical, you will be given instructions at the start of the lesson on what you should do during this practical and how to fill out the "Lab Report".
Make sure you are aware of any potential safety risk and protective measures, the procedure you should follow and additional information by using this pre-lab assessment.
11
The Practical:
Investigating the effect of water temperature on dissolving sodium chloride.
(Page 55-56 in the Learner's Book)

12
The Practical:
Groups of 4-5;
4 Classrooms will each have a different temperature workstation.

13
The Practical:
4 Classrooms; 4 temperatures:
Each classroom will have a different temperature for you to use to measure the solubility of NaCl (sodium chloride). The teacher will perform a quick run through in terms of procedure for each temperature.
601 (AWI) - Ice Bath - 0-5oC
604 (SSA) - Room Temperature - 20-25oC
605 (RGA) - Heated - 60-90oC (Student's Choice)
606 (SJA) - Boiled - 100oC

14
Equipment - each classroom will have:
Beakers
Thermometers - to measure water temperature.
Spatula - to measure the solute.
Stirring rod - to stir the solution.
Scale/Balance - to measure the salt.
Measuring cylinder (100mL) - to measure volume of water.
Funnel - to fill measuring cylinder.
Heating Element - Kettle, Ice Bath

15
Method / Procedure:
1) Measure 50mL of water (temperature specific) and add to your beaker.
2) Record the mass of your beaker and water.
3) Measure Xg of sodium chloride and add it to your weighing boat.
4) Add the salt and stir continuously.
5) Repeat steps 4-5 until no more salt can be added.
6) Record the mass of your beaker and solution.
7) Calculate how much solute you have dissolved.
8) Repeat for other water temperatures (different classrooms).
9) Generate a table and graph for the results.

16
The Practical: Variables


17
Plot a graph
The independent variable always goes along the horizontal axis.
The dependent variable always goes up the vertical axis.

18
Hotspot
Which speech bubble demonstrates the independent variable?
Click on the speech bubble to give your answer.
19
Hotspot
Which speech bubble demonstrates the dependent variable?
Click on the speech bubble to give your answer.
20
Hotspot
So that only the independent variable will affect the results we obtain, which speech bubble suggests this variable needs be to controlled to ensure the experiment is a fair test?
Click on the speech bubble to give your answer.
21

Complete the following questions as part of your pre-lab preparation and assessment.
22
Multiple Choice
23
Multiple Choice

What apparatus will you use to measure mass?
Spatula
Measuring cylinder
24
Multiple Choice

Which apparatus measures volume?
Spatula
Measuring cylinder
25
Multiple Select

Which step(s) involves the use of spatula?
2
3
4
5
6
26
Open Ended

Step 5 will require which piece of apparatus to mix the solvent and solution?
27
Open Ended

Suggest a hazard that is present within this practical investigation.
28
Open Ended

Suggest a way students can stay safe during this specific experiment; give a reason why.
29
Fill in the Blanks
Type answer...
30
Fill in the Blanks
Type answer...
31
Reorder
Reorder the following:
Record the mass of your beaker.
Measure 50 mL of water; add it to the beaker using a funnel.
Record the mass of your beaker and water.
Measure the amount of NaCl you want to add in a weighing boat.
Add the salt from the weighing boat to the beaker of water and stir.
32
Reorder
Reorder the following:
Add an additional measured amount of salt using the weighing boat and stir until the solution is saturated.
Record the mass of the solution.
Calculate the solute mass dissolved.
Repeat the practical procedure with different solvent temperatures.
Generate a graph using your tabulated data (data from your table).
33
Multiple Choice
How does stirring the solution affect the rate of dissolving salt in water?
Stirring the solution has no effect on the rate of dissolving salt in water.
Stirring the solution decreases the rate of dissolving salt in water.
Stirring the solution causes the salt to evaporate from the water.
Stirring the solution increases the rate of dissolving salt in water.
34
Multiple Choice
Explain why adding more salt to a fixed amount of water may reach a point where no more salt can dissolve.
Adding more salt to a fixed amount of water may reach a point where no more salt can dissolve because the water becomes saturated at its solubility limit.
Adding more salt to water will make it evaporate faster
The salt will disappear if more is added
The water will turn into a solid if more salt is added
35
Multiple Choice
What is the term used to describe a solution that has dissolved all the solute it can hold at a given temperature?
saturated solution
dilute solution
concentrated solution
unsaturated solution
maximized solution
36
Multiple Choice
Describe an experiment you could conduct to determine the solubility of salt in water at different temperatures.
Mixing salt with oil instead of water
Using different types of salt for each temperature
Conduct a series of experiments by preparing saturated salt solutions at different temperatures and recording the temperature at which no more salt can dissolve.
Measuring the solubility of sugar instead of salt
37
Multiple Choice

Use this graph to estimate the solubility of KCl at 70 oC.
about 48 g
about 42 g
about 40 g
50 g
38
Multiple Choice

Use the graph to estimate the temperature at which 120 grams of KNO3 dissolves in 100 g of water.
60 oC
65 oC
70 oC
20 oC
39
Multiple Choice
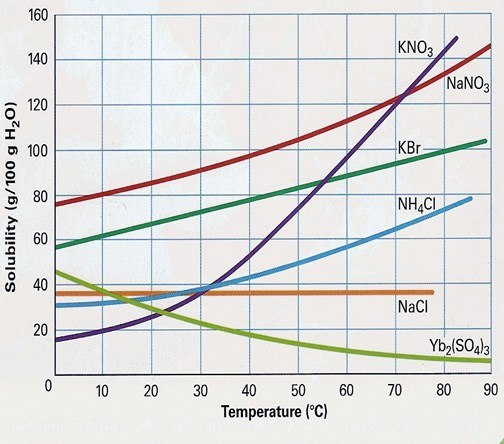
At 80oC, KBr's solubility is:
100g
90g
80g
0g
40
Multiple Choice
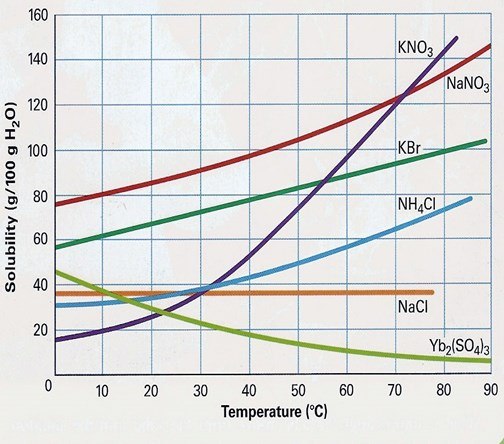
At which temperature do KBr and KNO3 have the same solubility?
60 oC
55 oC
50 oC
Never
Y8 - Unit 2 Solubility
Practical

Pre-Practical Assessment.
Show answer
Auto Play
Slide 1 / 40
SLIDE
Similar Resources on Wayground

37 questions
Heating Metal Practice
Presentation
•
7th Grade

32 questions
Synthetic Materials: Lesson 3 - Materials Science
Presentation
•
7th Grade

35 questions
Electric Current
Presentation
•
7th - 8th Grade

35 questions
Modeling Magnetic Force
Presentation
•
8th Grade

35 questions
Force of Gravity
Presentation
•
6th - 8th Grade

35 questions
Volcanoes and Earthquakes
Presentation
•
6th - 8th Grade

35 questions
Unit 2 Oceans Review 7th Grade Science
Presentation
•
7th Grade

35 questions
Inheritance and Punnett Squares
Presentation
•
7th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

20 questions
Moon Phases and Eclipses
Quiz
•
6th - 8th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

22 questions
6th & 8th Grade Science Material SOL Review
Quiz
•
6th - 8th Grade

20 questions
genetics, punnett squares, heredity
Quiz
•
7th Grade

17 questions
Human body systems
Quiz
•
7th Grade

20 questions
Food Webs + Energy Pyramids
Quiz
•
7th Grade

20 questions
Evolution and Natural Selection
Quiz
•
7th Grade