

Density Slides
Presentation
•
Science
•
9th - 12th Grade
•
Hard
Joseph Anderson
FREE Resource
8 Slides • 53 Questions
1

2
Density Definition
the degree of compactness of a substance

3

We need MASS- the amount of MATTER in a given object
We need VOLUME- the amount of SPACE something takes up
Calculating Density
4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
7
Multiple Choice
8
Multiple Choice
9
Multiple Choice
10
How do I calculate mass?
Pretty easy, you weight it on a scale
There are only 2 units we use for mass....
g(grams) and kG (kilograms)
11
You find the "displacement" Put the object in a cylinder, and measure the before and after units, and the difference in the volume. in mL
An irregular object
You measure Length x Width x Height measured in cm^3
A cube or normal object
How do I calculate volume?
12
Multiple Choice

13
Multiple Choice

14
Multiple Choice

Let's practice! Before reads 50 mL. After we drop in the object, the new water level goes up to 60 mL. What is the volume of the blocks? (Hint: new volume- old volume)
8 mL
10 mL
60 mL
5 mL
15
Multiple Choice

What is the volume of the two red spheres? (Hint: new volume - old volume)
4 mL
6 mL
2 mL
8 mL
16
Multiple Choice

What is the volume of the ring? (new volume - old volume)
64 mL
68 mL
8 mL
4 mL
17
Ok, got it. So, how do I calculate density?
D=M/V or...M= DxV or...V= M/V
Remember, units for
volume cm^3 OR mL
Mass g or kG
Density g/cm^3 or mL/g.......
18
Multiple Choice

19
Multiple Choice

20
Multiple Choice

21
Multiple Choice

22
Multiple Choice

What is the mass of a 25 cm3 liquid if it has a density of 5 g/cm3?
125 g
5 g
cm35g
0.2 cm3
23
Multiple Choice

24
Keys to remember
The density of water is 1.00 g/mL
If anything is higher than 1.00 g/mL, it will SINK
If anything is lower than 1.00 g/mL, it will float.
If it is "equal" to 1.00 g/ml, it will float/"flink"
25
Multiple Choice

26
Multiple Choice
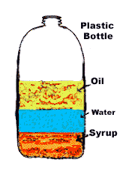
27
Let's review to make sure we got it!
28
Multiple Choice
29
Multiple Choice
30
Multiple Choice

31
Multiple Choice
32
Multiple Choice
33
Multiple Choice
34
Multiple Choice
35
Multiple Choice
What is the formula for Volume?
Mass/Volume
Density x Volume
Mass/Density
36
Multiple Choice
What is the formula for Mass?
Mass/Volume
Density x Volume
Mass/Density
37
Multiple Choice

38
Multiple Choice
39
Multiple Choice
40
Multiple Choice

41
Multiple Choice
42
Multiple Choice
43
Multiple Choice
A crayon has a mass of 10.0 g and volume of 5.0 mL. What is the density?
30 g/mL
15 g/mL
1.0 g/mL
2.o g/mL
44
Multiple Choice
A piece of copper has a mass of 89g and a volume of 10.0 cm3. What would be the density of the copper?
0.89 g/cm3
89 g/cm3
8.9 g/cm3
890 g/cm3
45
Multiple Choice
What is the volume of 150.0 grams of lead if it has a density of 11.3 g/cm3?
13.3 g
13.3 cm3
.075 g
1695 cm3
46
Multiple Choice
47
Multiple Choice
Calculate the density in the following question: A star has a mass of 15 g and a volume of 25 mL.
0.7 g/mL
0.6 g/mL
0.5 g/mL
0.4 g/mL
48
Multiple Choice
In a density column, is the substance with the highest density on the bottom or the top?
Bottom
Top
49
Multiple Choice
In a density column, is the substance with the lowest density on the bottom or on the top?
Bottom
Top
50
Multiple Choice
Which of the following is the correct definition of density?
The amount of stuff in an object
The amount of space an object takes up
A measure of the force of gravity on an object
The amount of matter (mass) packed into a space (object)
51
Multiple Choice

Which object has the highest density in this picture?
A
B
C
52
Multiple Choice

Which object has the lowest density in this picture?
A
B
C
53
Multiple Choice
54
Multiple Choice
M =
matter
Massachusetts
math
mass
55
Multiple Choice
V =
velocity
volume
vertex
vacuole
56
Multiple Choice
D =
diffusion
decay
decible
density
57
Multiple Choice
A bar of copper has a mass of 216g and a volume of 24 cm3. What is the density of copper?
9g/cm3
5184g/cm3
.11g/cm3
322mL
58
Multiple Choice
59
Multiple Choice
60
Multiple Choice
61
Multiple Choice

Using the picture, tell me which object is the least dense and why?
The fish because he is in the middle
The cork because it is floating at the top of the water
The rock because it is at the bottom
The cork and the rock because they have the same about of atoms.

Show answer
Auto Play
Slide 1 / 61
SLIDE
Similar Resources on Wayground

52 questions
Biodiversity Basics
Presentation
•
9th - 12th Grade

57 questions
Perimeter of Polygons & Circumference of Circles
Presentation
•
9th - 12th Grade

57 questions
Lesson 1: 1920s- A Return to Normalcy
Presentation
•
9th - 12th Grade

57 questions
Ecology part 1 review 2025 fall semester final
Presentation
•
9th - 12th Grade

58 questions
Speed, Velocity, and Acceleration
Presentation
•
9th - 12th Grade

53 questions
DNA Replication and Cell Cycle
Presentation
•
9th - 12th Grade

52 questions
Biodiversity Easy
Presentation
•
9th - 12th Grade

52 questions
Biodiversity Animals
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

40 questions
NCFE Earth and Environmental Science Released Test
Quiz
•
9th - 12th Grade

10 questions
Exploring Weather Influences and Map Reading
Interactive video
•
6th - 10th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

61 questions
End of the Year Trivia Fun
Quiz
•
9th Grade

15 questions
Protein synthesis
Quiz
•
9th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade