

Middle School Chemistry Review
Presentation
•
Science
•
8th Grade
•
Hard
Joseph Anderson
FREE Resource
12 Slides • 31 Questions
1
Chemistry Review
2

3
Multiple Choice
4
Multiple Choice
5

6
Multiple Choice
What is the outcome of observing a chemical property?
A new substance is formed with different properties
the substance does not change
7
Multiple Choice
What effect does observing the physical properties of a substance have on the substance?
Measuring physical properties changes the chemical identity of the substance
Observing physical properties has no effect on the substance
It makes to substance develop a deep seeded distrust of humans
The substance will undergo an exothermic reaction
8
Multiple Choice
Choose the one that is a chemical property
Magnetism
Size
Color
Reactivity with water
9

10
Multiple Choice

11
Multiple Choice

Physical or Chemical Change? Iron rusts
Physical Change
Chemical Change
12
Multiple Choice

Water freezing into ice is an example of a...
Physical Change
Chemical Change
13

14
Multiple Choice
15
Multiple Choice
Which of the following is a negatively charged subatomic particle?
Electron
Proton
Neutron
Quark
16

17
Multiple Choice

18
Multiple Choice
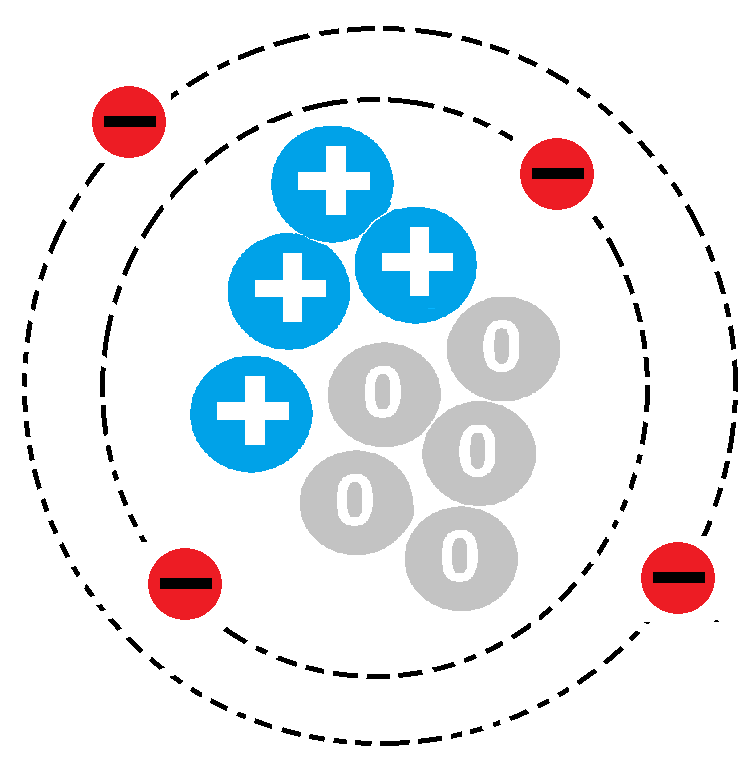
19

20
Multiple Choice
Which of the following properties uniquely identifies every element?
21
Multiple Choice
22
Multiple Choice
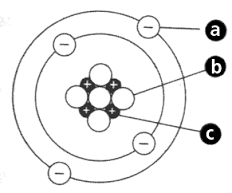
23

24
Multiple Choice

How many valence electrons does the element have?
1
2
3
4
25
Multiple Choice
What do the vertical columns on the periodic table tell us?
The number of orbitals
The number of valence electrons (electrons on the outer most shell)
26
Multiple Choice
What are the vertical columns on the periodic called?
groups/
families
periods
nonmetals
metals
metalloids
27
Multiple Choice
a row on the Periodic Table of Elements
family
period
row
isotope
28

29
Multiple Choice
Which elements are found on the left to middle of The Periodic Table and are shiny, good conductors, and malleable?
Metals
Nonmetals
Metalloids
Candles
30

31
Multiple Choice
What kind of substance is composed of two or more elements that are chemically combined?
element
mixture
solution
compound
32
Multiple Choice
How many different elements are in an amino acid with the formula C5H11NO2S?
5
20
18
4
33
Multiple Choice
How many atoms of Oxygen (O) are in each molecule of H2SO4?
1
4
2
7
34
Multiple Choice
Alanine is an amino acid with formula C3H7NO2. How many total atoms in all are in each molecule of alanine?
3
7
12
13
35

36
Multiple Choice
What does the number 4 represent in 4NH3?
Subscript
Superscript
Coefficient
37
Multiple Choice
What does the number 3 represent in 4NH3?
Subscript
Superscript
Coefficient
38
Multiple Select

Select all of the reactants in this chemical equation.
Hydrogen + Oxygen --> Water
H2 + O2 --> H2O
Hydrogen
H2
Oxygen
O2
Water
H2O
39
Multiple Choice

Select of the products in this chemical equation.
Hydrogen + Oxygen --> Water
H2 + O2 --> H2O
Hydrogen
H2
Oxygen
O2
Water
H2O
40

41
Multiple Choice
The Law of Conservation of Mass says that matter can not be __________ or __________.
created; destroyed
recycled; renewed
subtracted; added
joined; separated
42
Multiple Choice

Is this equation balanced?
Yes
No
43
Multiple Choice

WHY does this picture demonstrate the Law of Conservation of Mass?
the number of atoms of each element are the same on both sides
they start out separate and end up combined
the same elements are used
it doesn't demonstrate the Law of Conservation of Mass
Chemistry Review
Show answer
Auto Play
Slide 1 / 43
SLIDE
Similar Resources on Wayground

36 questions
Ecological Relationships
Presentation
•
7th Grade

37 questions
Physical and Chemical Properties and Changes
Presentation
•
8th Grade

35 questions
HR Diagram and The Sun
Presentation
•
8th Grade

39 questions
Earth and Solar Systen: Phenomena
Presentation
•
8th Grade

38 questions
Transferring Thermal Energy
Presentation
•
8th - 9th Grade

37 questions
Matter and Energy in Ecosystems
Presentation
•
7th Grade

37 questions
Properties and Atoms
Presentation
•
7th Grade

38 questions
Science 7 Year End Review - Unit C
Presentation
•
7th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

30 questions
GMAS Physical Science Review
Quiz
•
8th Grade

10 questions
Exploring the Rock Cycle
Interactive video
•
6th - 8th Grade

18 questions
Carbon Cycle
Quiz
•
8th Grade

10 questions
Exploring Earth's Seasons and Their Causes
Interactive video
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

10 questions
Evidence of Evolution
Presentation
•
6th - 8th Grade

20 questions
8th Grade Science NC EOG Review
Quiz
•
8th Grade