

Intermolecular Forces and Polarity
Presentation
•
Chemistry
•
10th Grade
•
Hard
Joseph Anderson
FREE Resource
9 Slides • 8 Questions
1
Polarity and Intermolecular Forces

2
Polar vs Nonpolar
A polar molecule is sharing electrons unequally (two different poles, one positive and the other negative)
A nonpolar is sharing electrons equally (tug of war with two trucks pulling with the exact same force)

3
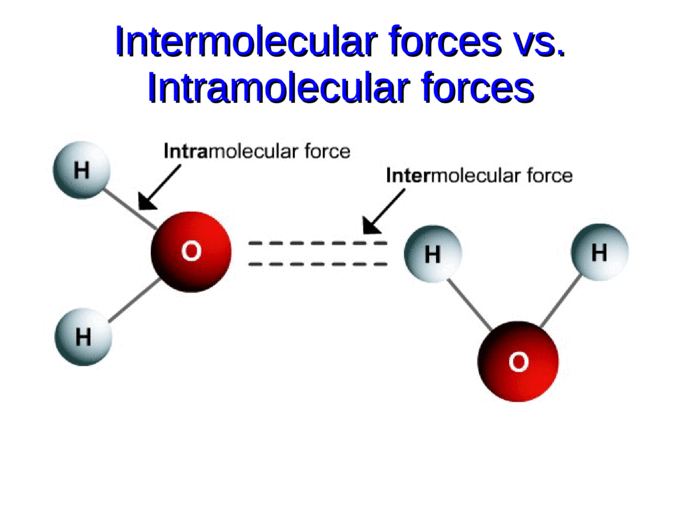
INTRAmolecular VS INTERmolecular Forces
INTRAmolecular forces are the bonds that occur inside or within a single moelcule.
INTERmolecular forces (IMF) exist between two or more molecules
4
Multiple Choice

Intermolecular Forces are the forces that exist
Between two or more molecules
Within a single molecule
Only in molecules containing carbon
In all molecules
5
INTERmolecular Forces
Examples of intermolecular forces include london dispersion (LDF), dipole/dipole, and hydrogen bonding

6

7
Multiple Choice
Examples of an intermolecular force include
london disperson
hydrogen bonding
dipole dipole
all of the above
8
Multiple Choice
Which type of intermolecular force is the strongest?
LDF
dipole-dipole
Hydrogen bonding
Covalent bonding
9
London Dispersion Force (LDF)
The weakest force
Occurs between ALL MOLECULES
If a molecule is nonpolar then the only force present will be LDF

10
Dipole Dipole Forces
Occurs between all polar molecules
The attraction of the positive end of one molecule to the negative end of another

11
Hydrogen Bonding
Must have a hydrogen directly bonded to a F, N or O.
Strongest type of intermolecular force

12
Multiple Choice
For hydrogen bonding to occur, a molecule must have a hydrogen bonded to
carbon
another hydrogen
Fluorine, Chlorine or Oxygen
Fluorine, Nitrogen or Oxygen
13
Polar and Nonpolar
Nonpolar molecules will contain only london dispersion forces
Polar molecules will contain london dispersion and dipole dipole
Molecules with hydrogen bonding will contain all three types of forces

14
Multiple Choice
Which of the following is an example of a polar molecule
water
propane
methane
carbon dioxide
15
Multiple Choice
Is methane (CH4) polar or nonpolar?
polar
nonpolar
16
Multiple Choice
What forces are present in methane (CH4)?
LDF
dipol-dipole
Hydrogen bonding
All of the above
17
Multiple Choice

Does this molecule contain hydrogen bonding?
yes
no
Polarity and Intermolecular Forces

Show answer
Auto Play
Slide 1 / 17
SLIDE
Similar Resources on Wayground

11 questions
Crude Oil and Hydrocarbons
Presentation
•
10th Grade

13 questions
Water & Life Review
Presentation
•
10th Grade

15 questions
Metals and Non-Metals
Presentation
•
10th Grade

14 questions
DNA!
Presentation
•
9th Grade

16 questions
periodic patterns of melting points, electrical conductivity, IE
Presentation
•
10th Grade

16 questions
stoichiometry
Presentation
•
10th Grade

13 questions
counting atoms
Presentation
•
10th Grade

11 questions
KMT & Phase Changes Recap
Presentation
•
10th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

10 questions
Exploring Properties and Models of Acids and Bases
Interactive video
•
6th - 10th Grade

19 questions
Review Set 3 - Moles & Chemical Formulas
Presentation
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Stoichiometry Practice
Quiz
•
10th Grade

10 questions
Identifying Types of Chemical Reactions
Interactive video
•
6th - 10th Grade

20 questions
Redox Reactions
Quiz
•
10th - 12th Grade

20 questions
pH and pOH Calculations
Quiz
•
10th - 11th Grade