

A&P Chapter 2: The Chemistry of Life
Presentation
•
Biology
•
11th Grade
•
Medium
Joseph Beshara
Used 3+ times
FREE Resource
49 Slides • 66 Questions
1

Chapter 2
Chemistry of Life
Copyright © 2020 by Elsevier Inc. All rights reserved.
2

• Matter -Anything that occupies space and has mass
• Atoms- basic units of matter
• Element -Pure substance. Made of only 1 type of atom.
Example: A bar of iron (Fe) is made of only iron atoms
•
• Molecules - particle of matter made of more than 1 type of atom
EXAMPLE Water (H2O) is made of hydrogen atoms and oxygen atoms.
Chemical Level of Organization
2
Copyright © 2020 by Elsevier Inc. All rights reserved.
3

⬤ Nucleus: The center of the atom
� Proton: Has a positive charge. In nucleus.
� Neutron: Has a neutral (0) charge. In nucleus
� Atomic number: Number of
protons in the nucleus
Atomic number tells you the name of the element
� Atomic mass: Number of
protons plus neutrons
Also called the MASS number
MASS NUMBER = # of protons + # of neutrons
Structure of an Atom: The
Copyright © 2020 by Elsevier Inc. All rights reserved.
3
4
Multiple Choice
Anything that takes up space and has mass is called...
atoms
matter
element
molecule
5
Multiple Choice
CHOOSE THE BEST ANSWER: Proteins are made of many Nitrogen, Oxygen, Carbon, and Hydrogen Atoms. This is an example of an.....
Matter
Atoms
Element
Molecule
6
Multiple Choice
CHOOSE THE BEST ANSWER: An aluminum can is made of only aluminum atoms. This is an example of a/an
Matter
Atoms
Element
Molecule
7
Multiple Choice
The center of an atom is called the________________
Energy levels
electrons
nucleus
atomic number
8
Multiple Select
Pick all that apply: In the nucleus _______________ can be found
Protons
electrons
neutrons
energy levels
9
Multiple Select
Pick all that apply: The atomic number tells you what 2 things?
Name of the element
How many protons
How many neutrons
energy levels
10
Multiple Choice
Which element has an atomic number 17?
Carbon
oxygen
chlorine
fluorine
11
Multiple Choice
Antimony (Sb) has an atomic number of _____
121.76
51
118.81
50
12
Multiple Choice
An atom has a mass number of 20 and 10 protons. How many neutrons does this element have?
20
10
15
30
13
Multiple Choice
An atom has a mass number of 16 and 8 neutrons. What is the name of the element?
chlorine
Chromium
oxygen
nitrogen
14
Multiple Choice
An atom as an atomic number of 12. This element also has 12 neutrons. What is its mass number? (HINT: What does atomic number tell you?)
12
6
24
48
15
Multiple Choice
How many protons does Gold (Au) have?
47
74
79
17
16
Fill in the Blanks
17
Fill in the Blanks
18


Copyright © 2020 by Elsevier Inc. All rights reserved.
4
19


⬤
Energy levels: Orbits around the nucleus. Contains Electrons
� Electron: has a negative charge
� Each energy level may contain a different number of electrons
� Number of electrons in outer energy level determines chemical
behavior
Energy Levels
6
20


Model of an Atom
Copyright © 2020 by Elsevier Inc. All rights reserved.
7
21
Match
Match each type of charge with the subatomic particle
Negative charge
positive charge
no charge or neutral
electrons
protons
neutron
electrons
protons
neutron
22
Multiple Choice

23
Multiple Choice

24

⬤ Molecule: Larger chemical unit of atoms
⬤ Element: Pure substance;
composed of only one element.
Sometimes can be a molecular element
The "gens": Oxygen (O2) Hydrogen (H2), Nitrogen (N2) Halogen (X2)
⬤ Compound:
2 or more different elements
⬤ Chemical formula determines atoms present in a
compound
Elements, Molecules, and
Compounds
Copyright © 2020 by Elsevier Inc. All rights reserved.
8
25
Categorize
H2
A bar of Pure Gold
Chlorine (a halogen)
Table Salt (NaCl)
Table sugar (C12H24O11)
Hydrochloric Acid (HCl)
Sort between an element or a compound
26

⬤ Isotope: Same element with a different number of neutrons
⬤ Radioactive isotope: Isotope that emits radiation
� Sometimes used in nuclear medicine to evaluate the
function of body parts
⬤ Exposure to high radiation levels may cause cancer
cells to develop
Radioactive Isotopes
Copyright © 2020 by Elsevier Inc. All rights reserved.
9
27
Multiple Choice
The same element with two different masses is called a/an...
isomer
molecule
isotope
ion
28

Types of Bonds
10
29

⬤ Chemical bonds form to make atoms more stable
� Stability occurs when outermost energy level of each
atom becomes full
⬤ Two types of chemical bonds we will examine:
Ionic bonds
• Between METAL and NON METALS
• electrons are TRANSFERRED
• Ex: Table Salt is Sodium Chloride (NaCl)
Covalent bonds – Covalent means sharing
• Between NON METALS
• Electrons are shared
• EX: Water (H2O) and Carbon Dioxide (CO2)
Chemical Bonding
Copyright © 2020 by Elsevier Inc. All rights reserved.
11
30


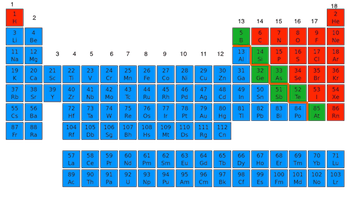
To see what is metal or nonmetal:Find the steps.
Left of the Steps:
METALS
Right of steps
NONMETALS
The steps are
called metalloids
The only
exception is
Hydrogen (it is
non metal)
Metals vs Nonmetals
12
31
Categorize
Chlorine (Cl)
oxygen
Carbon
Hydrogen
Sodium (Na)
Iron (Fe)
Nitrogen (N)
Phosphorus (P)
Sulfur (S)
Argon (Ar)
Copper (Cu)
Gold (Au)
Silver (Ag)
Aluminum (Al)
Lead (Pb)
Zinc (Zn)
Categorize each element as a metal or nonmetal
32
Multiple Choice
A chemical bond between a METAL and a NONMETAL is called a ___________ bond
acidic
covalent
polar
ionic
33
Multiple Choice
A chemical bond between one or more NONMETALs is called a ___________ bond
acidic
covalent
polar
ionic
34

To figure this out split the molecule by elements. Ignore the little
numbers
Example NaCl
Na is a METAL, Cl is a NON METAL
The bond between them is IONIC
Example 2: CO2 (carbon dioxide)
C is a NONMETAL, O is a NONMETAL
The bonds in this are COVALENT
Ionic or Covalent Bonds
13
35

⬤ Ions form when an atom gains or loses electrons in
its outer energy level to become stable
� Cation = The positive part. Always the metal
� Anion = The negative part. One or more nonmetals
⬤ Ionic compounds dissolve easily in water
⬤
Ionic bonds form when oppositely charged ions attract
each other
⬤
Electrolyte: Forms ions when dissolved in water
⬤
The formula of an ion always shows its charge by a “+”
or “−” after the chemical symbol
Ionic Bonds
Copyright © 2020 by Elsevier Inc. All rights reserved.
14
36


Ionic Bonding
Copyright © 2020 by Elsevier Inc. All rights reserved.
15
37

⬤ Covalent bonds
� Form when atoms share their outer energy to fill up
and thus become stable
� Do not ordinarily easily dissociate
in water
⬤ Hydrogen bonds
� Weak forces hold molecules in folded shapes
or in groups
� Do not form new molecules
� Found in bonds between H and F (Fluorine), H and O (oxygen) and H and N (nitrogen)
Covalent Bonds
Copyright © 2020 by Elsevier Inc. All rights reserved.
16
38


Covalent Bonding
Copyright © 2020 by Elsevier Inc. All rights reserved.
17
39

Why are they pretty strong?
Things H-bonds explain.
Hydrogen Bonds
18
40

How they work?
In some covalent molecules, there are partial positive and negative charges
This allows them to interact with similar molecules
Why are they pretty strong?
They form a tight network which gives it strength
In biology, A DNA molecule will have billions of these holding a double strand together
Things H-bonds explain.
The high boiling point of water compared to other similar sized molecules
How certain proteins fold
Hydrogen Bonds
18
41

Types of Bonds
Ionic: Between METALS AND NONMETALS
: Electrons are TRANSFERRED
Covalent: Between NONMETALS
: Electrons are _SHARED
Hydrogen bond: Between H and F, H and O, H and N
: A type of covalent bond
Summary of Bonds
19
42
Multiple Choice
The chemical bond between a non-metal and another non-metal will be a ________ bond.
metal
ionic
covalent
polar
43
Multiple Choice
44
Multiple Choice
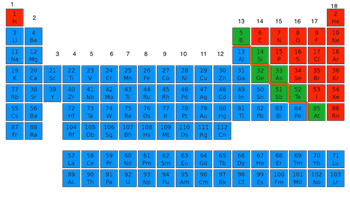
45
Multiple Choice

What type of elements form cations?
metals
nonmetals
metalloids?
46
Multiple Choice
Identify the following compound as ionic or covalent: MgO
ionic
covalent
47
Multiple Choice
Identify the following compound as ionic or covalent: CF4
ionic
covalent
48
Multiple Choice
Identify the following compound as ionic or covalent: SO2
ionic
covalent
49
Multiple Choice
Identify the following compound as ionic or covalent: Ca(OH)2
ionic
covalent
50
Multiple Choice
51
Multiple Choice
What property of water is responsible for its high boiling point?
Covalent bonding
Ionic bonding
Hydrogen bonding
Metallic bonding
52
Multiple Choice
Why do HF, H2O, and NH3 have higher boiling points compared to other molecules?
They have ionic bonds.
They exhibit hydrogen bonding.
They are larger molecules.
They have covalent bonds.
53
Multiple Choice
Which of the following molecules exhibit unusually high boiling points due to hydrogen bonding?
CO2
O2
NH3
Ne
54

Common Substances in Living
Systems
20
55

Organic Chemistry
Organic and Inorganic Chemistry
Copyright © 2020 by Elsevier Inc. All rights reserved.
21
organic molecules:
Carbon-carbon or Carbon Hydrogen bonds
Size:
Larger and more complex than inorganic
Examples
Proteins, fats, nucleic acids, and carbohydrates
Inorganic Chemistry
inorganic molecules:
Water, salts, gases, acid, and bases
Size:
Smaller and simpler than organic
Examples:
Water, Carbon Dioxide, Oxygen, Hydrochloric acid, vitamins.
56
Categorize
Carbon-based molecules
Molecules tend to be large and complex
DNA, Proteins, carbohydrates, and lipids
Salts, water, gases, acids, and bases
Smaller and simpler molecules
Carbon Dioxide, Oxygen, Hydrochloric acid, and vitamins
Classify the following as either organic or inorganic chemistry
57

Water
Carbon Dioxide
Ammonia
Mineral Salts
Common inorganic molecules in
the human body
22
58

⬤ Water
� Water is essential to life
� The most abundant compound in the body, found in
and around each body cell
� Water’s slightly gluelike nature helps hold the body
together
� Water is a solvent (liquid into which solutes are
dissolved), forming aqueous solutions in the body
• Universal solvent
Inorganic Chemistry
Copyright © 2020 by Elsevier Inc. All rights reserved.
23
59

⬤ Acids, bases, and salts
� Water molecules dissociate to form equal amounts of
H+ (hydrogen ion) and OH− (hydroxide ion)
� Acid: Substance that shifts the H+/OH− balance
in favor of H+; opposite of base
� Base: Substance that shifts the H+/OH− balance
against H+; also known as an alkaline; opposite
of acid
Inorganic Chemistry
Copyright © 2020 by Elsevier Inc. All rights reserved.
24
60

⬤ Acids, bases, and salts
� pH: Mathematical expression of relative H+
concentration in an aqueous solution
• pH 7 is neutral (neither acid nor base)
• pH values above 7 are basic; pH values below 7 are
acidic
� Neutralization: Acids and bases mix to form salts
� Buffers: Chemical systems that absorb excess acids
or bases and thus maintain a relatively
stable pH
Inorganic Chemistry
Copyright © 2020 by Elsevier Inc. All rights reserved.
25
61


The pH Scale
Copyright © 2020 by Elsevier Inc. All rights reserved.
26
What is the pH of Blood?
What is the pH of Stomach Acid?
Is ocean water (pH= 8) acidic or basic?
62
Multiple Choice
What is the pH of stomach acid?
0.8
2.8
7.4
11.9
63
Multiple Choice
What is the pH of blood?
0.8
2.8
7.4
11.9
64
Multiple Choice
Acids and bases mix to form
salt
65
Multiple Choice
pH above 7 is considered
neutral
acidic
basic
radioactive
66
Multiple Choice
pH below 7 is considered
neutral
acidic
basic
radioactive
67
Multiple Choice
Ocean water has a pH of 8.0. This would be considered
neutral
acidic
basic
radioactive
68
Multiple Choice
pH exactly 7 is considered
neutral
acidic
basic
radioactive
69
Multiple Choice
The most abundant compound in the body
Sodium Chloride (Salt)
Water
Carbon Dioxide
Ammonia
70

In humans, also known as macromolecules
The most common element in these molecules is
carbon
1. Carbohydrates also known as
sugars
2. Lipids also known as fats
3. Proteins
4. Nucleic acids
Organic Compound
27
71

⬤
Carbohydrates: Complex carbohydrates and sugars
� Contain carbon (C), hydrogen (H), and oxygen (O)
� Function of carbohydrates is to store energy
for later use
Carohydrates
Copyright © 2020 by Elsevier Inc. All rights reserved.
28
72

Carbohydrates
Another word for Carbs is SUGARS
Two Types
1. Simple Carbs-Two Types
a. Monosaccharides
b. Disaccharides
2. Complex Carbs
73


Simple Sugars : Monosaccharides
●
Mono = ONE
●
Saccharide = SUGAR
●
Simple Structure:
○Consists of …
■A 5-6 carbon ring
■Hydrogens or alcohol groups
coming off the ring
� Made up of six-carbon
subunits called
monosaccharides or single
sugars (e.g., glucose)
74


Examples of Monosaccharides
1.
Glucose:
a.
The preferred source of energy for the
body.
b.
The main component in ATP
PRODUCTION
c.CELLULAR ENERGY CURRENCY_= ATP
2.
Fructose:
a.
A Natural Sugar Found in FRUIT
b.
Enters paths that will eventually lead to
ATP production
3.
Galactose:
a.
Gets converted into glucose with the
body
4.
Ribose and Deoxyribose
a.
The R in RNA and the D in
DNA
b.One of the three major components of
nucleic acids
75


Simple Sugars Disacharrides
●
Di = TWO
●
Sucrose = Sugars
○
●
Lactose: Sugar found in Milk
○
●
Maltose: A sugar found in malts
� Disaccharide: Double sugar
made up of two
monosaccharide units (e.g.,
sucrose, lactose)
76


Complex Sugars =Polysaccharides
●3 or more sugars linked
●Found in beans and whole grains
●Cellulose
○Found in plants and vegetables
●Amylose: rices and wheats
●Amylopectin: Starch found in
potatoes and corn
All of these starches break down into
smaller molecules into the body
�Polysaccharide: Complex carbohydrate
made up of many monosaccharide units
(e.g., glycogen made up of many glucose
units)
77

Function of Carbs
●Structurally:
○ Basis for some hormones
○MAIN FUNCTION: Breakdown into glucose
○Glucose creates ATP
○ATP (Adenosine Triphosphate) is cellular energy currency
“Good vs Bad” Carbs
●Food that are least processed and refined, like eating an orange instead of an orange cake
●Natural Carb sources, like fruits are better than processed sugars and carbs.
78


Cellular
Respiration:
How we get our
energy
79


Carbohydrates
Copyright © 2020 by Elsevier Inc. All rights reserved.
36
80
Multiple Select
What are the two types of carbohydrates?
Complex sugars
Polyunsaturated sugars
Monounsaturated sugars
Simple sugars
81
Multiple Select
What are the two types of simple sugars?
Cellulose
Amylopectin
Monosaccharide
Disaccharide
82
Multiple Choice
Which is a monosaccharide?
lactose
sucrose
fructose
cellulose
83
Multiple Choice
The R in RNA is a sugar called
84
Multiple Choice
The D in DNA is a sugar called
85
Multiple Choice
The D in DNA is a sugar called
86
Multiple Choice
The D in DNA is a sugar called
87
Multiple Choice
The monosaccharide found in fruit is called
88
Multiple Choice
Another name for table sugar is
89
Multiple Choice
The disaccharide found in milk is called
90
Multiple Choice
The complex sugar found in plant cell walls is called
91

⬤ Lipids: Fats and oils
� Triglycerides
• Made up of one glycerol unit and three fatty acids
• Store energy for later use
� Phospholipids
• Similar to triglyceride structure, except with only two
fatty acids, and with a phosphorus-containing group
attached to glycerol
• The head attracts water (hydrophilic) and the double tail
does not (hydrophobic), thus forming stable double
layers (bilayers) in water
• Form membranes of cells
Organic Chemistry
Copyright © 2020 by Elsevier Inc. All rights reserved.
37
92


Triglyceride
Copyright © 2020 by Elsevier Inc. All rights reserved.
38
93


Phospholipids
Copyright © 2020 by Elsevier Inc. All rights reserved.
39
94

⬤ Lipids: Fats and oils
� Cholesterol
• Molecules have a steroid structure made up of multiple
rings
• Cholesterol stabilizes the phospholipid tails in cellular
membranes
• Cholesterol is converted into steroid hormones by the
body
Organic Chemistry
Copyright © 2020 by Elsevier Inc. All rights reserved.
40
95


Cholesterol
Copyright © 2020 by Elsevier Inc. All rights reserved.
41
96
Multiple Choice
Lipids that are a liquid at room temp are called
97
Multiple Choice
Triglycerides are made of
98
Multiple Choice
The structure of a phospholipid contains
99
Fill in the Blanks
100
Match
Match the following function with the type of lipid
Converted into hormones like steroid
Used for energy storage
Forms the cell membrane
cholesterol
triglycerides
phospholipids
cholesterol
triglycerides
phospholipids
101

⬤ Proteins
� Very large molecules made up of amino acids held
together in long, folded chains by peptide bonds
� Structural proteins
• Form various structures of the body
• Collagen: Fibrous protein that holds many tissues
together
• Keratin: Forms tough, waterproof fibers in the outer
layer of the skin
Organic Chemistry
Copyright © 2020 by Elsevier Inc. All rights reserved.
42
102



Protein
Copyright © 2020 by Elsevier Inc. All rights reserved.
43
103

⬤ Proteins
� Functional proteins
• Participate in chemical processes of the body
• Examples: Hormones, cell membrane channels
and receptors, enzymes
• Enzymes
� Catalysts: Help chemical reactions occur
� Lock-and-key model: Each enzyme fits a particular
molecule like a key fits into a lock
Organic Chemistry
Copyright © 2020 by Elsevier Inc. All rights reserved.
44
104


Enzyme Action
Copyright © 2020 by Elsevier Inc. All rights reserved.
45
105
Multiple Choice
Fibrous proteins that holds many tissues together
106
Multiple Choice
Structural protein that forms the tough, waterproof outer layer of the skin
107
Multiple Choice

Enzymes are ________ that act as ________.
proteins, catalysts
amino acids, proteins
proteins, amino acids
catalysts, carbohydrates
108
Multiple Choice
The ____________________ mechanism describes how each substrate must fit into the enzyme.
lock and key
hammer and nail
jigsaw puzzle
seesaw
109

⬤
Nucleic acids
� Made up of nucleotide units
• Sugar (ribose or deoxyribose)
• Phosphate
• Nitrogen base (adenine, thymine or uracil, guanine, cytosine)
� Deoxyribonucleic acid (DNA)
• Used as the cell’s “master code” for assembling proteins
• Uses deoxyribose as the sugar and uses A, T (not U), C, and
G as bases
• Forms a double helix shape
Organic Chemistry
Copyright © 2020 by Elsevier Inc. All rights reserved.
46
110
Multiple Choice
What are the three main parts of a nucleic acid?
111
Multiple Choice
What are the 4 bases for a DNA?
112


Deoxyribonucleic Acid
Copyright © 2020 by Elsevier Inc. All rights reserved.
47
113

⬤
Nucleic acids
� Ribonucleic acid (RNA)
• Used as a temporary “working copy” of a gene (portion of the
DNA code)
• Uses ribose as the sugar and uses A, U (not T), C, and G as
bases
� By directing the formation of structural and functional
proteins, nucleic acids ultimately direct overall body
structure and function
� Adenosine triphosphate (ATP): A modified nucleotide used
to transfer energy from nutrients to cellular processes, thus
acting as an energy-transfer “battery”
Organic Chemistry
Copyright © 2020 by Elsevier Inc. All rights reserved.
48
114
Categorize
single stranded
Adenine, Guanine, Cytosine, and uracil
Ribose
Intermediate in protein synthesis
Contains the blueprint for protein synthesis
Adenine, Guanine, Cytosine, Thymine
Double stranded helix
depxyribose
What are the differences between DNA and RNA?
115


Adenosine Triphosphate
Copyright © 2020 by Elsevier Inc. All rights reserved.
49

Chapter 2
Chemistry of Life
Copyright © 2020 by Elsevier Inc. All rights reserved.
Show answer
Auto Play
Slide 1 / 115
SLIDE
Similar Resources on Wayground

113 questions
Topik 1 LCTK Kimia UMC 2022
Presentation
•
10th Grade

108 questions
Wildlife and Exotic Radiology
Presentation
•
KG - University

108 questions
9.1 KOMUNITI DAN EKOSISTEM
Presentation
•
12th Grade

111 questions
Q1W3 ES Minerals Uses & Processing
Presentation
•
11th Grade

107 questions
Biology EOC Review 2
Presentation
•
9th - 10th Grade

106 questions
Heredity Notes - CP Bio
Presentation
•
11th Grade

107 questions
Lesson #35 - The Impact of World War II on Americans
Presentation
•
10th Grade

107 questions
Cognitive Bias and Logical Fallacies
Presentation
•
12th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Biology

16 questions
AP Biology: Unit 1 Review (CED)
Quiz
•
9th - 12th Grade

20 questions
AP Biology: Unit 3 Review (CED)
Quiz
•
9th - 12th Grade

21 questions
AP Biology: Unit 6 Review (CED)
Quiz
•
9th - 12th Grade

14 questions
AP Biology: Unit 4 Review (CED)
Quiz
•
9th - 12th Grade

20 questions
Food Chains and Food Webs
Quiz
•
7th - 12th Grade

30 questions
AP Biology Unit 8 Review
Quiz
•
9th - 12th Grade

15 questions
Evidence of Evolution
Quiz
•
8th - 12th Grade

15 questions
Pedigrees
Quiz
•
7th - 12th Grade