

Unit 5: States of Matter and Gas Laws
Presentation
•
Chemistry
•
8th Grade - University
•
Hard
+1
Standards-aligned
Kristina Bowden
FREE Resource
22 Slides • 13 Questions
1

2

3

4
Multiple Choice
Energy due in motion is ____________ energy.
5

6

7

8

9

10
Multiple Select
What phases were at point D in the last slide? (Pick 2)
Liquid
Solid
Gas
Plasma
11

12
Teacher note
You will not have to do any calculations on your CSA 5 for the gas laws. You need to be able to identify the relationships. Ex: What happens if the temperature increases?
13

14
Don't forget to label what types of Gas Law is occurring when taking your notes!
15

16

17
Multiple Choice
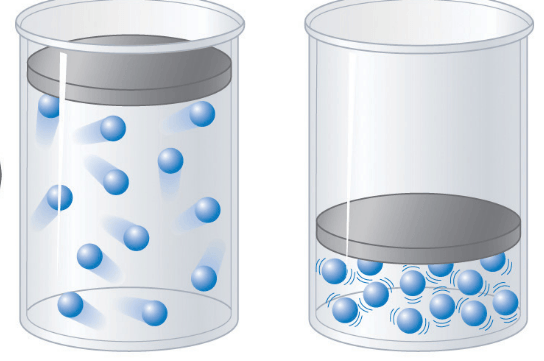
18
Multiple Choice
19

20

21

22

23

24

25

26

27
Multiple Choice
Boyles Law Tells That
The More The Volume The Less The Temperature
The More The Temperature The Less The Pressure
The More The Volume The More The Pressure
The More The Pressure The Less The Volume
28
Multiple Choice
Boyles Law Is A
Directly Proportional Relationship
Inversely Proportional Relationships
29
Multiple Choice
30
Multiple Choice
According to Charles Law, if Temperature of a gas increases Volume will also....
Increases
Decreases
Stays the same
31
Multiple Choice
According to Charles Law, the relationship between Temperature and Volume can best be described as....
Inverse Relationship
No Relationship
Direct Relationship
32
Multiple Choice
33
Multiple Choice

34
Multiple Choice
The burner of a hot air balloon start heating the air inside the envelope. This continues heating the air, until the balloon inflates all the way & starts to lift off the ground. What happens to the pressure of the balloon as it is being inflated?
Increases
Decreases
35
Multiple Choice
You accidentally leave a can of soda in the car on a hot summer day. What will happen to the volume of the carbon dioxide ( CO2 ) in the soda?
Increase
Decrease

Show answer
Auto Play
Slide 1 / 35
SLIDE
Similar Resources on Wayground

29 questions
Separation Techniques
Presentation
•
KG - University

27 questions
Bonding and Formulas
Presentation
•
9th - 12th Grade

26 questions
Monetary and Fiscal Policy Analysis
Presentation
•
8th - 12th Grade

26 questions
Apostrophes
Presentation
•
8th - 12th Grade

25 questions
Electron Configuration, Orbital Notation, & Dot diagrams
Presentation
•
9th - 12th Grade

26 questions
Stoichiometry Lesson
Presentation
•
9th - 12th Grade

27 questions
MS-ESS1-1: Earth, Sun, Moon Cycles
Presentation
•
8th Grade - University
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

15 questions
Acids and Bases Review
Quiz
•
8th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Counting Atoms Practice
Quiz
•
8th Grade