

2nd Sem M4 Chem Chapter 1
Presentation
•
Science, Chemistry
•
8th - 10th Grade
•
Practice Problem
•
Hard
Lady Alias
Used 3+ times
FREE Resource
59 Slides • 75 Questions
1
Atomic Mass and Mole
Learning Objectives:
Define the mole as a unit of measurement in chemistry and explain its importance.
Differentiate between atomic mass and molar mass, and understand the relationship between them.
Calculate molar mass of elements and compounds using atomic masses from the periodic table.
Perform conversions between moles, mass, and number of particles (atoms, molecules).
2
The Mole


3

4

5
Multiple Choice
Which of the following statements regarding the mole is NOT correct?
A mole is a counting unit
Equal to 6.02 x 1023 particles.
The concept of mole is similar to the concept of dozen
none of the above
6
Multiple Choice
When comparing a mole of oxygen and a mole of sulfur, how many atoms does each have?
oxygen has 16 grams of atoms
sulfur has 32.06 grams of atoms
both contains 6.02 x 1023 atoms
can not be determined
7
Multiple Choice
1 mole of Li
1 mole of Au
1 mole of Si
None, all are equal
8

9

10
Multiple Choice
How many atoms of iodine are in a mole of iodine (I)?
53
63.55g
126.9
6.02 x 1023
11
Multiple Choice
2
2 x (6.02 x 1023)
2 x (atomic mass)
2 ÷ (6.02 x 1023)
12
Multiple Choice
How many particles are found in a mole?
6.022 x 1023
6.022 x 1023
6022 x 1023
6022 x 1023
13
Multiple Choice
How many Carbon Atoms are in 4 moles of Carbon?
6.02 x 1023
1.33 x 1023
4.52
2.408 x 1024
14
Multiple Choice
How many particles are there in 2 moles of carbon-12 atoms
2
6.02 x 1023
3.01 x 1023
1.204 x 1024
15
Multiple Choice
How many atoms are there in 3 moles of carbon dioxide?
3
6.02 x 1023
1.806 x 1024
5.418 x 1024
16
Multiple Choice
How many particles are there in 3 moles of Argon atoms
3
6.02 x 1023
1.806 x 1024
1.806 x 1023
17

18

19

20

21
Multiple Choice
A mole = Avogadro's number = 6.022 x 1023
true
false
22
Multiple Choice
Molar number
Scientist's number
23
Multiple Choice
Which of the following statements regarding the mole is correct?
A mole is a counting unit equal to 6.02 x 1023 particles.
The number of particles in a mole is known as Avogadro’s number.
1 mole = Avogdro's number
all of the above
24
The Mole Concept
By Guzman
25
Avogadro's Number & Molar Mass
The mass of one mole of a substance is called Molar Mass (MM)
Units are g/mol
In an element 1 mole is equal to its Atomic Mass
26
Molar Mass
1 mole of atoms of any element will have a mass equal to its relative Atomic Mass
Its shown in grams
Once Again

1 Mol = 23 grams
2 Mol = 2 x 23 grams =
3 Mol = 3 x 23 grams =
27
Examples
How many grams are in 2 mol of Carbon (C)?
Look for the element
Look at the atomic mass
multiply the mol they are asking for

28
Multiple Choice
What are the units used for Molar Mass
grams
mols
g/mol
mol/g
29
Multiple Choice

How many grams are in 1 mole of Mg?
12 grams
24 grams
36 grams
6.022 x 1023 grams
30
Multiple Choice

How many grams are in 3 mol of carbon?
6 grams
12 grams
18 grams
36 grams
31
Multiple Choice

How many grams are in 5 mol of lithium
3 grams
15 grams
35 grams
7 grams
32
Multiple Choice

How many grams are in 10 mol of fluorine?
19 grams
190 grams
90 grams
9 grams
33
Formula Weight
Its the sum of all the atomic weights in a chemical formula.
to find the formula weight of a compound:
find the number of atoms of each element present in the compound
multiply the number of atoms by their respective weight
add the atomic weights of all the elements present.
34
Example: H2O
H2O contains 2 atoms of Hydrogen and 1 atom of Oxygen
Hydrogen: 2 x 1.0080 = 2.016
Oxygen: 1 x 16 = 16
2.016 + 16 = 18.016
35
Multiple Choice
what are the units used for Atomic Weight (Molecular Weight)?
amu
grams
g/mol
mol
36
Multiple Choice

Find the atomic weight (molecular weight) of Sugar - C12H22O11
198.176 amu
342.308 amu
144.132 amu
29.019
37
Multiple Choice

Find the atomic weight (molecular weight) of Vitamin A - C20H30O
286.46 amu
240.22 amu
29.019 amu
129.46 amu
38
Multiple Choice

Find the atomic weight (molecular weight) of carbon monoxide - CO
16 amu
12.011 amu
28.011 amu
29.019 amu
39
Multiple Choice

Find the atomic weight (molecular weight) acetic acid - CH3COOH
24.022 amu
31.035 amu
29.019 amu
60.054 amu
40
Putting these together
Gram Formula Mass (Molar Mass)
This is the combination of formula weight and mol
Moles are used not only to describe elements, but compounds as well.
When asked about the weight of 1 mol of a compound, you will follow the same steps as finding the molecular weight of your compound.
its units are g/mol
41
Molar Mass
molar mass = 1 mole
CO2
C=12 , O2 =16 x 2 = 32
12 + 32 = 44 g/mol

42
Example
Determine the gram formula mass of each compound: KMnO4
(this is asking for the mass of one mol of your compound)
K = 1 x 39.098 = 39.098
Mn = 1 x 54.938 = 54.938
O = 4 x 16 = 64
= 158.036 g/mol
43
Multiple Choice
What are the units used for Gram Formula Mass ?
amu
grams
mol
g/mol
44
Multiple Choice
Determine the gram formula mass of KCl
74.548 amu
35.45 amu
74.548 g/mol
35.45 g/mol
45
Multiple Choice

Molar Mass of CH4
12 g/mol
16 g/mol
4 g/mol
46
Multiple Choice
Determine the gram formula mass (Molar Mass) of H2SO4
98.076 mol
98.076 g/mol
98.076 g
98.076 amu
47
Multiple Choice
CO2 = 44 g/mol
How much would 3 moles of CO2 equal?
132g/mol
33g/mol
404g/mol
48
Multiple Choice
Molar Mass of K2CO3
138.205 g/mol
35 g/mol
78.2 g/mol
49
Multiple Choice
Molar Mass of Ag2O
231.7 g/mol
56.2 g/mol
734.4 g/mol
50
Calculating moles, given mass (or inverse)


51
Facts on The Mole
1 mole= 6.022 x 1023 atoms
The mole can either be a mass or a number of particles
Abbreviation is 'mol'
Commonly used term is molar mass = mass of one mole of a substance
has the units of g/mol or g.mol-1
We can easily convert between mass, moles, and particles
We can also easily calculate mass, molar mass, or moles (as long as we have two of these numbers)
52

53
Calculating number of particles, given moles (or inverse)



54

55
Multiple Choice
How many particles are there in 1 mole of oxygen gas?
[1st Hint: 1 molecule is counted as 1 particle]
1
6.02 x 1023
1.204 x 1024
12.04 x 1023
56
Fill in the Blanks
Type answer...
57
Fill in the Blanks
Type answer...
58
Fill in the Blanks
Type answer...
59
Multiple Choice
800 grams of 2NH3 (34 g/mol) is how many moles?
23.52 moles
.0425 moles
834 moles
60
Multiple Choice
How many grams are in 3 mol of carbon?
6 grams
12 grams
18 grams
36 grams
61
Examples
How many grams are in 2 mol of Carbon (C)?
Look for the element
Look at the atomic mass
multiply the mol they are asking for

62
Chemical Formulas
63

64

65
Multiple Choice
How many different elements are present in the following chemical formula: CO2
1
2
3
4
66
Multiple Choice
How many different elements are present in the following chemical formula: H2SO4
1
2
3
4
67
Multiple Choice
How many different elements are present in the following chemical formula: CoSO4
1
2
3
4
68
Multiple Choice
How many different elements are present in the following chemical formula: K2CO3
1
2
3
4
69
Multiple Choice
How many different elements are present in the following chemical formula: Ag2O
1
2
3
4
70

71
Multiple Choice
How many Hydrogen atoms are present in glucose: C6H12O6
6
12
24
1
72
Multiple Choice
How many Oxygen atoms are present in H2O?
1
2
3
4
73
Multiple Choice
How many Bromine atoms are present in AlBr3?
1
2
3
4
74
Multiple Choice
How many Calcium atoms are present in CaCO3?
1
2
3
4
75
Multiple Choice
How many Sulfur atoms are present in Na2S04?
1
2
3
4
76

77
Multiple Choice
How many Sulfur atoms are in Cr(SO4)3?
1
3
4
12
78
Multiple Choice
How many Oxygen atoms are in Cr(SO4)3?
1
3
4
12
79
Multiple Choice
How many Chromium atoms are in Cr(SO4)3?
1
3
4
12
80

81

82
Multiple Choice
How many molecules of H2O are present in: 3H2O
1
2
3
4
83
Multiple Choice
How many atoms of Hydrogen are present in: 3H2O
9
2
3
6
84
Multiple Choice
How many atoms of Oxygen are present in: 3H2O
9
2
3
6
85
86
Multiple Choice
The elements in water are hydrogen and _________.
chlorine
oxygen
nitrogen
potassium
87
88

89
Fill in the Blanks
Type answer...
90
Multiple Select
1. How many total atoms are in this compound?
2. How many atoms of Hydrogen are in C6H12O6?
12 total atoms
24 total atoms
12 atoms of Hydrogen
24 atoms of Hydrogen
91
Fill in the Blanks
Type answer...
92
Valence Electrons
These are the number of ELECTRONS on the OUTER SHELL and they are responsible for how the atom BONDS to other atoms.

93
Multiple Choice
What are valence electrons?
The total number of electrons in an atom
The number of electrons in the outermost shell
The number of electrons in the second shell
The number of protons in the outermost shell
94
Multiple Choice
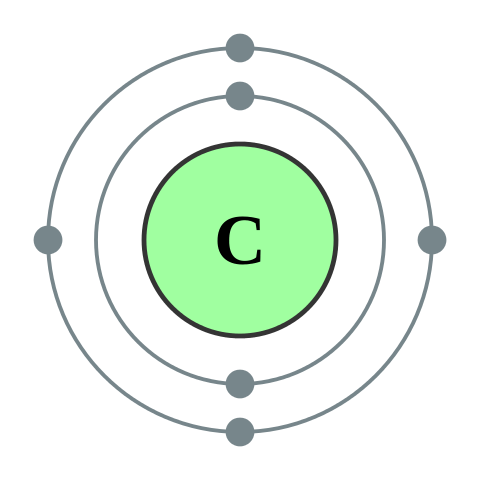
How many valence electrons does carbon have?
4
5
6
7
95
Multiple Choice

How many ELEMENTS are in this chemical equation?
3
4
9
10
96
Multiple Choice
Acetic acid is a compound found in vinegar. The chemical formula for acetic acid is CH3COOH.
How many different ELEMENTS are in acetic acid?
3
5
6
8
97

98

99

100
Multiple Choice
All ____________ are made up of two or more atoms bonded together.
atoms
molecules
electrons
elements
101
Multiple Choice
102
The REACTANTS are the starting atoms. Basically the reactants mix together and REACT! The PRODUCTS are what you end up with after the chemical reaction occurs. The atoms rearrange to make something new. The something NEW are the PRODUCTS.

103
Use RAP to memorize which part of the chemical equation are the reactants and which part are the products

104
Multiple Choice
NaOH + HCl --> NaCl + H2O Identify the reactants in the chemical equation above.
NaOH and HCl
NaCl and H2O
HCl and H2O
HCl and NaCl
105
Multiple Choice
2AgNO3 + CaCl2 --> 2AgCl + Ca(NO3)2
Identify the products in the chemical equation above.
2AgNO3 and CaCl2
2AgCl and Ca(NO3)2
CaCl2 and Ca
Ag, Cl, N, and Ca
106
Multiple Choice
Cu + 2AgNO3 ⟶ 2Ag + Cu(NO3)2 How many copper (Cu) atoms are there in Cu(NO3)2 ?
1
2
5
6
107
Multiple Choice
How many MOLECULES are on the PRODUCT side of the chemical equation NaOH + HCl --> NaCl + H2O
1
2
4
5
108
Poll

THE END
Did you learn anything today????
Yes, I did and excited about learning more.
No. I don't care about my grade.
Yes, kinda. The holiday can't come any sooner!!!!
Yes. I also like my science teacher and class.
109
Counting Atoms & Chemical Formulas

110

111

112

113

114

115

116

117

118

119

120
Multiple Choice
What elements are present in glucose, C6H12O6?
Calcium, Hydrogen, and Oxygen
Carbon, Hydrogen, and Oxygen
Carbon and Oxygen
Calcium and Oxygen
121
Multiple Choice
How many carbon (C) atoms are present in glucose, C6H12O6?
6
12
18
24
122
Multiple Choice
How many hydrogen (H) atoms are in glucose, C6H12O6?
6
12
18
24
123
Multiple Choice
How many total elements make up glucose, C6H12O6?
1
3
6
24
124
Multiple Choice
How many total atoms make up glucose, C6H12O6?
3
6
12
24
125

126

127

128

129

130

131

132
Multiple Choice
How many atoms of potassium (K) are in 2 molecules of K2SO4?
1
2
4
8
133
Multiple Choice
How many atoms of sulfur (S) are in 2 molecules of K2SO4?
1
2
4
8
134
Multiple Choice
How many atoms of oxygen (O) are in 2 molecules of K2SO4?
1
2
4
8
Atomic Mass and Mole
Learning Objectives:
Define the mole as a unit of measurement in chemistry and explain its importance.
Differentiate between atomic mass and molar mass, and understand the relationship between them.
Calculate molar mass of elements and compounds using atomic masses from the periodic table.
Perform conversions between moles, mass, and number of particles (atoms, molecules).
Show answer
Auto Play
Slide 1 / 134
SLIDE
Similar Resources on Wayground

129 questions
A&P I Chapter 5 Review
Presentation
•
University

118 questions
Biology EOC Review Macromolecules
Presentation
•
11th - 12th Grade

134 questions
Carbon Lesson 1
Presentation
•
11th Grade

122 questions
OP 3/4 Industrialization 1865-1898 25/26
Presentation
•
11th Grade

136 questions
P4Care Outcome 1 Revision
Presentation
•
10th Grade - University

123 questions
untitled
Presentation
•
KG - University

119 questions
Biology Summary EOC Review
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade