

S P D F Block Elements Periodic Table
Presentation
•
Science
•
8th - 11th Grade
•
Hard
Joseph Anderson
FREE Resource
4 Slides • 30 Questions
1
Periodic Table/Element Block Review

2
Multiple Choice
3
Multiple Choice
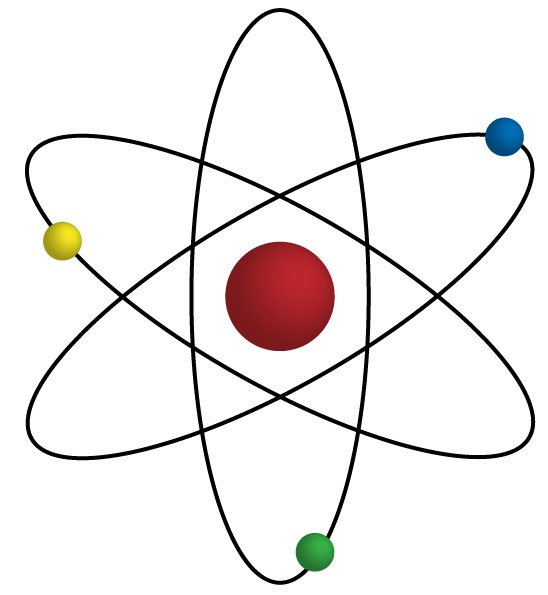
The central region of an atom where its neutrons and protons are is called the _____.
4
Multiple Choice
5
Multiple Choice
The particle that moves around the outside of nucleus is a(n)...
Proton
Neutron
Electron
Quark
6
Multiple Choice
7
Multiple Choice
True or False? Electrons have a negative charge.
8
Multiple Choice
9
Multiple Choice

10
Multiple Choice
11

12
Multiple Choice
Elements on the periodic table are organized by which of these?
symbols
atomic mass
state of matter
atomic number
13
Match

atomic number
mass number
chemical symbol
Always larger except for in hydrogen
Always smaller except for in hydrogen
Tells how many protons (and sometimes electrons) there are
tells how heavy the nucleus is (protons and neutrons)
abbreviation for an element's name
mass number
atomic number
Tells how many protons (and sometimes electrons) there are
tells how heavy the nucleus is (protons and neutrons)
abbreviation for an element's name
mass number
atomic number
14
Multiple Choice
True or false: 2 elements can have the same atomic number?
true
false
15

16
Multiple Choice
What does the atomic number tell about an atom?
the number of protons and electrons
the number of protons and neutrons
the number of neutrons
17
Multiple Choice

18
Multiple Choice
What does the mass number tell about an atom?
the number of protons
the number of protons and neutrons
the number of neutrons
the number of protons and electrons
19
Multiple Choice
20
Multiple Choice

21
Multiple Choice

What is the atomic mass of Fluorine?
22
Multiple Choice

What is the number of protons that the element in this image contain?
23
Multiple Choice
What is the atomic number for an element with three protons?
24
Multiple Choice

How many neutrons are in the atom potassium "K"??
7
2
39
20
25
Multiple Choice
An atom with atomic number 6 would have how many protons
26
Multiple Choice
If an atom has 10 protons, 10 neutrons, and 10 electrons, what is the mass of the atom?
27
Multiple Choice

How many neutrons would you expect the average gold atom to have given the information shown?
79
118
197
276
28

29
Multiple Choice

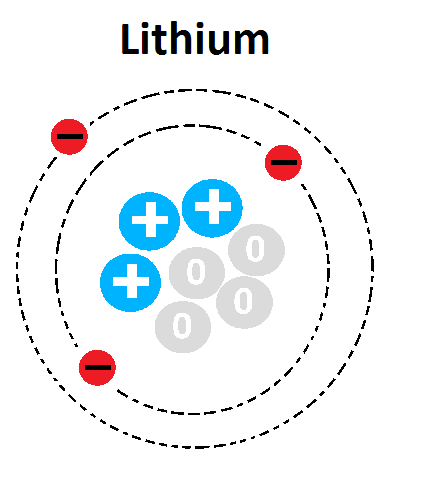
Lithium has the following atomic information:
3 protons / 3 electrons /4 neutrons - is this the correct Bohr model for lithium?
Yes
No
30
Multiple Choice

What element is this?
Neon - Element 10
Magnesium - Element 12
Sodium - Element 11
Aluminum - Element 13
31
Multiple Choice

Which element is this?
Calcium - Element 20
Chlorine - Element 17
Silicon - Element 14
Potassium - E;ement 19
32
Multiple Choice

What is the atomic number of this element?
17
18
35
35.453
33
Multiple Choice

What element is this?
Nitrogen - Element 7
Chlorine - Element 17
Argon - Element 18
Phosphorous - Element 15
34
Multiple Choice

Which element is this?
Nickel - Element 28
Neon - Element 10
Sodium - Element 11
Nitrogen - Element 7
Periodic Table/Element Block Review

Show answer
Auto Play
Slide 1 / 34
SLIDE
Similar Resources on Wayground

31 questions
Chapter 19.2: Classifying Chemical Reactions
Presentation
•
9th - 11th Grade

27 questions
CK-12 1.4 Scientific Theory
Presentation
•
9th - 12th Grade

26 questions
Circuits and Symbols
Presentation
•
9th - 12th Grade

24 questions
Chemical Reactions - Balancing and Writing Chemical Equations
Presentation
•
9th - 12th Grade

28 questions
Citing Textual Evidence using R.A.C.E
Presentation
•
8th - 10th Grade

21 questions
Ions
Presentation
•
8th - 11th Grade

26 questions
Atoms and Ions
Presentation
•
8th - 10th Grade

24 questions
Ch. 4.3 - Distinguishing Among Atoms
Presentation
•
9th - 11th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

12 questions
PS.8.1.2, PS.8.1.3, PS.8.1.5 Review
Quiz
•
8th Grade

20 questions
Moon Phases and Eclipses
Quiz
•
6th - 8th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

22 questions
6th & 8th Grade Science Material SOL Review
Quiz
•
6th - 8th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade