

Redox Reactions
Presentation
•
Chemistry
•
10th Grade - University
•
Hard
Joseph Anderson
FREE Resource
8 Slides • 18 Questions
1
Redox Reactions Review

2
3
Assigning Oxidation Numbers
4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
7
Multiple Choice
8
Multiple Choice
9
Multiple Choice
10
Multiple Choice
11
Identifying Reduction and Oxidation
LEO GER
12

13

14
Multiple Choice
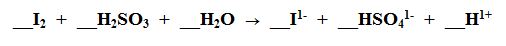
H
S
O
15
Multiple Choice
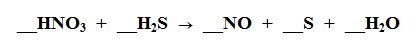
16
Multiple Choice
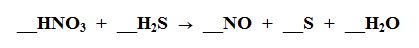
17
Multiple Choice
Sn(s) → Sn4+(aq) + 4e−
18
Multiple Choice

In any redox reaction, the substance that undergoes reduction will
lose electrons and have a decrease in oxidation number
lose electrons and have an increase in oxidation number
gain electrons and have a decrease in oxidation number
gain electrons and have an increase in oxidation number
19
Multiple Choice
Which reaction is an example of an oxidation-reduction reaction?
AgNO3 + KI --> AgI + KNO3
Cu + 2 AgNO3 --> Cu(NO3)2 + 2 Ag
2 KOH + H2SO4 -> K2SO4 + 2 H2O
Ba(OH)2 + 2 HCl --> BaCl2 + 2 H2O
20
Multiple Choice
Which half-reaction correctly represents reduction?
Ag --> Ag+ + e-
F2 --> 2 F- + 2e-
Au3+ + 3e- --> Au
Fe2+ + e- --> Fe3+
21
Balancing Redox Reactions
22
23
Multiple Choice
WRITE THIS OUT ON PAPER, YOU MAY USE THE ANSWER TO THIS PROBLEM AGAIN IN ANOTHER QUESTION:
Balance the following ACIDIC reactions using the half-reaction method.
MnO4−(aq) + H+(aq) + I− (aq) → Mn2+(aq) + I2(s)
Water is a product of this reaction. What is the coefficient of water in the final balanced equation?
8
5
2
16
24
Multiple Choice
WRITE THIS OUT ON PAPER, YOU MAY USE THE ANSWER TO THIS PROBLEM AGAIN IN ANOTHER QUESTION:
Balance the following reactions using the half-reaction method.
MnO4−(aq) + H+(aq) + I− (aq) → Mn2+(aq) + I2(s)
How many electrons did you cross out on both side?
1 e-
2 e-
5 e-
10 e-
25
Multiple Choice
WRITE THIS OUT ON PAPER, YOU MAY USE THE ANSWER TO THIS PROBLEM AGAIN IN ANOTHER QUESTION:
Consider the following REDOX reaction:
Cl2(g) → ClO−(aq) + Cl−(aq) (basic solution)
How many OH- molecules did you add to the reactant side of this reaction when balancing?
1
2
3
4
26
Multiple Choice
WRITE THIS OUT ON PAPER, YOU MAY USE THE ANSWER TO THIS PROBLEM AGAIN IN ANOTHER QUESTION:
Consider the following REDOX reaction:
Cl2(g) → ClO−(aq) + Cl−(aq) (basic solution)
Which species is the result of oxidation?
Cl−
ClO−
Redox Reactions Review

Show answer
Auto Play
Slide 1 / 26
SLIDE
Similar Resources on Wayground

21 questions
Chemistry Unit 9: Lesson 1 Solutions and Molarity
Presentation
•
10th - 12th Grade

20 questions
fast food
Presentation
•
KG

21 questions
Addition and Subtraction
Presentation
•
KG

22 questions
Accuracy v. Precision, Sig Figs, and Scientific Notation
Presentation
•
9th - 12th Grade

18 questions
Mole Conversions
Presentation
•
9th - 12th Grade

21 questions
NOBLE
Presentation
•
KG - University

20 questions
Periodic Trends
Presentation
•
10th - 12th Grade

20 questions
Types of Reactions
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Unit 7 Chemical Reactions
Quiz
•
10th Grade

25 questions
Unit 8 Stoichiometry Review
Quiz
•
10th Grade

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

15 questions
Nuclear Chemistry: Balancing
Quiz
•
10th Grade

20 questions
Naming Acids and Bases
Quiz
•
10th Grade

10 questions
Exploring Stoichiometry in Chemistry
Interactive video
•
6th - 10th Grade