

Gas Chemistry
Presentation
•
Chemistry
•
University
•
Hard
Joseph Anderson
FREE Resource
13 Slides • 12 Questions
1
Chapter 11- Gases
Presenter: Dr Luis Bello

2

3
Multiple Choice
4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
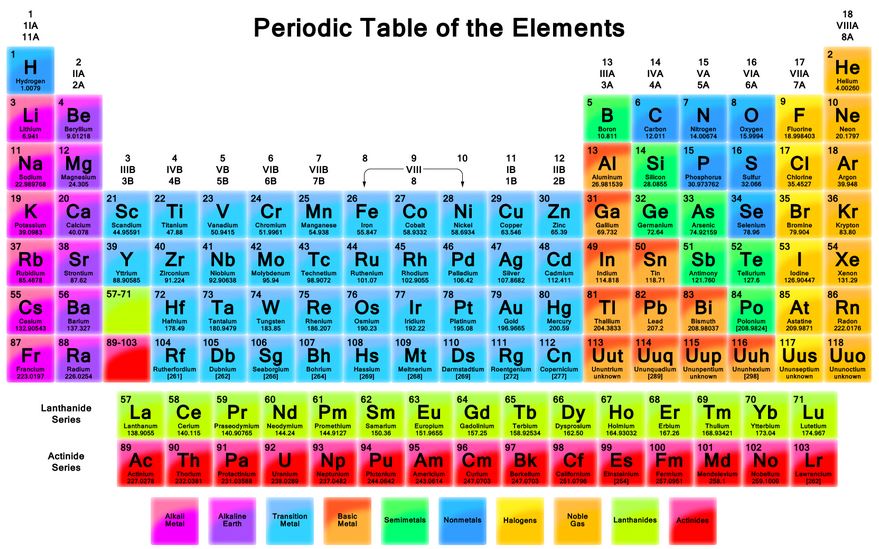
Elements on the LEFT side of the periodic table will most likely form:
Positive ions
Negative ions
Neutral Ions
None of these
7
Multiple Choice
The theory that is used to predict the arrangement of atoms about a central atom is called?
Valence shell repulsion
Valence shell electron sharing
Valence shell electron pair repulsion
Valence electron shell repulsion pair
8
Multiple Choice

9
Multiple Choice

10
Multiple Choice
11

12

13
14
Kinetic-molecular theory:
Gases consist of large numbers of molecules that are in continuous, random motion
The volume of all the molecules of the gas is negligible compared to the total volume in which the gas is contained
Attractive and repulsive forces between gas molecules are negligible
Energy can be transferred between molecules during collisions, but the average kinetic energy of the molecules does not change with time, as long as the temperature of the gas remains constant
The average kinetic energy of the molecules is proportional to absolute temperature. At any given temperature, the molecules of all gases have the same average kinetic energy
15
16
17

18

19

20
21
Multiple Choice
A student inflates a balloon with helium then places it in the freezer. The student should expect
the balloon's volume to increase
the balloon's volume to decrease
the balloon's moles to increase
the balloon's moles to decrease
22
Multiple Choice
23
Multiple Choice
24
Multiple Choice
25
Chapter 11- Gases
Presenter: Dr Luis Bello

Show answer
Auto Play
Slide 1 / 25
SLIDE
Similar Resources on Wayground

21 questions
ATIN2_PART4_S26
Presentation
•
University

21 questions
IN ON AT prepositions of place
Presentation
•
University

20 questions
alkane
Presentation
•
KG - University

21 questions
TCRM Lesson 10/26
Presentation
•
KG - University

21 questions
Whole numbers GCSE AQA Maths
Presentation
•
University

18 questions
going to
Presentation
•
University - Professi...

20 questions
Topic 9: Reproduction & Development Part 2
Presentation
•
University

14 questions
Electron Math
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
"What is the question asking??" Grades 3-5
Quiz
•
1st - 5th Grade

20 questions
“What is the question asking??” Grades 6-8
Quiz
•
6th - 8th Grade

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

34 questions
STAAR Review 6th - 8th grade Reading Part 1
Quiz
•
6th - 8th Grade

20 questions
“What is the question asking??” English I-II
Quiz
•
9th - 12th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

47 questions
8th Grade Reading STAAR Ultimate Review!
Quiz
•
8th Grade
Discover more resources for Chemistry

15 questions
LGBTQ Trivia
Quiz
•
University

36 questions
8th Grade US History STAAR Review
Quiz
•
KG - University

25 questions
5th Grade Science STAAR Review
Quiz
•
KG - University

16 questions
Parallel, Perpendicular, and Intersecting Lines
Quiz
•
KG - Professional Dev...

20 questions
5_Review_TEACHER
Quiz
•
University

10 questions
Applications of Quadratic Functions
Quiz
•
10th Grade - University

10 questions
Add & Subtract Mixed Numbers with Like Denominators
Quiz
•
KG - University

20 questions
Block Buster Movies
Quiz
•
10th Grade - Professi...