

Properties of Water
Presentation
•
Science
•
7th Grade
•
Medium
+3
Standards-aligned
James Gonzalez
Used 1+ times
FREE Resource
1 Slide • 19 Questions
1
2
Multiple Choice
What type of bond is found BETWEEN water molecules?
Hydrogen bonds
Ionic bonds
Nonpolar covalent bonds
Polar covalent bonds
3
Multiple Choice
What word describes when water is attracted to other substances?
Cohesion
Adhesion
Capillary action
Surface tension
4
Multiple Choice
When water molecules stick easily to other water molecules, this is called what?
Cohesion
Adhesion
Solution
Polar molecule
5
Multiple Choice
The tightness across the surface of water that enables insects to walk on it is
Adhesion
Capillary action
Surface tension
Polarity
6
Multiple Choice
Large bodies of water help to keep nearby climates from being too hot or too cold because they
Absorb heat in the spring and slowly release that heat in the fall
Absorb heat in the fall and slowly release that heat in the spring
Absorb heat in the summer and slowly release that heat in the winter
Absorb heat in the winter and slowly release that heat in the summer
7
Multiple Choice

8
Multiple Choice

9
Multiple Choice
Which of the following can water NOT dissolve?
Salt
Sugar
Oil
Baking Soda
10
Multiple Choice
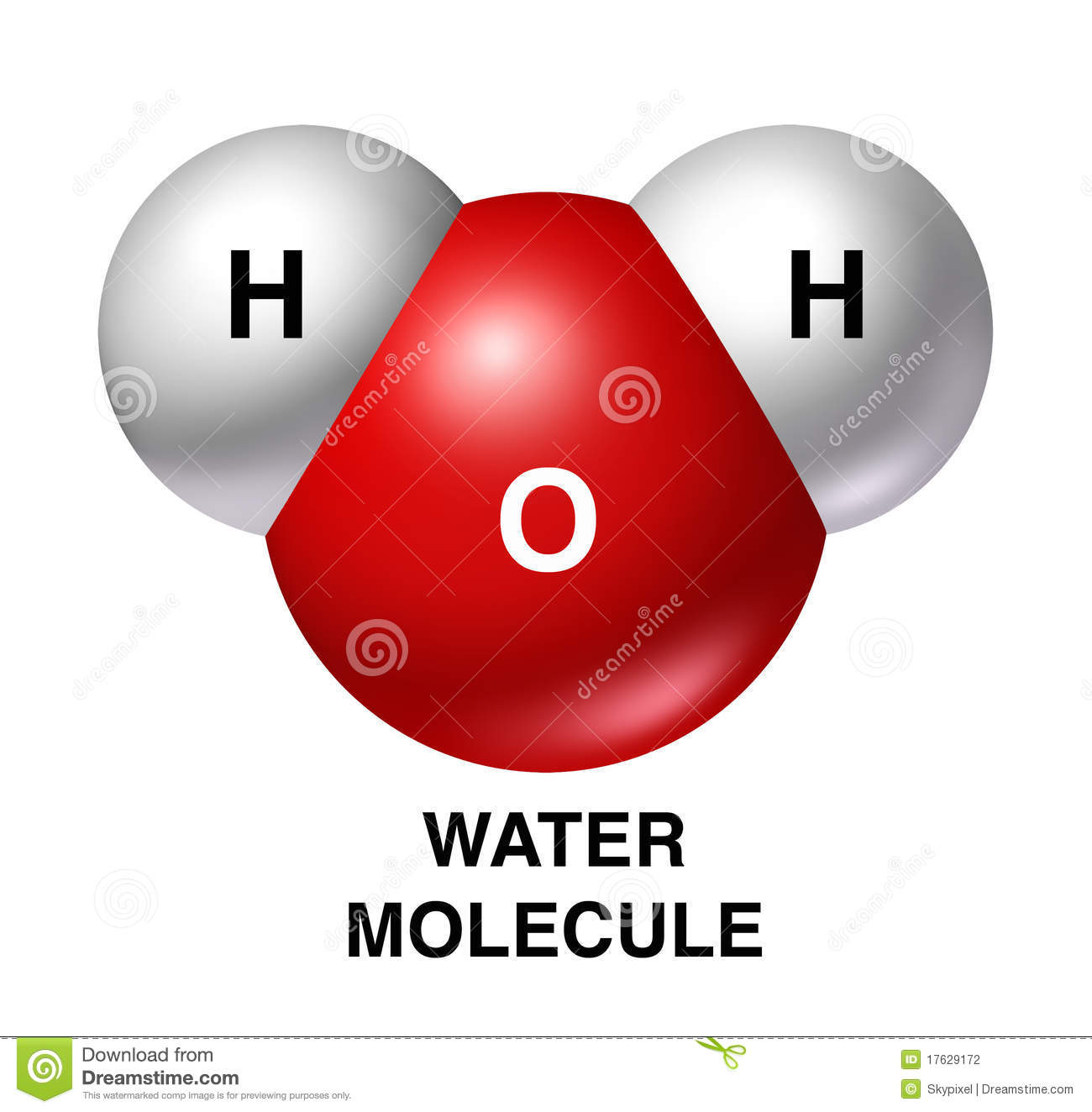
Which part of a water molecule has a Positive Charge?
The Hydrogen
The Oxygen
The Top
The Bottom
11
Multiple Choice
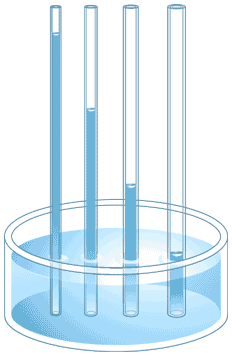
What is the term for water's ability to defy gravity and climb up a tube?
Capillary Action
Specific Heat
Universal Solvent
Magic
12
Multiple Choice
13
Multiple Choice

Hydrogen bond
weak attraction between two molecules (ex: water to water) that is formed due to positive and negative charges attracting
Strong attraction between two molecules.
Weak attraction between two molecules that is formed due to charges repelling.
Strong attraction due to repelling of charges.
14
Multiple Choice

Capillary Action
Tendency of a liquid in a tube to decrease
Tendency of a solid to dissolve
Tendency of a gas to condense
the tendency of a liquid in a tube to rise
15
Multiple Choice
Which of the following is the correct molecular structure for water, H2O?
16
Multiple Choice

Refer to the picture of water sticking to a leaf; this is occurring because of:
Adhesion
Cohesion
17
Multiple Choice
18
Multiple Choice
Water's ability to stick to glass is
adhesion
cohesion
surface tention
capillary action
19
Multiple Choice
20
Multiple Choice

As you add drops of water to the surface of a coin, each drop sticks to the other water droplet because of ________________, and sticks to the penny because of ______________.
adhesion, cohesion
cohesion, adhesion
capillary action, specific heat
specific heat, capillary action
Show answer
Auto Play
Slide 1 / 20
SLIDE
Similar Resources on Wayground

13 questions
The Five Senses
Presentation
•
7th Grade

13 questions
Mitosis
Presentation
•
7th Grade

18 questions
7.8B Weathering, Erosion, and Deposition
Presentation
•
7th Grade

15 questions
Chemical Compounds
Presentation
•
7th Grade

15 questions
Bacteria
Presentation
•
7th Grade

16 questions
Roman Numerals
Presentation
•
7th Grade

16 questions
Net Force
Presentation
•
7th Grade

17 questions
7th Science- Human Impacts (Water Edition)
Presentation
•
7th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

20 questions
genetics, punnett squares, heredity
Quiz
•
7th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

20 questions
Food Webs + Energy Pyramids
Quiz
•
7th Grade

10 questions
Exploring the Rock Cycle
Interactive video
•
6th - 8th Grade

10 questions
Exploring Earth's Seasons and Their Causes
Interactive video
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

19 questions
Natural and Artificial Selection
Quiz
•
7th Grade