

Introduction to the mole
Presentation
•
Chemistry
•
10th Grade
•
Practice Problem
•
Medium
+2
Standards-aligned
ELIZABETH JOANNA FAYE FOSTER
Used 6+ times
FREE Resource
7 Slides • 10 Questions
1
Introduction to the Mole (the science one)
And some practice problems
2
The Video below is a review of what we did in class on Wednesday January 8th. You don't have to watch it, but it may help if you get stuck.
(It is also on Google Classroom if you need it).
3
The Mole is a quantity
Just like the saying "a dozen" refers to a specific number of something (12), saying "a mole" refers to a certain number of a thing.
A mole of a thing is equal to Avogadro's number of that thing.
4
Avogadro's number
"Avogadro's number" is the specific name we give to the number
6.02214076 x 1023
5
Fill in the Blanks
6
Multiple Choice
Based on the information on the last few slides, how many blueberries would be in 1 mole of blueberries?
6.02214076 x 1023
12
600000
you can't use moles to measure blueberries! That would be like using a scale to measure volume.
7
Hopefully it is obvious we use moles to measure very tiny things.
Specifically, atoms and molecules. The reason that moles are helpful is because
1 mole of a specific substance weighs the same amount in grams as 1 molecule that substance weighs in atomic mass units (amu)
8
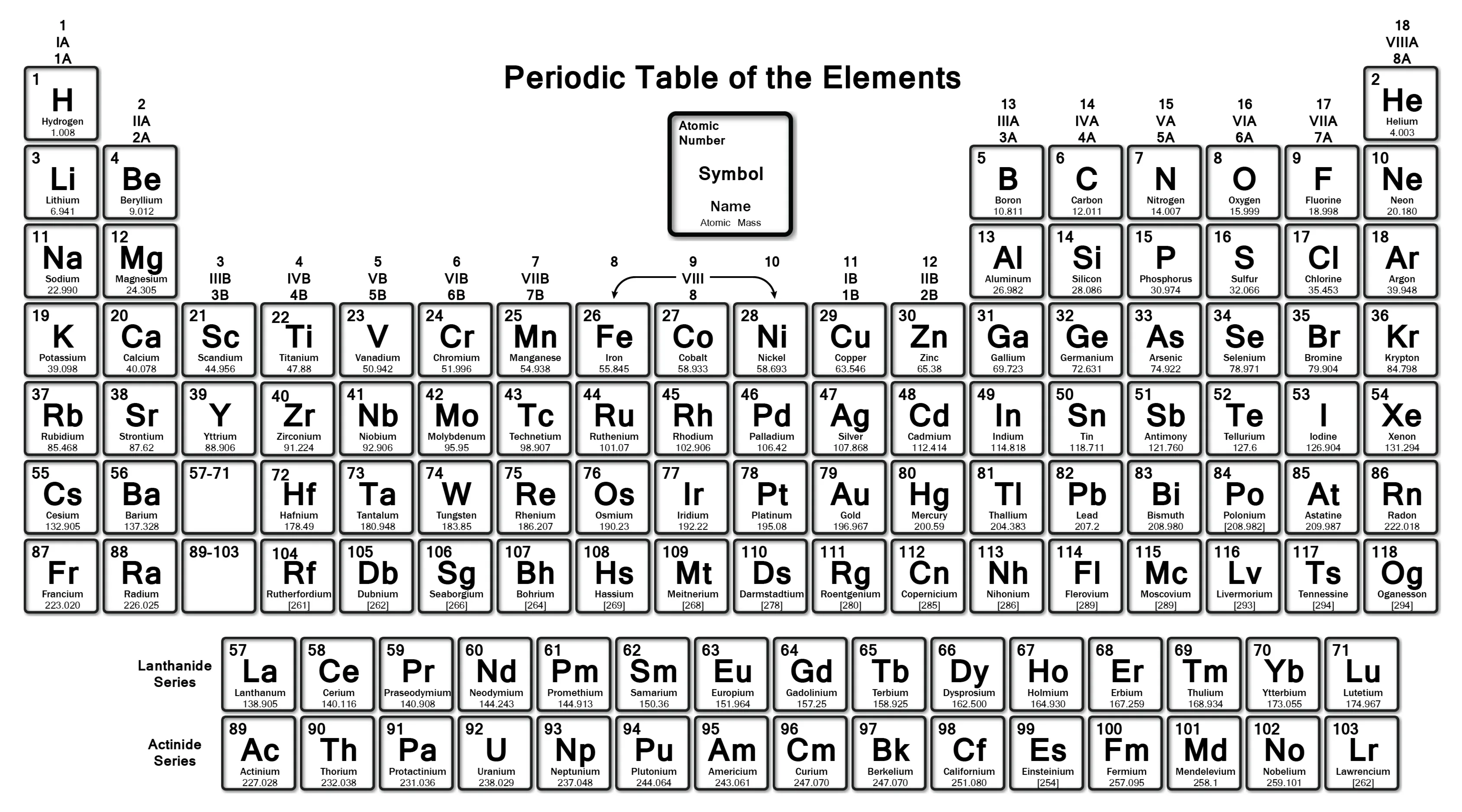
How to figure out the weight of a molecule:
First you figure out the number of each type of atom in the molecule, then multiply the number of each atom by the weight of the atom (based on the periodic table) and finally add all those together.
(I show you how in the video at the beginning and on Google Classroom)
9
Dropdown
10
Fill in the Blanks
Type answer...
11
amu to grams
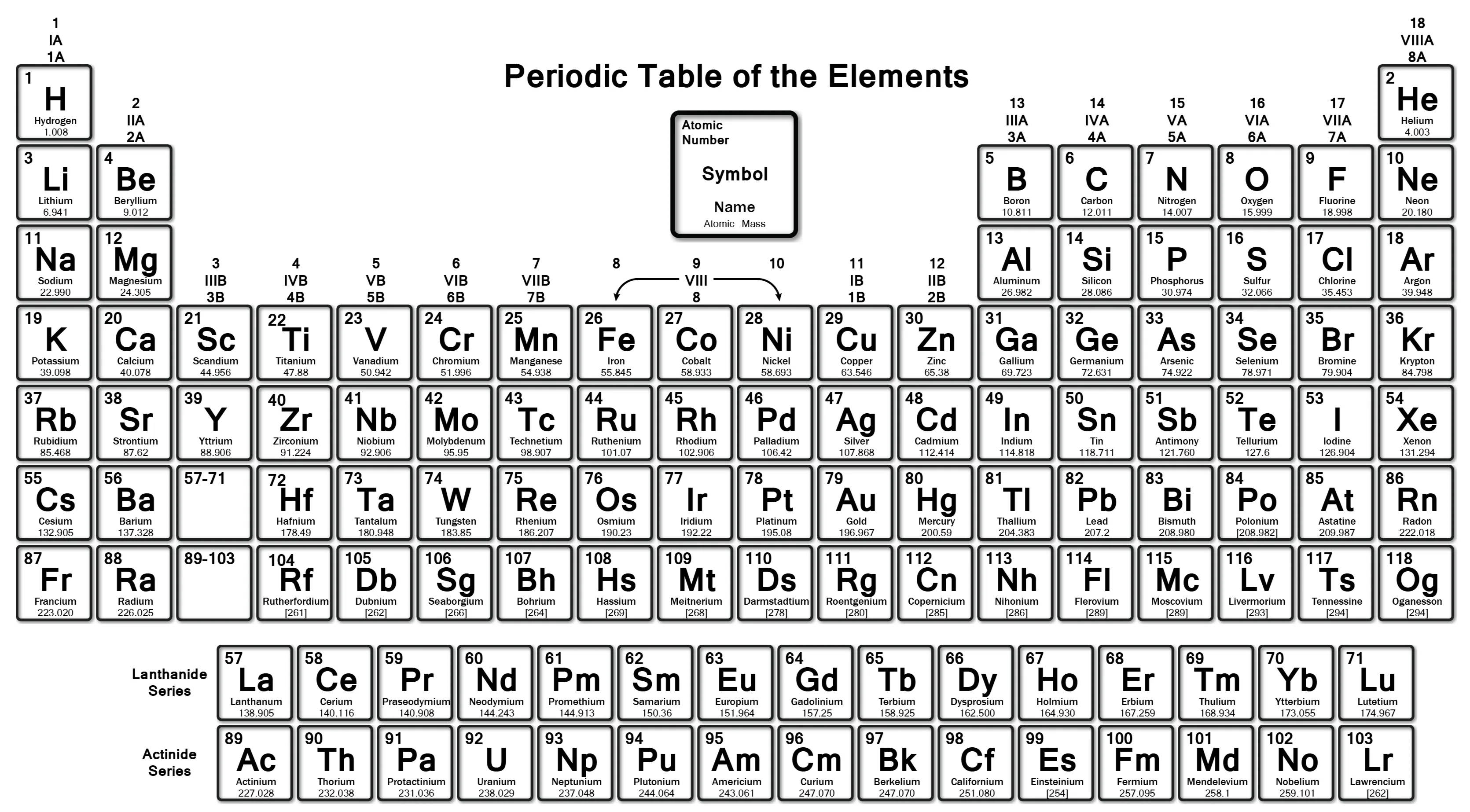
Since 1 molecule of NaOH weighs 40 amu, 1 mole of NaOH weighs 40 grams!
See how convenient that is!
Now for some practice!
12
Fill in the Blanks
Type answer...
13
Fill in the Blanks

Type answer...
14
Fill in the Blanks

15
Multiple Choice

Which weighs more, one gram of water or one gram of carbon dioxide?
the water
the carbon dioxide
they both weigh the same
16
Multiple Choice

Which weighs more, one milliliter of water or one milliliter of carbon dioxide?
the water
the carbon dioxide
they both weigh the same
17
Multiple Choice

Which weighs more, one mole of water or one mole of carbon dioxide?
the water
the carbon dioxide
they both weigh the same
Introduction to the Mole (the science one)
And some practice problems
Show answer
Auto Play
Slide 1 / 17
SLIDE
Similar Resources on Wayground

12 questions
Living Things
Presentation
•
KG

11 questions
Teenager 5 Session 24: Pronunciation & Speaking
Presentation
•
KG

13 questions
Sentence Fragments/Run-Ons Lesson
Presentation
•
10th Grade

13 questions
counting atoms
Presentation
•
10th Grade

12 questions
Intro to Thermochemistry
Presentation
•
10th Grade

16 questions
stoichiometry
Presentation
•
10th Grade

10 questions
IGCSE Chemistry Revision
Presentation
•
10th Grade

12 questions
Comma Rules
Presentation
•
10th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

10 questions
Exploring Properties and Models of Acids and Bases
Interactive video
•
6th - 10th Grade

19 questions
Review Set 3 - Moles & Chemical Formulas
Presentation
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Stoichiometry Practice
Quiz
•
10th Grade

10 questions
Identifying Types of Chemical Reactions
Interactive video
•
6th - 10th Grade

20 questions
Redox Reactions
Quiz
•
10th - 12th Grade

20 questions
pH and pOH Calculations
Quiz
•
10th - 11th Grade