

Chemistry Semester Review
Presentation
•
Chemistry
•
9th Grade
•
Hard
Joseph Anderson
FREE Resource
10 Slides • 41 Questions
1
Semester Exam Review
Pre AP Chemistry 2022-2023
December 5, 2022
The Chemistry Semester Exam is TOMORROW, TUESDAY! Try to review as much as you can :)
2
Periodic Table trends

3
How to calculate molar mass?
Subject | Subject
Some text here about the topic of discussion

1.Identify the elements
2. Count how many atoms of each element.
3.Find the atomic mass in the periodic table.
4.Multiply it and then add each mass.
4
Multiple Choice

What is the molar mass of NF3?
33.01 g/mol
61.03 g/mol
71.01 g/mol
99.03 g/mol
5
Percent Composition


You need to remember the formula and steps to calculate percent composition in the next slide question.
6
Match
The percent composition of Mg and O in MgO? Remember you need to first calculate the molar mass of MgO. Then use the formula.
Mg
O
Molar Mass of MgO
60%
40%
40.30 gr/mol
60%
40%
40.30 gr/mol
7
How to calculate Formal Charge?
Some text here about the topic of discussion

You need to remember the formula for formal charge to answer the next question.
8
Match

Calculate the formal charge of each atom represented in the molecule in the picture. (Look for the Formal charge formula from previous slide.)
H
N
C (between hydrogens)
C (between O's)
O (with double bond)
0
+1
0
0
0
0
+1
0
0
0
9
VSEPR Formula

10
Multiple Choice

The VSEPR Formula that best represents this molecule...3
AB3E
AB2E
AB2E3
A3B2E
11
Molecular & Electron Geometry
In order for you to determine the electron or molecular geometry of a molecule. You will have access to the table. It is very helpful to be able to know the ABE (VSEPR formula) of the molecule to correctly identify the type of shape.

12
Fill in the Blanks

Type answer...
13
Fill in the Blanks

Type answer...
14
Structural Isomers
Isomers in which atoms are completely arranged in a different order with the same molecular formula.
Same formula, but different order of atoms.

15
Multiple Choice
Which formulas represent compounds that are isomers of each other?
16
Multiple Choice

Yes
No
17
Polarity of molecules
Covalent bonds are divided into polar and non polar bonds.
Polar bonds
Asymmetrical
The branches might not match (atoms connected to central atom are not all the same)
There will be lone pairs (dots) on the central atom
Nonpolar bonds
Symmetrical
The branches will match (atoms connected to central atom are the same)
There will be lone pairs (dots) on the central atom
There will be no lone pairs on central atom.



18
Multiple Choice
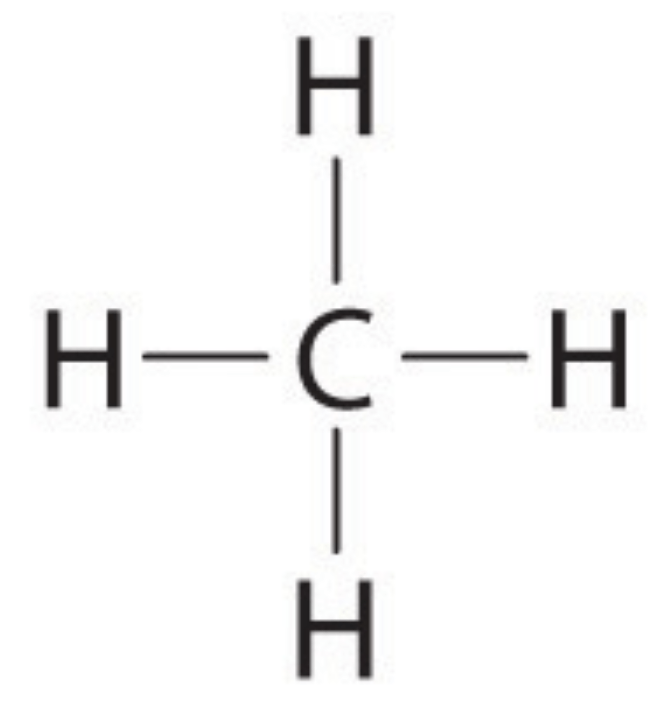
Classify the following molecule.
polar
nonpolar
19
Multiple Choice
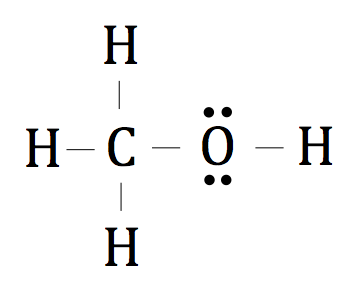
Is the following molecule polar or nonpolar?
polar
nonpolar
20
Multiple Choice

Is this molecule polar or non-polar?
Polar
Non-polar
21
Multiple Choice

Is this molecule polar or non-polar?
Non-polar
Polar
22
Dimensional analysis

Write and analyze the example problem below to answer the problem in the next slide. Remember that the units are the most important to set up correctly to get the correct result.
23
Multiple Choice
A 270.27 g sample of glucose C6H12O6 is measured in the laboratory. How many molecules of glucose are in
the sample? Assume molar mass of glucose is 180.18 g/mol. If you want to get this right in your exam, please make sure you get a piece of paper and try to work out the problem. 1023
6.02 x 1023 molecules of C6H12O6
9.03 x 1023 molecules of C6H12O6
1.20 x 1024 molecules of C6H12O6
4.01 x 1023 molecules of C6H12O6
24
Multiple Choice
An Ionic Compound is formed between what two types of elements?
Metal and Metal
Metal and Nonmetal
Nonmetal and Nonmetal
Metal and Metalloid
25
Multiple Choice
In an Ionic Bond Valence Electrons are what?
created
destroyed
gained or lost = transferred
shared
26
Multiple Choice
Compounds held together with these bonds are malleable with high thermal and electrical conductivity due to freely moving electrons.
Ionic Bond
Metallic Bond
27
Multiple Choice
Occurs when cations are fixed within a sea of shared delocalized electrons...
Ionic Bond
Metallic Bond
28
Multiple Choice
Form a crystal lattice based on electrostatic attraction and repulsion in repeating patterns...
Ionic Bond
Metallic Bond
29
Multiple Choice
K1+ S2-
KS
K8S
K6S3
K2S
30
Multiple Choice
Identify the following compound as ionic or covalent: SO2
ionic
covalent
31
Multiple Choice
Which Lewis Dot Structure is correct for the particular element?
32
Multiple Choice
What is the correct Lewis Dot Structure for ammonia NH3
33
Multiple Choice

Classify the following molecule.
polar
nonpolar
34
Multiple Choice

Covalent
Metallic
Ionic
Oxygen
35
Multiple Choice
A bond between a nonmetal and a nonmetal is called a(n)
covalent bond
ionic bond
metallic bond
transfer bond
36
Multiple Choice
Identify the following compound as metallic, ionic or covalent: SO2
ionic
covalent
metallic
37
Multiple Choice
Cations
Gain electrons, has an overall positive charge
Lose electrons, has an overall positive charge
Lose electrons, has an overall negative charge
Gain electrons, has an overall negative charge
38
Multiple Choice
How many Aluminum atoms are in Al2O3?
3
5
2
1
39
Multiple Choice
An ion with a negative charge is called...
Anion
Cation
40
Multiple Choice
Na1+ F1-
NaF
NaF3
Na3F
Na1F2
41
Multiple Choice
AlCl3
Al3Cl
AlCl
Al3Cl3
42
Multiple Choice
you add more and more neutrons
you add more and more protons
you add more and more shells (energy levels)
you add more atomic mass
43
Multiple Choice
Francium (Fr) has the lowest ionization energy in Group 1 because -
it has the smallest number of valence electrons
it has the greatest atomic mass
it has the greatest number of protons, so it attracts its electrons the strongest
its 1 valence electron is very far from the nucleus with a lot of shielding from the nucleus, so little energy is needed to remove it
44
Multiple Choice
Helium (He)
Titanium (Ti)
Calcium (Ca)
Magnesium (Mg)
45
Multiple Choice
the ability of an atom to attract/ accept electrons
the ability of an atom to lose electrons
the energy required to remove an electron from a specific atom
how easy it is to make friends.
46
Multiple Choice
the atoms have more mass.
the atoms have less mass
the atoms have more protons.
the atoms have less electrons.
47
Multiple Choice
Protons
Neutrons
Electrons
Valence Electrons
48
Multiple Choice

The ionization energy drops significantly as you move down a group because the valence electrons are in a shell further from the nucleus.
The ionization energy increases gradually as you move right across a period because the valence electrons are in a shell further from the nucleus.
The ionization energy increases gradually as you move right across a period because you are adding more protons.
The ionization energy increases when the valence electrons are more attracted to the nucleus.
49
Multiple Choice
the energy required to add an electron to a specific atom
how much energy it takes to remove an electron from an atom
the energy required to shield the outer electrons from the nucleus
a measure of the ability of an atom to attract electrons
50
Multiple Choice
Ionization energy __________ down a column and ________ to the right.
decreases, increases
increases, decreases
increases, increases
decreases, decreases
51
Multiple Choice

Which atoms has the HIGHEST first ionization energy?
He
Ar
Xe
Rn
Semester Exam Review
Pre AP Chemistry 2022-2023
December 5, 2022
The Chemistry Semester Exam is TOMORROW, TUESDAY! Try to review as much as you can :)
Show answer
Auto Play
Slide 1 / 51
SLIDE
Similar Resources on Wayground

45 questions
Mitosis & Meiosis Review
Presentation
•
9th - 10th Grade

46 questions
Motion-Distance, Displacement, Average Speed
Presentation
•
9th Grade

47 questions
Realidades 4B Test
Presentation
•
9th Grade

50 questions
C3.1 Chemical Formulae and Balancing Equations
Presentation
•
8th - 9th Grade

44 questions
Major Cell Organelles-Eukaryotic Cells
Presentation
•
10th Grade

44 questions
8/25 - Human Impact on Ocean Systems
Presentation
•
8th Grade

44 questions
Intro to Chemical Concepts Bellwork Review
Presentation
•
9th Grade

43 questions
Cell Transport
Presentation
•
9th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

10 questions
Exploring Stoichiometry in Chemistry
Interactive video
•
6th - 10th Grade

5 questions
DOL PreAP: Intro to Nuclear Chemistry
Quiz
•
9th - 10th Grade

12 questions
IP PreAP: Intro to Nuclear Chemistry
Quiz
•
9th - 10th Grade

17 questions
Types of Chemical Reaction
Presentation
•
7th - 12th Grade

120 questions
Chemistry Final Review
Quiz
•
9th - 12th Grade

15 questions
Half-Life
Quiz
•
9th - 12th Grade

15 questions
Unit 10 Review
Quiz
•
9th - 12th Grade