

Types of Radiations
Presentation
•
Chemistry
•
9th - 10th Grade
•
Hard
Joseph Anderson
FREE Resource
8 Slides • 22 Questions
1
Alpha Beta and Gamma radiation
by Cameron Scott
2
In this lesson
You will be able to understand the differences between the three typpes of particles apha, beta and gamma
You will watch some videos and answer multiple choice questions related to the video
You will improve your understanding of different forms of radiation and how they are used
3
Some isotopes are unstable
As seen earlier on this means they can decay and change into another isotope or produce alpha, beta or gamma radiation.
This is known as radiactive decay
The time is takes for half an unstable isotope to decay is known as a half life
4
Multiple Choice
An isotope has
time it takes for half to break down
the same protons but differing number of neutrons in the nucleus
the same electrons but differing number of neutrons in the nucleus
Time it takes for it to double
5
Open Ended
Define the word decay in relation to teeth
6
Watch the clip
Multiple choice questions are to follow in relation to the clip
7
Multiple Choice
Aplhaparticles can be stopped very easily by
Lead
air
stop sign
paper
8
Multiple Choice
Aplha particles are affected by
Gravity field
Rain
magnetic field
electricity
9
Multiple Choice
Alpha particles bend right, beta particles....
bend left
bend right
go up
go down
10
Multiple Choice
Beta particles have a ________ charge
positive
neutral
negative
11
Multiple Choice
Gamma radiation can be stopped by
Aluminium
oxygen
lead
copper
12
Multiple Choice
Gamma radiation is
A type of particle
a wave of pure energy
type of band
13
Open Ended
Name two features of Gamma radiation
14
15
Now over to you
please complete the questions from 5.7 in Science books and email a photo of completion
16
Multiple Select
Select all the correct answers.
Unstable nuclei have........
Too many neutrons
Too many protons
Too many electrons
stable amount of everything
17
Multiple Choice
Unstable nuclei are
Stable
have too many electrons
radioactive
18
Multiple Choice
Alpha particles form
nothing
new element two places below in the periodic table
new element two places higher in the periodic table
19
Multiple Choice
When there are too many protons an ______ forms
electron
proton
neutron
positron
20
Multiple Choice
True or false the number of neutrons was on the y axis
True
False
21
This is a random process and we cannot predict the radiactive nuclei in a sample will decay at any given moment.
However the rate of radioactive decay follows a pattern. As a radiocative sample decays less and less of the original substance is left and the radiactivity drops
This is a half life. When radioactivity reaches one half of its orginal level, one half life has passed

22
Now time to put this information into practice
Following are a selection that you can take your time and select the correct answer.
Please don't rush and you can do this more than once to improve your result
23
Multiple Choice
What is Half-life?
The amount of time it takes for some of the nuclei in a sample of the isotope to decay
The amount of time it takes for half the electrons in a sample of the isotope to decay
The amount of time it takes for half the nuclei in a sample of the isotope to decay
the amount of time it takes to double the nuclei in a sample of the isotope to decay
24
Multiple Choice
If 10 mg of iodine 131 is given to a patient, how much is left after 24 days? The half-life of iodine-131 is 8 days.
1.25mg
1.25g
10g
10mg
25
Multiple Choice
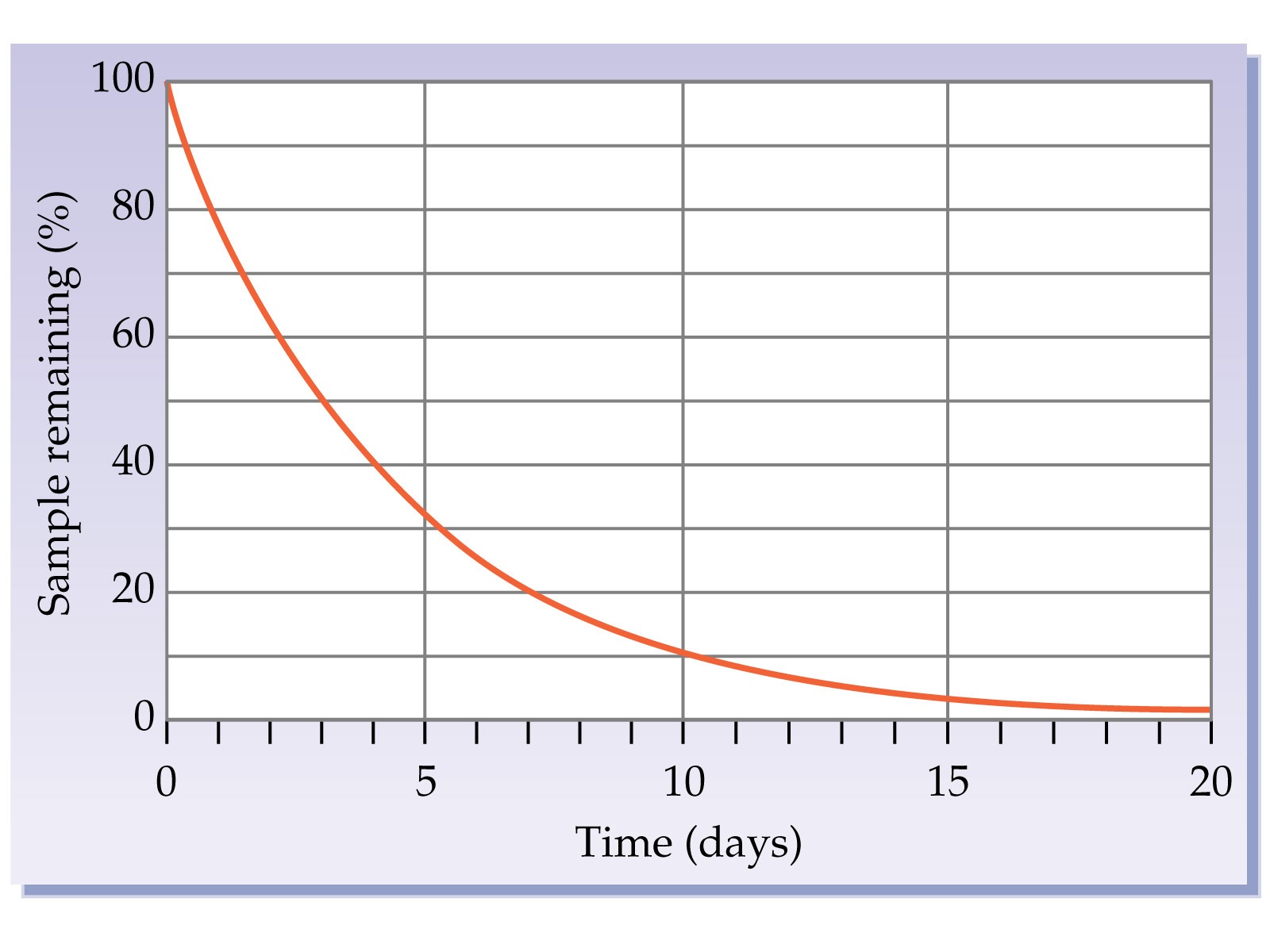
What is the Half Life of this isotope?
3 days
5 days
8 days
10 days
26
Multiple Choice

How many protons does this isotope of titanium have?
48
22
26
70
27
Multiple Choice

How many neutrons does the isotope of lithium have?
8
3
4
5
28
Multiple Choice
An element is defined by its number of
protons
electrons
neutrons
protons + electrons
29
Multiple Choice
An atom's atomic number is equal to
The number of protons in the atom's nucleus
The number of neutrons in the atom's nucleus
The number of electrons in the atom
The total of protons + neutrons
30
Multiple Choice

_______ are atoms with the same number of protons but varying numbers of neutrons.
ions
elements
allotropes
isotopes
Alpha Beta and Gamma radiation
by Cameron Scott
Show answer
Auto Play
Slide 1 / 30
SLIDE
Similar Resources on Wayground

22 questions
Arithmetic Sequences
Presentation
•
9th - 10th Grade

25 questions
Physical and Chemical Changes Review
Presentation
•
8th Grade

24 questions
Properties of Acids and Bases
Presentation
•
10th Grade

22 questions
Valence electrons and valency
Presentation
•
9th Grade

23 questions
Box and whisker
Presentation
•
9th - 10th Grade

23 questions
Sp. 3 Preterite vs. Imperfect Review
Presentation
•
9th - 10th Grade

24 questions
Asexual and sexual reproduction
Presentation
•
9th - 10th Grade

22 questions
Cell Cycle and Cancer Review
Presentation
•
9th - 10th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Unit 7 Chemical Reactions
Quiz
•
10th Grade

25 questions
Unit 8 Stoichiometry Review
Quiz
•
10th Grade

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

15 questions
Nuclear Chemistry: Balancing
Quiz
•
10th Grade

20 questions
Naming Acids and Bases
Quiz
•
10th Grade

10 questions
Exploring Stoichiometry in Chemistry
Interactive video
•
6th - 10th Grade