

Bohr Models
Presentation
•
Education
•
10th Grade
•
Easy
Anya Solomon
Used 19+ times
FREE Resource
7 Slides • 35 Questions
1

The Bohr Model
LQ: How many energy levels does an
atom of F have? How do you know?
2

Quick Review
What is an electron?
What charge does it have?
Where are they located in the atom?
3
Fill in the Blanks
4
Multiple Choice
Where are electrons located in an atom?
Inside the nucleus
Outside the nucleus
Near the protons
Next to the neutrons
5


Atomic Models
6


The Bohr Model
Electrons ONLY exist at these
levels, not in between.
– We call these levels “SHELLS ”
• Shells = period
The Bohr Model says that electrons exist in
very specific energy levels surrounding the
nucleus.
7
Multiple Choice
8


● The period (row) number tells us how many shells
● We can use the periodic table to predict how many
energy levels or shells an atom’s Bohr model will have.
The Bohr Model
9
Multiple Choice

On the periodic table, Groups are
Horizontal rows
Vertical columns
10
Multiple Choice

On the periodic table, Periods are
Horizontal rows
Vertical columns
11


Each shell can only hold a certain number of electrons
The Bohr Model
Electron Shell
Max # e-
1
2
2
8
3
18
4-7
18-32
12
Multiple Choice
The innermost energy level has a maximum of how many electrons?
1
2
6
8
13
Multiple Choice
The second energy level has a maximum of how many electrons?
1
2
6
8
14
Multiple Choice
The third energy level has a maximum of how many electrons?
2
6
8
18
15
Poll
I have watched the following video on how to draw a Bohr Model:
Yes I watched the video
No I didn't need to because I understand
16
Multiple Choice
Which Bohr model represents Neon?
17
Multiple Choice

Which element does this Bohr model represent?
Aluminum
Silicon
Magnesium
Cobalt
18
Multiple Choice
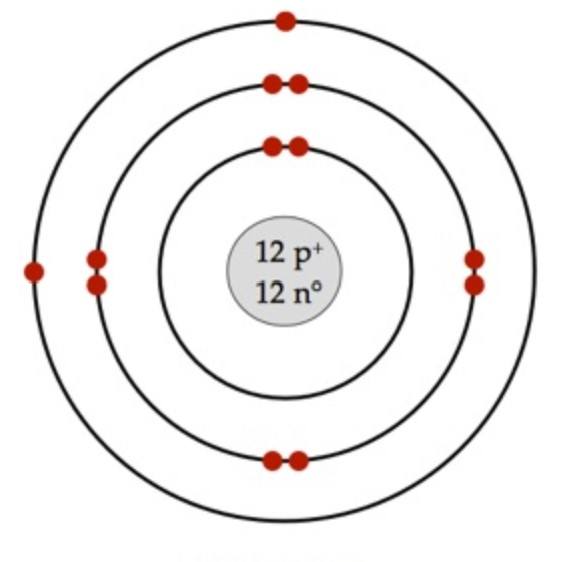
What element does this Bohr model represent?
Chromium
Magnesium
Carbon
Krypton
19
Multiple Choice

20
Multiple Choice

What element is represented in this Bohr Model?
Carbon
Hydrogen
Aluminum
Lithium
21
Multiple Choice

What is this element?
Beryllium (atomic #4)
Boron (atomic #5)
Carbon (atomic #6)
Nitrogen (atomic #7)
22
Multiple Choice

Name this element.
Neon
Magnesium
Sodium
Manganese
23
Multiple Choice
Which Bohr model represents Neon?
24
Multiple Choice

What element is this?
Nitrogen
Chlorine
Argon
Phosphorous
25
Multiple Choice

What element is this?
P
F
B
Mg
26
Multiple Choice
Which Bohr model represents Be?
27
Multiple Choice

How many electrons does this element have?
12
11
13
10
28
Multiple Choice

How many energy levels does this element have?
1
2
3
4
29
Multiple Choice
Which Bohr model represents Oxygen?
30
Multiple Choice

Is this the correct Bohr model for carbon (C)?
Yes
No
31
Multiple Choice

What element is this the Bohr model of?
Fluorine (F)
Potassium (K)
Aluminum (Al)
Boron (B)
32
Multiple Choice

What is the atomic number of this atom?
2
4
6
8
33
Multiple Choice

What is the mass number of this atom?
1
3
4
7
34


The Bohr Model
Valence Electrons:
electrons in the outermost
shell
Elements in the same group
act the same chemically
because they have the same
number of valence
electrons!
35
Multiple Choice
What is a valence electron?
Electrons in the first energy shell
Electrons in the second energy shell
Electrons in the outer shell
Total number of electrons
36
Multiple Choice

How many valence electrons?
2
4
6
8
37
Multiple Choice

How many valence electrons?
2
3
4
5
38
Multiple Choice

How many valence electrons?
2
3
4
5
39
Multiple Choice

What group does this element belong to?
1
2
17
18
40
Multiple Choice

Which element will have the most similar physical and chemical properties to the element Sulfur (S)?
Carbon
Neon
Phosphorus
Hydrogen
Oxygen
41
Multiple Choice

Which element will have the most similar physical and chemical properties to the element Barium (Ba)?
Caesium
Strontium
Astatine
Lanthanum
Oxygen
42
Multiple Choice

The Bohr Model
LQ: How many energy levels does an
atom of F have? How do you know?
Show answer
Auto Play
Slide 1 / 42
SLIDE
Similar Resources on Wayground

37 questions
Unit 2 Team Power Lesson A
Presentation
•
10th Grade

33 questions
Run-on Sentences
Presentation
•
10th Grade

34 questions
身体部位
Presentation
•
KG

39 questions
Cell terms part 3 2023
Presentation
•
10th Grade

36 questions
GAMBAR TEKNIK
Presentation
•
10th Grade

36 questions
Levels of Organization - Cells to Biome
Presentation
•
KG

36 questions
Regulation of Gene Expression
Presentation
•
9th - 10th Grade

34 questions
Preparing Vegetable dishes -Market Form of Vegetable
Presentation
•
10th Grade
Popular Resources on Wayground

25 questions
The Ultimate College Knowledge Quiz
Quiz
•
8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Math Review
Quiz
•
6th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade
Discover more resources for Education

10 questions
Fact Check Ice Breaker: Two truths and a lie
Quiz
•
5th - 12th Grade

40 questions
Math Large Group Session 1
Quiz
•
9th - 12th Grade

14 questions
Attributes of Linear Functions
Quiz
•
9th - 12th Grade

100 questions
NC Biology EOC Review
Quiz
•
9th - 10th Grade

15 questions
Memorial Day Trivia
Quiz
•
KG - 12th Grade

21 questions
Hidden Figures
Quiz
•
6th - 12th Grade

20 questions
Scatter Plots, Lines of Best Fit, Sampling
Quiz
•
9th - 11th Grade

20 questions
Food Chains, Food Webs, and Ecological Pyramids
Quiz
•
9th - 10th Grade