

Chemical Compounds
Presentation
•
Chemistry
•
10th - 12th Grade
•
Hard
Joseph Anderson
FREE Resource
17 Slides • 18 Questions
1
Naming Chemical Compounds
Nov. 12th - 22nd

2

3

4

5

6

7
Multiple Choice
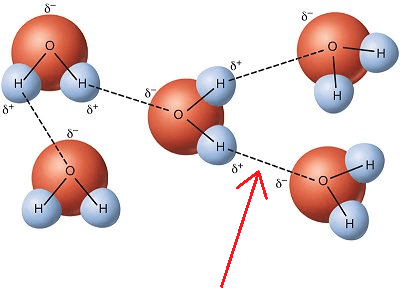
8
Multiple Choice
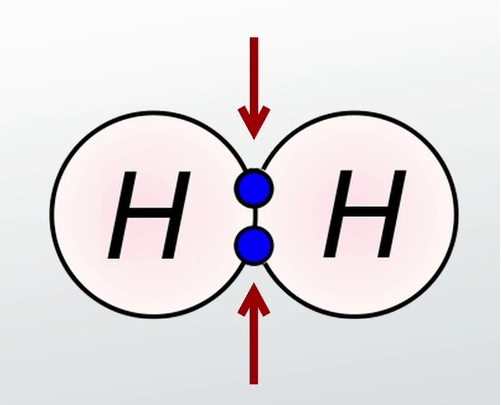
9

10
Multiple Choice
In metallic bonds electrons are:
shared
move around the nuclei randomly
transferred
11
Multiple Choice
Malleability and ductility are characteristics of compounds with
covalent bonds
metallic bonds
ionic bonds
alloy bonds
12
Multiple Choice
Which of the following lists of elements contain only metals?
Li, Al, Te
Ni, Pb, H
Na, Os, Ga
Ca, Zn, I
13

14

15
Multiple Choice
Why do all bonds form?
so the number of protons equals the number of electrons
so an atom can become unstable
to fill the outermost energy level
16
Multiple Choice
17
Multiple Choice
18
Multiple Choice
Valence electrons are found
in the innermost energy level of an atom
in the middle energy levels of an atom
in the outermost energy levels of an atom
19
Multiple Choice

The negative ions tend to _____________ electrons.
lose
gain
20
Multiple Choice

The positive ions tend to _____________ electrons.
lose
gain
21

22
Multiple Choice
Formula for barium sulfide
B2S3
BaS
Ba2S
BaS2
23

24
Multiple Choice
Formula for lead (II) chloride
PbCl2
Pb2Cl
Pb2Cl3
Pb3Cl2
25

26
Multiple Choice
27
Multiple Choice

28
Multiple Choice
29
Multiple Choice
Name for PBr2
phosphorus bromide
phosphorus dibromide
phosphorus dibromine
phosphorus bromine
30

31
Multiple Choice
32

33

34

35

Naming Chemical Compounds
Nov. 12th - 22nd

Show answer
Auto Play
Slide 1 / 35
SLIDE
Similar Resources on Wayground

28 questions
History and Organization of the Periodic Table
Presentation
•
10th - 12th Grade

28 questions
Ions
Presentation
•
10th - 12th Grade

25 questions
Electron Configuration, Orbital Notation, & Dot diagrams
Presentation
•
9th - 12th Grade

31 questions
Polarity and Intermolecular Forces
Presentation
•
11th - 12th Grade

27 questions
Bonding and Formulas
Presentation
•
9th - 12th Grade

28 questions
1.1 c & 1.2 a: Periodic Trends/Isotopes/Unstable Isotopes
Presentation
•
10th - 12th Grade

28 questions
Chemistry I Final Exam Review
Presentation
•
10th - 12th Grade

30 questions
Atoms & The Periodic Table
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Unit 7 Chemical Reactions
Quiz
•
10th Grade

25 questions
Unit 8 Stoichiometry Review
Quiz
•
10th Grade

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

15 questions
Nuclear Chemistry: Balancing
Quiz
•
10th Grade

20 questions
Naming Acids and Bases
Quiz
•
10th Grade

10 questions
Exploring Stoichiometry in Chemistry
Interactive video
•
6th - 10th Grade