

Heat and Temperature
Presentation
•
Science
•
5th Grade
•
Practice Problem
•
Medium
Rudi Setyawan
Used 2+ times
FREE Resource
24 Slides • 50 Questions
1
Temperature and Heat

2
When you think of the word temperature, you probably think of it as a way to measure heat.
WRONG!!!!!!!!!
3
There are words that we use in our everyday lives that are incorrectly used in a scientific context. Heat and temperature are two examples. Starting today, you will need to begin thinking about these words differently.
4
Let's begin by taking a look at the word temperature. Temperature is actually a measure of the average kinetic energy of the atoms or molecules of a substance. Watch the video on the next screen.
5
6
Multiple Choice
As the kinetic energy increases, what happens to temperature?
It decreases.
It increases.
7
Watch the next video that also shows you that temperature is a measure of kinetic energy.
8
9
We also use the word "heat" incorrectly. Heat is actually a process. Heat is the transfer of energy from a substance at a higher temperature to a substance at a lower temperature. We will be exploring heat in the next lab.
10
Heat and temperature
If a hot body is brought into contact with a cold body, heat will be transferred from the hot body to the cold body.
In order to measure the quantity of heat, we must have a suitable heat unit.
Heat is a form of energy and therefore has the same unit as any other form of energy, ie the joule (J).

11
12
Heat
the thermal Energy that is transferred from one system to another
–Movement of atoms
–Total potential and kinetic energy of molecular motion in a substance.
–Depends on speed, number (size and mass), and type of particles.
–Transfers between objects
–Units Joules, Calorie
symbol is Q, Q = mCΔT
A large object can have more heat that a small object with a higher temperature. Ex. water trough and hot cup of coffee
13
Temperature
a measurement of the average kinetic energy of the particles in a system
–Thermo =heat
–Meter = to measure
–Measurement of the average kinetic energy of molecular motion in a substance.
–Hotness or coldness
–Does not depend on the speed or type of particle.
–Units Celcius, K, F
14

15
16

17

18

19

20

21
22

23
24
Measuring using a thermometer
The correct technique to read from a thermometer is as shown in Diagram C.

25
26
Multiple Choice
How does heat transfer when you hold an ice cube in your hand?
from the cold ice cube to your warm hand
the cold ice cube makes your hand cold
from your warm hand to the cold ice cube
very carefully
27
Multiple Choice
McDonald's keeps my Big Macs warm under a heat lamp which transfers heat through
conduction
convection
radiation
science stuff
28
Multiple Choice
Smoke rises at a campfire because
the heated gas is less dense
the heated gas is more dense
the heated gas conducts heat to space
smoke follows beauty
29
Multiple Choice
When I drink coffee in the morning, I warm my hands on the mug by what type of heat transfer?
conduction
convection
radiation
coziness
30
Multiple Choice
Energy that transfers from one substance to another until the temperatures are equal is called...
Joe
temperature
MELTS
heat
31
Multiple Choice
Heat transfer is really neat. It always moves from..
hot to cold
cold to hot
big to little
stop singing
32
Multiple Choice
When I cook bacon, it touches the pan. It is cooked because
heat transfers from the pan to the bacon through convection
heat transfers from the pan to the bacon through conduction
heat transfers from the bacon to the pan through conduction
heat is transferred as energy
33
Multiple Choice
Which type of heat transfer occurs as heat moves in a circular motion in a liquid or gas?
conduction
convection
radiation
cooking
34
Multiple Choice
Which type of heat transfer travels through empty space and does need a state of matter?
conduction
convection
radiation
waves
35
Multiple Choice
Is heat energy?
Yes, its thermal energy
No, Temperature is the one that is energy
Yes, its Kinetic Energy
No, its just a measurement
36
Multiple Select
If a pan is heated at 40°C and then the stove is turned on, is the pan going to instantly get hotter?
Yes, the pan will get hot as soon as the stove is on
Yes, the pan will get hot after a while
No, first it will lower its temperature to get into equilibrium and then it will get hotter
No, it is already hot
37
Multiple Choice
How does heat transfer when you hold an ice cube in your hand?
from the cold ice cube to your warm hand
the cold ice cube makes your hand cold
from your warm hand to the cold ice cube
very carefully
38
Multiple Select
Some Examples of man-made sources of heat are:
Iron
Oven
Sun
Stove
39
Multiple Choice
The liquid material inside the thermometer is called:
Water
Mercury
Silver
solution
40
Multiple Choice
41
Multiple Choice
42
Multiple Choice
What is the meaning of temperature?
The amount of water in a container.
The size of a shadow.
The measure of how cold or hot something is.
Celsius
43
Multiple Choice
_______________________ are materials that do not allow heat to move easily through them.
Insulators
Electricity
Sun
Conductors
44
Multiple Choice
45
Multiple Choice
46
Multiple Choice
47
Multiple Choice
48
Multiple Choice
49
Multiple Choice
50
Multiple Choice
51
Multiple Choice
52
Multiple Choice
53
Multiple Choice
54
Multiple Choice
55
Multiple Choice
56
Multiple Choice

When we put a spoon in the hot coffee ...
... the spoon heats up, because the coffee transfers heat to the spoon.
... its temperature remains the same.
... the spoon gets colder.
... the coffee temperature remains the same.
57
Multiple Choice

When on a cold day we open the window ...
... the cold enters.
... the cold goes out.
... the heat comes out.
... none of the options.
58
Multiple Choice

We say that two objects brought into contact reach thermal equilibrium when ...
... one of the objects gets hotter.
... they remain with the same temperature.
... the two objects are at the same temperature.
... objects increase their temperatures.
59
Multiple Choice

A substance loses thermal energy (heat) to the surroundings at a steady rate. The graph shows how the temperature of the substance changes with time.
What could the portion PQ of the graph represent?
gas condensing
gas cooling
liquid cooling
liquid solidifying
60
Multiple Choice
61
Multiple Choice
62
Multiple Choice
63
Multiple Choice
64
Multiple Choice
65
Multiple Choice
66
Multiple Choice
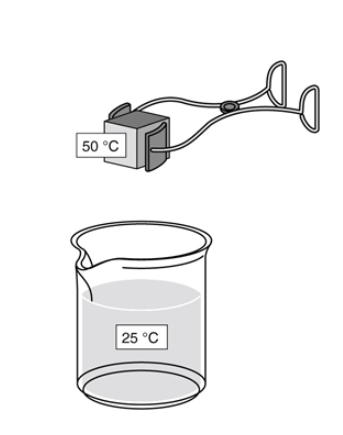
As part of a lab experiment, Tasha drops a metal cube into a beaker of water, as shown in the figure below.
After energy in the form of heat transfers between the substances, what is the final temperature of the water?
The final temperature is 25 °C because there is more water than metal.
The final temperature is 50 °C because the metal warms the water to the temperature of the metal.
The final temperature is between 25 °C and 50 °C because energy is transferred from the metal to the water.
The final temperature is between 25 °C and 50 °C because most of the thermal energy is lost to the air around the substances.
67
Multiple Choice
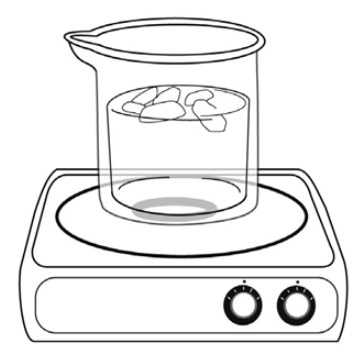
Emerson places a beaker of water on a hotplate and turns the hotplate on. The temperature in the room is 25 ºC. After the water heats to 75 ºC, she adds pieces of ice to the beaker.
Which of the following describes a transfer of heat energy that occurs during this experiment?
from the air to the water
from the ice to the beaker
from the water to the ice
from the water to the hotplate
68
Multiple Choice

69
Multiple Choice

The egg is cooking because heat is transferred from the frying pan via
Conduction
Convection
Radiation
70
Multiple Choice
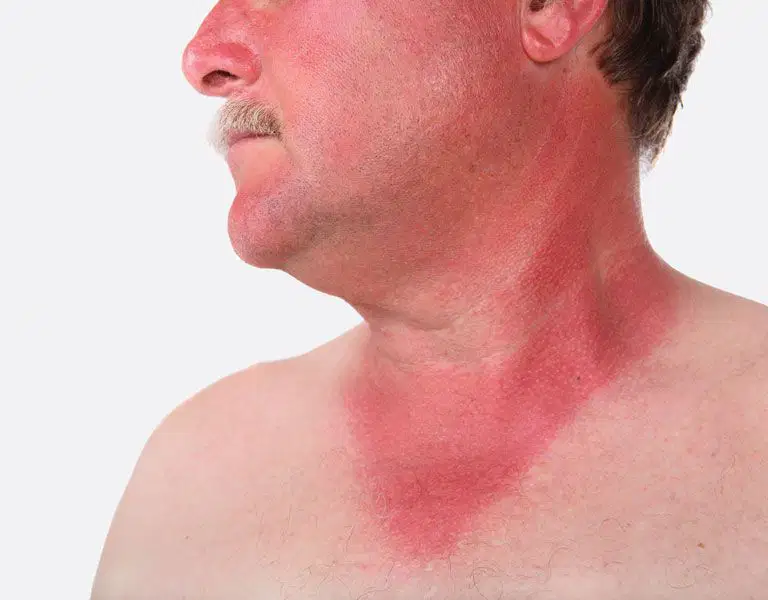
A sunburn is due to
conduction
convection
radiation
71
Multiple Choice

The movement of water in a boiling kettle is an example of
conduction
convection
radiation
72
Multiple Choice

During the summer, Chang takes a glass of milk from the refrigerator and places it on the kitchen counter. Over time, energy as heat is transferred to the milk from the air. Which change would most likely occur?
The temperature of the milk will increase.
The temperature of the milk will decrease.
The milk will change from a liquid to a gas.
The milk will change from a liquid to a solid.
73
Multiple Choice

Kito puts his spoon into a helping of fresh, hot, mashed potatoes as shown in the picture. Which process takes place when the objects come together?
The temperature of the spoon increases, but its thermal energy does not change.
Energy in the form of heat is transferred by conduction from the warmer mashed potatoes to the cooler spoon.
The thermal energy of both the mashed potates and the spoon increase as the energy in the form of heat is transferred between them.
The average temperature of the spoon does not change.
74
Multiple Choice

An ice cube floats in warm water in a container placed outside on a hot, sunny day. Identify the processes by which the warm water contacting the ice cube and the sun warming the air transfer energy.
Water transfers energy to ice through radiation. Sun transfers energy to the air through conduction.
Ice transfers energy to water through conduction. Air transfers energy to the Sun through radiation.
Water transfers energy to ice through conduction. Sun transfers energy to the air through radiation.
Both transfer energy through conduction.
Temperature and Heat

Show answer
Auto Play
Slide 1 / 74
SLIDE
Similar Resources on Wayground

68 questions
Classification of Living Things
Presentation
•
5th Grade

68 questions
Laws of Motion Review Honors 23-24
Presentation
•
6th Grade

65 questions
Ch. 7 - Amazing Animals - Animal habitats
Presentation
•
5th Grade

68 questions
TA 5 - Unit 8 - What are you reading?
Presentation
•
5th Grade

68 questions
Earth Structure and Plate Tectonics
Presentation
•
6th Grade

68 questions
Classification of Animals
Presentation
•
5th Grade

70 questions
Water Cycle Quiz
Presentation
•
5th Grade

67 questions
Matter and Their Three States
Presentation
•
5th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

20 questions
Science Review
Quiz
•
5th Grade

20 questions
Food Chains and Food Webs Review
Quiz
•
5th Grade

45 questions
Science SOL Review 1
Quiz
•
5th Grade

118 questions
5th Grade SSA Review_Mr. Hite
Quiz
•
3rd - 5th Grade

50 questions
5th Grade Science EOG Review
Quiz
•
5th Grade

43 questions
4th and 5th Grade VA Science SOL Review
Quiz
•
5th Grade

34 questions
5th Grade MAP Science Session 2 Released
Quiz
•
5th Grade

20 questions
Life Science Review
Quiz
•
5th Grade