

5.2 Reaction Rate Notes
Presentation
•
Science
•
9th Grade
•
Hard
Matthew Koch
Used 7+ times
FREE Resource
28 Slides • 10 Questions
1

2
Multiple Choice
3
Multiple Choice
4
Multiple Choice

How could we make this reaction happen more quickly?
Decrease the concentration of the acid
crush the chalk to increase the surface area
Put the test tube in an ice bath
5
Multiple Choice
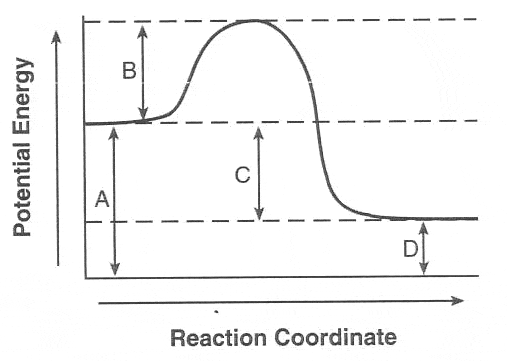
The minimum amount of energy needed for colliding particles to react is called
chemical energy.
kinetic energy.
activation energy.
potential energy.
6
Multiple Choice
they slow down
nothing
they stop moving
they speed up
7

8

9

10

11

12

13

14

15

16

17

18

19

20

21

22

23

24

25

26

27

28

29

30

31

32

33

34
Multiple Choice
Products will form faster during a chemical reaction if
the particle size of the reactants are larger.
temperature is decreased.
concentration of the reactants are increased.
the reaction is not stirred.
35
Multiple Choice
What is the purpose of a catalyst?
they help to slow down a reaction
they raise the activation energy
they lower the activation energy
they are consumed by the reaction
36
Multiple Choice

The following graph shows two different reaction pathways for the same overall reaction at the same temperature.
Which pathway is slower and why?
red, because the activation energy is larger
blue, because the activation energy is lower
both reactions progress at the same rate
37
Multiple Choice
Increasing pressure causes the reacting particles
to bond together
to repel from each other
gain more kinetic energy
to move closer together
38
Multiple Choice

Show answer
Auto Play
Slide 1 / 38
SLIDE
Similar Resources on Wayground

35 questions
Chapter 10 Forces and Motion Lesson 1 & 2
Presentation
•
8th Grade

34 questions
Pax Romana & Contributions
Presentation
•
9th Grade

31 questions
Intro to Genetics Lesson
Presentation
•
10th Grade

33 questions
Diffusion and Osmosis Notes
Presentation
•
9th Grade

33 questions
Wave Interaction Notes
Presentation
•
8th Grade

33 questions
Chemical Properties
Presentation
•
8th Grade

35 questions
Cell Organelles
Presentation
•
9th Grade

31 questions
Cell division terms 2 - mitosis prep
Presentation
•
10th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Science

20 questions
Earth Day Trivia
Quiz
•
9th - 12th Grade

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

20 questions
Earth Day
Quiz
•
3rd - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

10 questions
Exploring Natural Selection Concepts
Interactive video
•
6th - 10th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

10 questions
Exploring Air Masses and Weather Fronts
Interactive video
•
6th - 10th Grade

42 questions
Practice Missouri Biology EOC Test
Quiz
•
9th Grade