

Atomics
Presentation
•
Chemistry
•
8th - 12th Grade
•
Hard
Joseph Anderson
FREE Resource
10 Slides • 8 Questions
1
Quizizz Lesson
Atomic Structure and the Periodic Table

You will need your class notes and your periodic table.
2
The Periodic Table is organized by increasing atomic number.
Atomic Number = Number of Protons
There are 7 rows
There are 18 groups.
Each group has the same number of valence electrons

3
Poll
How many Groups are in the Periodic Table?
2
7
8
18
4
Poll
What is the maximum number of energy levels in an atom?
We have no way to know
up to 7
Each row adds another energy level
up to 18
5
Models: An atomic image focusing on electrons
The nucleus is a single ball in the center
The large gold spheres represent electrons
The rings represent energy levels

6
Multiple Choice

What is the atomic number for this atom?
2
8
7
5
7
Open Ended

What is this element?
8
Model of a Nitrogen Atom
How do you know?
7 total electrons = 7 protons = atomic number 7
OR
5 valence electrons - Group 15
2 rings - second row of the Periodic Table

9
Can you identify these two atoms?

Write your guess on a piece of scrap paper. Then check it on the next slide.
One Valence Electron (Group 1)
Two Rings so second row of periodic table
One Valence Electron (Group 1)
Three Rings so third row of periodic table
10

First atom
Group 1, Second Row = Lithium
It also has 3 total electrons so Atomic Number of 3 = Lithium
Second atom
Group 1, Third Row = Sodium
It also has 11 total electrons so Atomic Number of 11 = Sodium
11
What are these elements? Answer on scrap paper, then check on the next slide.
Top: Valence Electrons = ______, Rings = 2
Element =
Lower: Valence Electrons = ____ Rings = 3
Element =

12
What are these elements?

Top: Valence Electrons = 7,
Rings = 2
ELEMENT = Fluorine
Lower: Valence Electrons = 7
Rings = 3
ELEMENT = Chlorine
13
Nobel Gases have full outer energy levels
The Octet Rule
The first energy level can hold only 2 electrons
All others want 8 to be stable (happy)
An atom want either a full or an empty outer energy level

14
Multiple Choice

TRUE OR FALSE Lithium and Beryllium have the same number of valence electron.
TRUE
FALSE
15
Multiple Choice
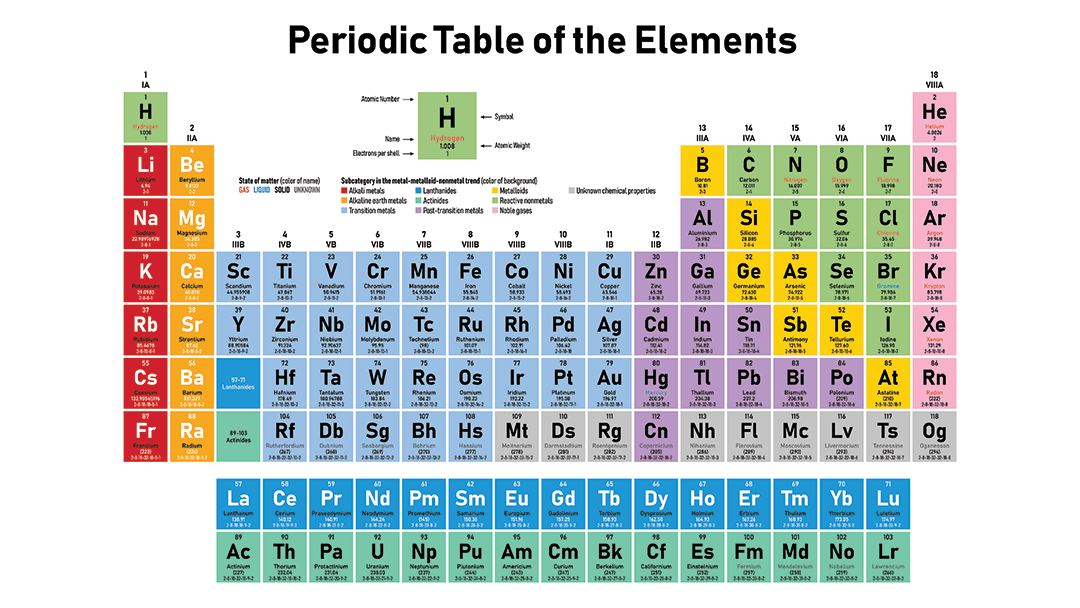
How many valence electrons does Na have?
1
5
6
11
16
Multiple Choice
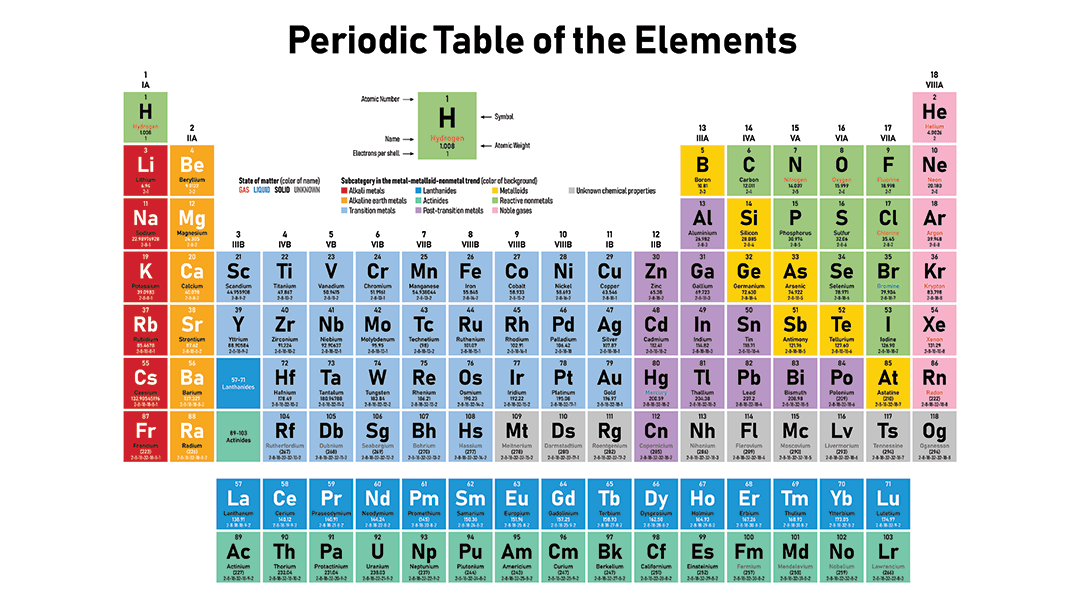
Chlorine ( Cl) has _________ valence electrons.
3
5
7
8
17

Atoms form bonds to be stable. These are now compounds.
18
Multiple Choice
Valence electrons play a key role in chemical reactions their number increases from left to right across a row of the periodic table.
TRUE
FALSE
Quizizz Lesson
Atomic Structure and the Periodic Table

You will need your class notes and your periodic table.
Show answer
Auto Play
Slide 1 / 18
SLIDE
Similar Resources on Wayground

13 questions
Radioactivity Review
Presentation
•
9th - 12th Grade

13 questions
Alpha Decay
Presentation
•
9th - 12th Grade

12 questions
Mole to Gram and Gram to Gram Conversions
Presentation
•
9th - 12th Grade

11 questions
Balancing, Conservation and Reaction Types Review
Presentation
•
9th - 12th Grade

18 questions
Quizizz Lesson: Atomic Structure and the Periodic Table
Presentation
•
8th - 12th Grade

17 questions
Balancing Equations and Predicting Products Guided Practice
Presentation
•
9th - 12th Grade

13 questions
Nobel Gas Configuration w/some Lewis Dot
Presentation
•
9th - 12th Grade

12 questions
EXERp1STRUCTud
Presentation
•
KG
Popular Resources on Wayground

20 questions
"What is the question asking??" Grades 3-5
Quiz
•
1st - 5th Grade

20 questions
“What is the question asking??” Grades 6-8
Quiz
•
6th - 8th Grade

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

34 questions
STAAR Review 6th - 8th grade Reading Part 1
Quiz
•
6th - 8th Grade

20 questions
“What is the question asking??” English I-II
Quiz
•
9th - 12th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

47 questions
8th Grade Reading STAAR Ultimate Review!
Quiz
•
8th Grade
Discover more resources for Chemistry

15 questions
Balancing and Reaction Types
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

16 questions
STAAR Review Quizziz 5
Quiz
•
10th Grade

20 questions
Counting Atoms
Quiz
•
8th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

5 questions
DOL REC: pH and pOH Calculations
Quiz
•
10th - 11th Grade

11 questions
Balancing Chemical Equations
Presentation
•
9th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade