

Stoichiometry Review
Presentation
•
Chemistry
•
10th Grade
•
Hard
James Gonzalez
FREE Resource
8 Slides • 24 Questions
1
Review of Stoichiometry
2
Stoichiometry – Using the mole ratio from the balanced equation and information about one compound in the reaction to determine information about another compound in the equation.
3

What do the coefficients really mean?
4

C3H8 + 7O2 → 6CO + 8H2O
5
C3H8 + 7O2 → 6CO + 8H2O
How many moles of Oxygen will react with 25.8 moles of Propane (C3H8) completely?

C3H8 + 7O2 → 6CO + 8H2O
6
Mole to Mole Ratio

C3H8 + 7O2 → 6CO + 8H2O
7
Limiting Reactant
vs.
Excess Reactants
8

9
Multiple Select
1 mole (mol) is equal to ...
6.02 x 10²³ atoms
Molar Mass of compound or element
22.4 Liters (L)
6.02 x 10²³ molecules
6.02 x 10²³ particles
10
Fill in the Blanks
11
Multiple Choice
When using a chemical equation to change one molecule into a different molecule, you use this.
Mole to Mole Conversion
T-Chart
Cross Multiplication
12
Multiple Choice

Mole to Mole Conversion Practice
2
3
4
6
13
Multiple Choice

How many moles of oxygen are necessary to react completely with four moles of propane?
5
4
12
15
20
14
Multiple Choice

How many grams are in 5.6 moles of sodium bromide (NaBr)?
Na = 23 g/mol
Br = 80 g/mol
576 g
756 g
15
Multiple Choice

16
Multiple Choice

17
Multiple Choice

How many moles are in 550 g of carbon dioxide?
C = 12 g/mol
O = 16 g/mol
18
Multiple Choice
Molar mass= ____ mol
1
2
19
Multiple Choice
is the volume of 1 mole of a gas at STP and is 22.4L/mol for any ideal gas
Molar volume
Molar Area
20
Multiple Choice
1 mole = 6.02 x 1023 representative particles
This is also known as ______
Avogadro’s number.
Beethoven's number.
21
Multiple Choice
S8 + 12 O2 → 8 SO3
How many moles of S8 is needed to produce 12 moles of SO3?
1
1.5
2
12
22
Multiple Choice
23
Multiple Choice
What is a Excess Reactant?
24
Multiple Choice
25
Multiple Choice
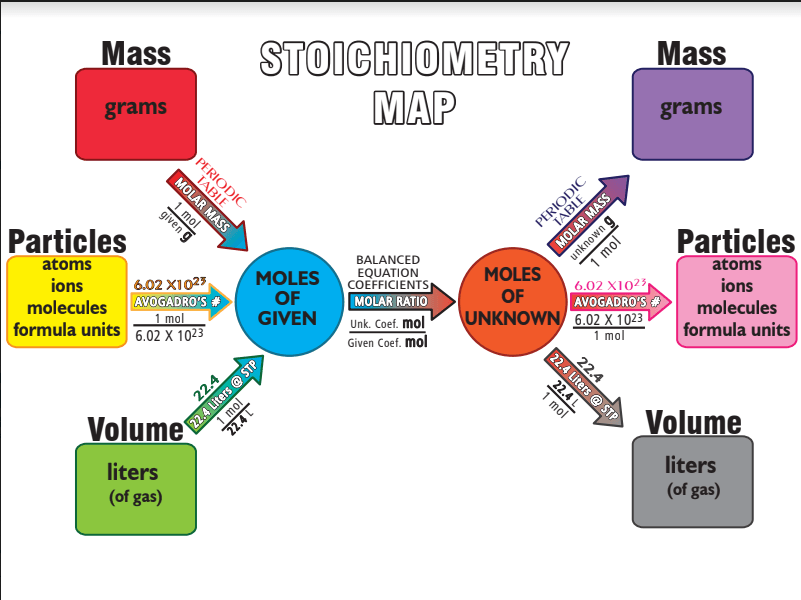
How many step will it take for me to go from grams to moles unknown
1
2
3
4
26
Multiple Choice
I use molar mass when the problem gives me grams?
False
True
27
Multiple Choice
I use mole ratio (coefficients) from balanced chemical equation?
True
False
28
Multiple Choice
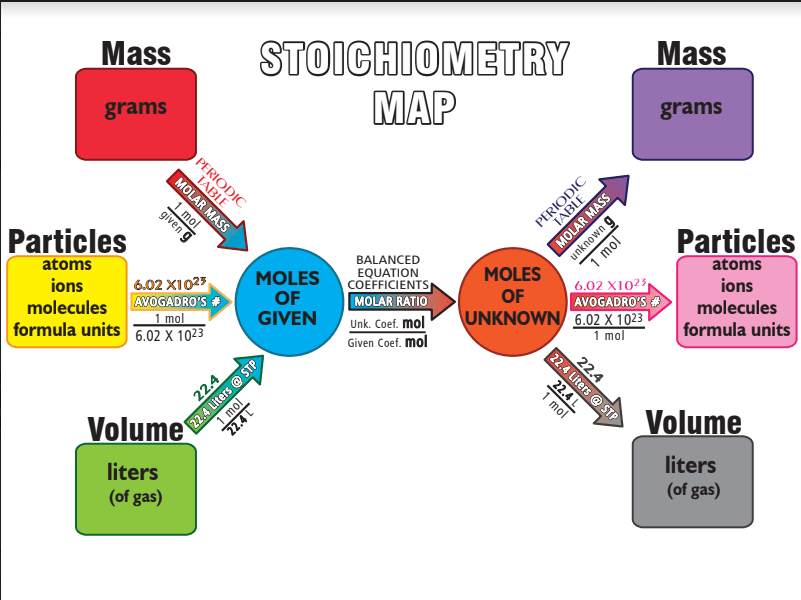
How many steps will it take me to go from grams to grams?
4
3
2
1
29
Multiple Choice

How many step will it take for me to go from moles given to moles unknown?
1
2
3
4
30
Multiple Choice
2 C2H6 + 7 O2 → 4 CO2 + 6 H2O
Ethane (C2H6) combusts in the above reaction. What is the mole ratio of ethane to oxygen gas?
31
Multiple Choice

In the equation: C2H4 + 3O2 --> 2CO2 + 2H2O
How many Liters of CO2 will be produced from 20g of C2H4 ?
31.4L
28L
36L
32L
32
Multiple Choice

What volume will 0.75 moles of Nitrogen gas occupy at STP?
15L
16L
16.8L
15.8L
Review of Stoichiometry
Show answer
Auto Play
Slide 1 / 32
SLIDE
Similar Resources on Wayground

25 questions
8.1 Stoichiometry Pt.3 Practice
Presentation
•
10th Grade

26 questions
11.1 Gases
Presentation
•
10th Grade

25 questions
Gas Laws
Presentation
•
10th Grade

26 questions
Dot Diagrams and Bonding Lesson
Presentation
•
9th Grade

21 questions
Balancing Chemical Equations
Presentation
•
9th Grade

27 questions
Introduction to Hydrocarbons
Presentation
•
10th - 11th Grade

26 questions
Introduction to Chemical Reactions
Presentation
•
9th Grade

28 questions
Covalent Bonding
Presentation
•
9th Grade
Popular Resources on Wayground

25 questions
The Ultimate College Knowledge Quiz
Quiz
•
8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Math Review
Quiz
•
6th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade