

Mass Stoichiometry Solutions
Presentation
•
Mathematics
•
10th Grade
•
Hard
Joseph Anderson
FREE Resource
14 Slides • 6 Questions
1
Stoichiometry
the relationship between substances that are included in reactions and solutions. This includes conversions of different measurements in order to show and give accurate proportions in reactions.

2
All about converting
Think of stoichiometry as converting feet to inches or minutes to hours and vice versa. For example, if you have 90ft you know that there are 12inches in one foot; therefore you should have 1080inches in total. The same goes for stoichiometry; there are several conversions that we have and use in order to calculate moles (mols) , molecules, atoms, formula units (fu) , and mass (g).
3
Conversions
Molecules/particles/molecules/formula units -> moles
Grams -> moles
Mole to mole ratio
4
Avogrado's Number (6.022 x 1023)
a constant that is used in stoichiometry problems when atoms or molecules are involved
5
Molecules/atoms/formula units/particles -> moles
Involves Avogrado's number
6
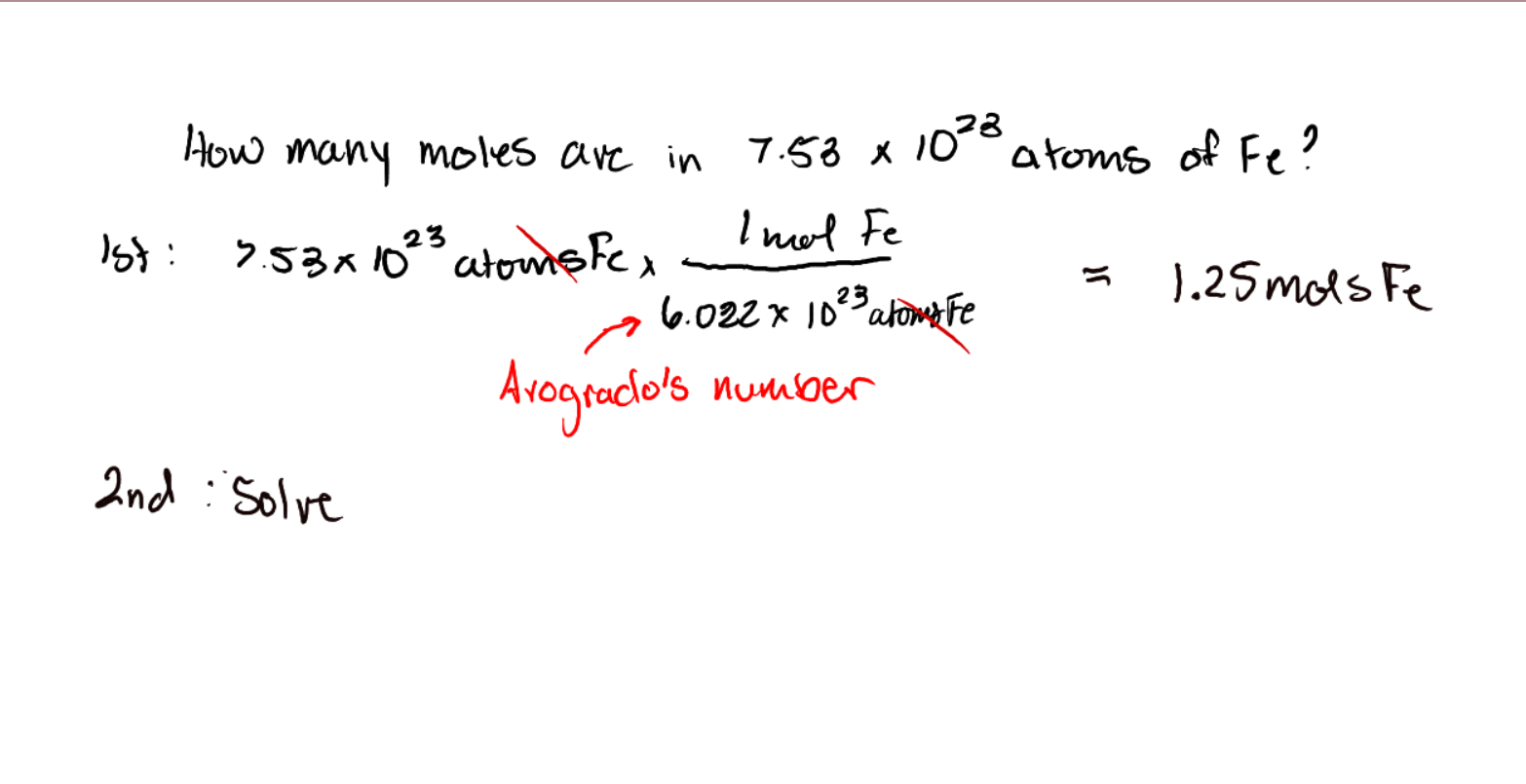
7

8
Fill in the Blanks
Type answer...
9
Fill in the Blanks
Type answer...
10
Mass (in grams) -> moles
the amount of mass given = 1 mole
11
Periodic Table
We use the periodic table in order to determine how much a compound weighs

12
Examples: Compound masses
CH4 = 16g (1 mole of methane)
H2O = 18g (1 mole of water)
CO2 = 44g (1 mole of carbon dioxide)
NH4 = 18g (1 mole of ammonia)
13
Explanation
When calculating the mass of a compound or substance one must look at the periodic table.
As you know CH4 has 1 Carbon atom and 4 Hydrogen atoms. If you look at the periodic table, you will see that Carbon's relative atomic mass is 12 and Hydrogen's relative atomic mass is 1.
Since there is only one carbon atom that is 12 grams, and because there are 4 hydrogen atoms in CH4 that adds another 4 grams to the compound. Adding the carbon and hydrogens together would mean you are adding the masses as well; therefore your relative molar mass would be 16g.
14
Fill in the Blanks
Type answer...
15
Fill in the Blanks
Type answer...
16
Mole Ratio
the correlation between the number of reacting particles and utilizes the coefficients in balanced equations
17

18

19
Fill in the Blanks
Type answer...
20
Fill in the Blanks
Type answer...
Stoichiometry
the relationship between substances that are included in reactions and solutions. This includes conversions of different measurements in order to show and give accurate proportions in reactions.

Show answer
Auto Play
Slide 1 / 20
SLIDE
Similar Resources on Wayground

13 questions
Lewis dot diagram (for elements)
Presentation
•
10th Grade

14 questions
5.1 Temperature, Thermal Energy, and Heat
Presentation
•
10th Grade

16 questions
Physical and Chemical Properties of Matter
Presentation
•
9th Grade

17 questions
Rockets Part 2: How Rockets Move
Presentation
•
10th Grade

12 questions
Dimensional Analysis
Presentation
•
11th Grade

12 questions
Density: Area & Volume
Presentation
•
9th Grade

19 questions
Radioactive Decay
Presentation
•
10th Grade

19 questions
History of the Cell Theory
Presentation
•
10th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Mathematics

5 questions
A.EI.1-3 Quizizz Day 1
Quiz
•
9th - 12th Grade

5 questions
A.EI.1-3 Quizizz Day 2
Quiz
•
9th - 12th Grade

5 questions
A.EI.1-3 Quizizz Day 4
Quiz
•
9th - 12th Grade

5 questions
G.PC/DF Quizizz Day 2
Quiz
•
9th - 12th Grade

5 questions
A.F/ST Quizizz Day 5
Quiz
•
9th - 12th Grade

5 questions
G.PC/DF Quizizz Day 1
Quiz
•
9th - 12th Grade

5 questions
A.EI.1-3 Quizizz Day 3
Quiz
•
9th - 12th Grade

25 questions
Algebra 1 EOC Review
Quiz
•
8th - 10th Grade