

Chemical Nomenclature Lesson
Presentation
•
Science
•
9th Grade - University
•
Hard
Joseph Anderson
FREE Resource
32 Slides • 6 Questions
1
Organic Chemistry Introduction
Module 7 - Year 12
2
What we will cover:
What is organic chemistry?
Syllabus inquiry questions
IUPAC nomenclature
Alkanes
Alkenes
Alkynes
Isomerism
3
Open Ended
What is your understanding of organic chemistry?
4
What is organic chemistry?
Life is carbon based
all Earthly life
pharmaceuticals, petrochemicals, agrichemicals, lubricants, solvents, plastics, fuels, explosives
Synthesising compounds or creating new ones

5
What we will learn in this module:






6

7
IUPAC Nomenclature
Naming is essential and specific
allows us to name incredibly complex chemical structures and every chemist around the world knows what we are talking about



8
generally it is based on the:
functional groups off this chain
number of carbons in the chain
the position of the functional groups
alphabetical order
different ways to represent:
molecular
structural
condensed structural
skeletal structural

9
Types of structures



Structural
10

11
IUPAC name or preferred name?
2-hydroxypropane - 1, 2, 3-tricarboxylic acid
Citric acid

12
Prefixes
We count the longest carbon chain, and this forms the basis of the organic compounds name
You need to remember these!

13
Hydrocarbons
Alkanes, alkenes alkynes

14
Multiple Choice
Choose the alkane
15
Alkanes
contain only C and H atoms (hydrocarbon)
contain single bonds
saturated molecule - cannot bond with anything else
has the general formula: CnH2n+2
Naming alkanes:
identify the longest continuous carbon chain
identify any alkyl side chains
number the carbon atoms in the chain (to give lowest numbers to side chains)
16
Example:
Count the number of carbons in the longest chain
Use the prefix and attach to
-ane

17
Alkyl side chains:
Branches off of the main carbon chain
suffix is -yl
Helps us to number the larger structure
Must have the lowest number possible attached
alphabetical order
3-ethyl-2-methylhexane

18
Your turn:

19
Your turn:

20
Multiple Choice
21
Multiple Choice
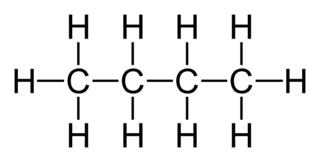
22
Multiple Choice

23
Multiple Choice
Which is the structures with IUPAC nomenclature
2,2,4-trimethylpentane
24
Alkenes
have a double bond between at least two carbons
unsaturated
general formula: CnH2n
Naming:
same as alkanes
replace -ane with -ene
place number before -ene to indicate where the double bond is

25
Your turn:

26

27
Your turn:

28

29
Alkynes
contains at least one triple bond between 2 carbons
uses the suffix -yne
unsaturated
general formula: CnH2n-2
naming:
same as alkanes
replace -ane with -yne
place number before -yne to indicate where the triple bond is

prop-1-yne
30
Your turn:

31

32
Your turn
Draw: 2,2-dimethyloct-3-yne
33
Your turn
Draw: 2,2-dimethyloct-3-yne

34
Isomers
molecules that have the same number and type of atoms, but have a different structure
can have very different physical and chemical properties
3 types of isomers:
Chain = have differing numbers of alkyl groups
Position = change in position of bonds or functional groups
Functional group = same atoms, but different functional groups (we will learn more about this later)

35
Chain Isomers

36
Position Isomers


37
Functional Isomers

38
What we will cover next:
Functional groups
haloalkanes, alcohols, amines, aldehydes, ketones, carboxylic acids, amides, esters
Organic Chemistry Introduction
Module 7 - Year 12
Show answer
Auto Play
Slide 1 / 38
SLIDE
Similar Resources on Wayground

33 questions
CCU4 Lewis
Presentation
•
9th Grade - University

33 questions
Heat Transfer Notes
Presentation
•
9th Grade - University

33 questions
Energy & Energy Transformations
Presentation
•
9th - 12th Grade

33 questions
Population Ecology Review
Presentation
•
9th - 12th Grade

31 questions
Genetic Technologies
Presentation
•
8th Grade - University

30 questions
Evidence of Evolution
Presentation
•
9th - 12th Grade

30 questions
Eukaryotic Cell Structure
Presentation
•
9th - 12th Grade

36 questions
Plant Hormones and Responses (Tropisms)
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

40 questions
NCFE Earth and Environmental Science Released Test
Quiz
•
9th - 12th Grade

10 questions
Exploring Weather Influences and Map Reading
Interactive video
•
6th - 10th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

61 questions
End of the Year Trivia Fun
Quiz
•
9th Grade

15 questions
Protein synthesis
Quiz
•
9th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade