

Atom/Periodic Table
Presentation
•
Biology
•
7th Grade
•
Medium
Standards-aligned
JULIAN SOTO
Used 14+ times
FREE Resource
35 Slides • 21 Questions
1

2

3

4

5

6

7

8

9

10

11

12

13

14

15

16
Multiple Choice

How many neutrons in Fluorine?
9
18
19
10
17
Multiple Choice
If I had an atom with 5 electrons and 5 protons, what is its atomic number?
5
10
15
20
18
Multiple Choice

From this element key of Nitrogen, determine the atomic mass of nitrogen
7
7.01
14.01
21.01
19
Multiple Choice

What does the atomic number tell us about an element?
number of neutrons
number of protons
number of isotopes
atomic mass
20
21

22

23
Multiple Choice

Look at the periodic table. Which list of elements forms a group on the periodic table?
Li, Be, B, C, N, O, F, and Ne
He, Ne, Ar, Kr, Xe, and Rn
B, Si, As, Te, and At
Sc, Ti, V, Cr, Mn, Fe, Co, Cu, Ni, and Zd
24

25
Multiple Choice

When elements are in the same ROW they are all in the same-
Chemical Symbol
Period
Family
26

27
Multiple Choice
The periodic table is mostly comprised of what type of element?
Metals
Nonmetals
Metalloids
28
Explanation Slide...
Metals are good conductors of electricity, making this the correct property among the options provided.
29
Multiple Choice
Which of the following is a property of metals?
Poor conductor of heat
Brittle and dull
Good conductor of electricity
Does not react with acids
30

31
Multiple Choice
What are the vertical columns on the Periodic Table known as?
Periods
Metals
Groups
Nonmetals
32
Multiple Choice

What classification of element is Lithium?
Metal
Nonmetal
Metalloid
33

34

35
Multiple Choice
36
Multiple Choice

Which of the following elements is in Period 2 Group 18?
Iodine
Seaborgium
Neon
Gold
37
Multiple Choice

Identify the element from Period 3 Group 13.
Aluminum
Neon
Nickel
Hydrogen
38

39
Multiple Choice

40
Multiple Choice
41
Multiple Choice

42
Multiple Choice
43

44
45

46

47

48

49
50
Multiple Choice
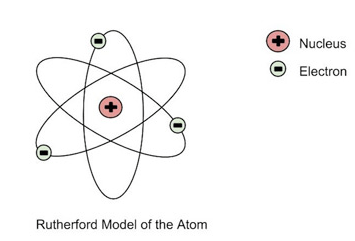
51

52
Multiple Choice

53

54
Multiple Choice
What is the most current atomic model?
55
Multiple Choice
What is the quantum mechanical model of the atom and how does it differ from Bohr's model?
The quantum mechanical model considers electrons as having both particle-like and wave-like properties, represented by probability distributions called orbitals, allowing for more accurate predictions compared to Bohr's model.
The quantum mechanical model only considers electrons as particles, ignoring their wave-like properties.
The quantum mechanical model uses circular orbits for electrons, similar to Bohr's model.
The quantum mechanical model predicts electron positions with absolute certainty, unlike the probabilistic nature of Bohr's model.
56


Show answer
Auto Play
Slide 1 / 56
SLIDE
Similar Resources on Wayground

55 questions
Meiosis
Presentation
•
7th Grade

51 questions
Microorganisms
Presentation
•
7th Grade

49 questions
Measures of Variation 2020
Presentation
•
7th Grade

47 questions
Proportional Relationships
Presentation
•
7th Grade

52 questions
LEAP REVIEW #1
Presentation
•
6th Grade

49 questions
8th Grade 1-6 Scientific Measurement & Math
Presentation
•
8th Grade

54 questions
KS3 E-Safety 1: Account Security
Presentation
•
7th Grade

52 questions
Properties of Waves Interactive Notes
Presentation
•
7th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Biology

20 questions
Food Chains and Food Webs
Quiz
•
7th - 12th Grade

18 questions
Population Dynamics
Quiz
•
7th - 12th Grade

10 questions
Exploring Evidence of Evolution
Interactive video
•
6th - 10th Grade

10 questions
Exploring Food Webs and Energy Pyramids
Interactive video
•
6th - 10th Grade

10 questions
Photosynthesis and Cellular Respiration Concepts
Interactive video
•
6th - 8th Grade

20 questions
Cell Division
Quiz
•
7th Grade

10 questions
Comparing Mitosis and Meiosis: Key Differences and Stages
Interactive video
•
6th - 10th Grade

10 questions
Comparing Prokaryotic and Eukaryotic Cells
Interactive video
•
6th - 10th Grade